CN1068331C - 含喹啉氧基或吡啶氧基配位体的过渡金属催化剂体系 - Google Patents
含喹啉氧基或吡啶氧基配位体的过渡金属催化剂体系 Download PDFInfo
- Publication number
- CN1068331C CN1068331C CN96194004A CN96194004A CN1068331C CN 1068331 C CN1068331 C CN 1068331C CN 96194004 A CN96194004 A CN 96194004A CN 96194004 A CN96194004 A CN 96194004A CN 1068331 C CN1068331 C CN 1068331C
- Authority
- CN
- China
- Prior art keywords
- catalyzer
- catalyst
- catalyst system
- transition metal
- metal catalyst
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F10/00—Homopolymers and copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/18—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms
- B01J31/1805—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms the ligands containing nitrogen
- B01J31/181—Cyclic ligands, including e.g. non-condensed polycyclic ligands, comprising at least one complexing nitrogen atom as ring member, e.g. pyridine
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/18—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms
- B01J31/1805—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms the ligands containing nitrogen
- B01J31/181—Cyclic ligands, including e.g. non-condensed polycyclic ligands, comprising at least one complexing nitrogen atom as ring member, e.g. pyridine
- B01J31/1815—Cyclic ligands, including e.g. non-condensed polycyclic ligands, comprising at least one complexing nitrogen atom as ring member, e.g. pyridine with more than one complexing nitrogen atom, e.g. bipyridyl, 2-aminopyridine
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/18—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms
- B01J31/1805—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms the ligands containing nitrogen
- B01J31/181—Cyclic ligands, including e.g. non-condensed polycyclic ligands, comprising at least one complexing nitrogen atom as ring member, e.g. pyridine
- B01J31/1825—Ligands comprising condensed ring systems, e.g. acridine, carbazole
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/22—Organic complexes
- B01J31/2204—Organic complexes the ligands containing oxygen or sulfur as complexing atoms
- B01J31/2208—Oxygen, e.g. acetylacetonates
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/22—Organic complexes
- B01J31/2204—Organic complexes the ligands containing oxygen or sulfur as complexing atoms
- B01J31/2208—Oxygen, e.g. acetylacetonates
- B01J31/2226—Anionic ligands, i.e. the overall ligand carries at least one formal negative charge
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/22—Organic complexes
- B01J31/2282—Unsaturated compounds used as ligands
- B01J31/2295—Cyclic compounds, e.g. cyclopentadienyls
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F17/00—Metallocenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/003—Compounds containing elements of Groups 4 or 14 of the Periodic System without C-Metal linkages
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/28—Titanium compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F10/00—Homopolymers and copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F10/02—Ethene
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/40—Complexes comprising metals of Group IV (IVA or IVB) as the central metal
- B01J2531/46—Titanium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/40—Complexes comprising metals of Group IV (IVA or IVB) as the central metal
- B01J2531/48—Zirconium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/40—Complexes comprising metals of Group IV (IVA or IVB) as the central metal
- B01J2531/49—Hafnium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/50—Complexes comprising metals of Group V (VA or VB) as the central metal
- B01J2531/56—Vanadium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/50—Complexes comprising metals of Group V (VA or VB) as the central metal
- B01J2531/57—Niobium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/60—Complexes comprising metals of Group VI (VIA or VIB) as the central metal
- B01J2531/62—Chromium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/60—Complexes comprising metals of Group VI (VIA or VIB) as the central metal
- B01J2531/66—Tungsten
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/80—Complexes comprising metals of Group VIII as the central metal
- B01J2531/82—Metals of the platinum group
- B01J2531/828—Platinum
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/22—Organic complexes
- B01J31/2204—Organic complexes the ligands containing oxygen or sulfur as complexing atoms
- B01J31/226—Sulfur, e.g. thiocarbamates
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/24—Phosphines, i.e. phosphorus bonded to only carbon atoms, or to both carbon and hydrogen atoms, including e.g. sp2-hybridised phosphorus compounds such as phosphabenzene, phosphole or anionic phospholide ligands
- B01J31/2404—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F110/00—Homopolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F110/02—Ethene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F210/00—Copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F210/16—Copolymers of ethene with alpha-alkenes, e.g. EP rubbers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2420/00—Metallocene catalysts
- C08F2420/04—Cp or analog not bridged to a non-Cp X ancillary anionic donor
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
- C08F4/65912—Component covered by group C08F4/64 containing a transition metal-carbon bond in combination with an organoaluminium compound
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S526/00—Synthetic resins or natural rubbers -- part of the class 520 series
- Y10S526/943—Polymerization with metallocene catalysts
Abstract
公开了一种助催化剂和通式(1)的二齿吡啶过渡金属催化剂的催化剂组合物,式中Y为O、S、NR、(2)或(3)。R各独立地选自氢或C1-6烷基,M为钛、锆或铪;X各独立地选自卤素、C1-6烷基、C1-6烷氧基或(4);L为X、环戊二烯基、C1-6烷基取代的环戊二烯基、茚基、芴基或(5);R′各独立地选自R、C1-6烷氧基、C1-16芳基、卤素或CF3;n为1-4。
也公开了制备聚α-烯烃的方法,该方法包括用该催化剂组合物或具有下述通式(6)的催化剂的催化剂组合物聚合α-烯烃单体,式中Y、M、L、X和R′如前所定义。
Description
本发明涉及用于聚合α-烯烃的催化剂,特别涉及使用含吡啶或喹啉部分的二齿配位体的过渡金属催化剂进行的乙烯聚合。与US3,900,452中公开的反应产物不同,其中TiCl3 -吡啶盐中的钛形成一与杂环氮连接的非σ键,而本发明化合物含有的钛与氮形成一非σ键,同时又与氧原子形成一σ键,氧原子又与杂环连接。
直到最近,聚烯烃的生产主要还是使用常规的齐格勒催化体系。典型的齐格勒催化剂是由含过渡金属的化合物和一种或多种有机金属化合物组成的。例如聚乙烯是用诸如三氯化钛和氯化二乙基铝,或四氯化钛、三氯氧化钒与三乙基铝的混合物之类的齐格勒催化剂生产的。这些种类的催化剂比较便宜,但是它们的活性低,必须以高浓度使用。残留于聚合物中的催化剂使聚合物产生黄或灰色,而且紫外线稳定性和长期稳定性差;再者,含氯的残留物能腐蚀聚合物加工设备。因此时常需要从聚合物里清除催化剂残留物或在聚合物中加中和剂和稳定剂,以克服残留物的有害作用,这样就增加了生产成本。另外,用齐格勒催化剂产生分子量分布较宽的聚合物,这是在许多应用中(诸如注射模塑)不希望有的。在掺入α-烯烃共聚单体时,它们也是较差的,这使聚合物密度控制产生困难。要达到某一密度可能要求大量过量的共聚单体。许多高级α-烯烃(诸如1-辛烯),如果需要掺入,则只能掺入极低的量。
自从发现齐格勒催化体系以来,虽然已有了大量的改进,但这些催化剂现在已被近期发现的金属茂催化剂所代替。典型的金属茂催化剂是由具有一个或多个环戊二烯基环状配位体的过渡金属化合物组成的。当金属茂与和传统齐格勒催化剂一起使用的有机金属化合物(诸如烷基铝)一起使用时,其活性很低,但当与作为助催化剂的铝氧烷一起使用时,则有很高的活性。其活性一般很高而无须自聚合物中除去其残留物。再者,用它们生产的聚合物分子量很高并且分子量分布很窄。它们也能很好地掺入α-烯烃共聚单体。
然而,在较高的温度下,金属茂催化剂趋于生产分子量较低的聚合物。因此,它们能用于约在80-90℃下进行的乙烯气相和淤浆聚合,但一般在增加温度时它们不能很好地进行。在较高温度下于溶液中进行的乙烯聚合是理想的,因为它具有较大的灵活性以生产分子量范围和密度较宽同时又可以用各种不同种类的共聚单体的聚合物。溶液聚合可以生产能在许多不同应用中使用的聚合物。例如可生产用作食品包装隔离膜的高分子量和高密度聚乙烯(PE)薄膜,和韧性好和抗中强度大的低密度乙烯共聚物。
现已发现了作为α-烯烃聚合催化剂其活性非常好的新颖二齿吡啶过渡金属化合物;也已发现至今为止无疑是具有任何催化性能的二齿喹啉过渡金属化合物,这些化合物也是优良的α-烯烃聚合催化剂。这些催化剂能生产性质与用金属茂催化剂生产的聚合物非常接近的聚合物,即聚合物有窄的分子量分布并均匀引入共聚单体。
含有二齿吡啶基配位体的本发明过渡金属催化剂的通式如下:式中Y为O、S、NR、
R各独立地选自氢或C1-6烷基,R'各独立地选自R、C1-6烷氧基、C6-16芳基、卤素或CF3,M为钛、锆或铪,X各独立地选自氢、C1-6烷基、C1-6烷氧基或
L为X、环戊二烯基、C1-6烷基取代的环戊二烯基、茚基、芴基或m为0-4,n为1-4。式中,基团Y最好是氧,因为这样的化合物较容易制备。同理,基团R最好是甲基,R'最好是氢。基团L最好是卤素,更优选氯,因为这样的催化剂有较优越的性能,并且容易制备。同理,基团X最好是卤素,特别是氯;基团M最好是钛。
二齿吡啶配合物的制备在实施例中说明,但一般它们能通过将具有酸式质子的取代吡啶前体与具有式MX3L的化合物在HX清除剂存在下进行反应而制备。反应是化学计量的,优选清除剂的化学计量。适合的清除剂的实例包括比取代吡啶碱性更强的化合物,诸如三乙胺、吡啶、氢化钠和丁基锂。如果清除剂是比取代吡啶更强的碱,就可制取取代吡啶的盐,并以取代吡啶盐为原料。当反应优选在溶剂中进行时,反应物只要求有部分的溶解度。可以使用非质子溶剂,诸如四氢呋喃(THF)、乙醚、甲苯、或二甲苯,浓度可用约0.2-20wt%固体,优选约5-10wt%固体。反应的发生温度约为-78℃至室温左右。反应进行时有沉淀形成,产物可用甲苯、二氯甲烷、乙醚或类似的提取剂进行提取。
喹啉过渡金属催化剂的制取方法与吡啶过渡金属催化剂类似,只是以取代喹啉(诸如8-羟基喹啉)代替取代吡啶。也可在溶剂中使用丁基锂以制取8-羟基喹啉的锂盐,8-羟基喹啉的锂盐也可用作起始原料。
因为催化剂在正常情况下是与有机金属助催化剂结合使用的,所以最好是将催化剂溶于助催化剂也能溶解的溶剂中。例如,如果甲基铝氧烷(MAO)或聚甲基铝氧烷(PMAO)为助催化剂,那么可以使用甲苯、二甲苯、苯或乙苯作溶剂。优选的助催化剂是MAO,因为用它能得到高的活性和分子量分布较窄的聚合物。在聚合中使用的有机金属助催化剂与催化剂之摩尔比一般在0.01∶1-100,000∶1的范围,优选1∶1-10,000的范围。
可以替代的助催化剂是含非配位的惰性阴离子的酸式盐(见US5,064,802)。酸式盐一般是非亲核化合物,它是由庞大的配位体连接在硼原子或铝原子上组成的,诸如四(五氟苯基)硼酸锂、四(五氟苯基)铝酸锂、四(五氟苯基)硼酸苯胺翁和它们的混合物。当这些化合物与催化剂反应时,所得到的阴离子相信是与含金属的阳离子成弱配位的。酸式盐与催化剂的摩尔比可在约0.01∶1-约1000∶1的范围,但最好是约1∶1-10∶1。从催从剂和酸式盐制备活性催化剂体系的方法没有限制,但优选的方法是在-78℃至约150℃的温度范围将它们在惰性溶剂中混合;需要时也可在单体的存在下进行混合。酸式盐可与上述的有机金属助催化剂结合使用。
催化剂和助催化剂可置于诸如硅胶、矾土、硅石、氧化镁或二氧化钛之类的载体上使用,但因它们对聚合物有污染,所以载体不是优选的。然而根据所使用的方法也可以需要载体。例如在气相聚合法和淤浆聚合法中一般需要有载体,以便控制所生产的聚合物的颗粒大小和阻止反应器壁结垢。使用载体时是将催化剂和助催化剂溶于溶剂,然后例如用蒸发溶剂的方法将其沉淀在载体材料上。助催化剂也可沉积在载体上或与载体化催化剂分别导入反应器。
催化剂是以常规方法用于烯属烃单体的聚合中。不饱和单体(诸如苯乙烯)可用本发明的催化剂来聚合,但它特别适用于聚合诸如丙烯、1-丁烯、1-己烯、1-辛烯、特别是乙烯之类的α-烯烃。
催化剂也可用常规方法用于诸如乙烯、丙烯、1-丁烯、1-己烯、1-辛烯等的不饱和单体混合物的共聚合,乙烯和诸如1,3-丁二烯、1,4-己二烯、1,5-己二烯等的二烯烃的混合物的共聚合;乙烯和诸如降冰片烯、亚乙基降冰片烯、乙烯基降冰片烯、降冰片二烯等的不饱和单体的混合物的共聚合。
本发明的催化剂可在各种不同的聚合方法中使用。它们可用于液相聚合法(淤浆、溶液、悬浮液、本体相聚合或其结合),高压流体相或气相聚合法。这些方法可在系列或单级中使用。聚合反应区的压力可为约15psia至约50,000psia,温度可为约-78℃至约300℃。
实施例1
双(2-吡啶氧基)二氯化钛的合成
实施例2
(环戊二烯基)(2-吡啶氧基)二氯化钛的制备
于0℃下将2-羟基吡啶(0.002摩尔)和三乙胺(0.002摩尔)于50ml乙醚中的溶液加入0.002摩尔环戊二烯基三氯化钛于50ml乙醚中的溶液,并搅拌过夜。从乙醚滤液中回收产物。产物结构如下:
实施例3
喹啉氧基过渡金属催化剂制备通法
在-78℃下将各种8-羟基喹啉衍生物的锂盐(用丁基锂制备)甲苯浆液与相应的钛或锆化合物(四氯化钛、四氯化锆、环戊二烯基三氯化钛或环戊二烯基三氯化锆)混合,并在室温下搅拌过夜。用甲苯或二氯甲烷提取,从反应混合物回收配合物。制备8-喹啉氧基三氯化钛(Ⅲ)是在-78℃下将0.01摩尔8-羟基喹啉的锂盐于30ml甲苯中的浆液(用1.45克(0.01摩尔)羟基喹啉和MeLi制备)加入1.9克(0.01摩尔)TiCl4于20ml甲苯中的溶液,并在室温下搅拌过夜。分离出沉淀,用甲苯洗涤,以100ml CH2Cl2提取。除去CH2Cl2后,分离得到棕色微晶状固体0.7克。
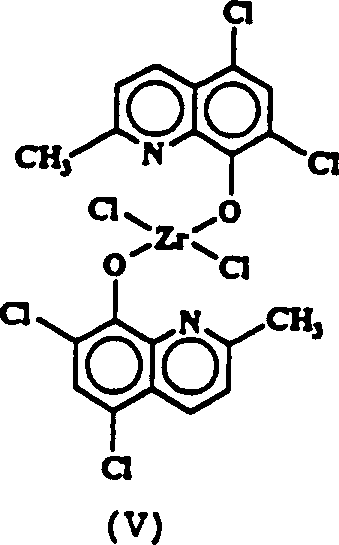
用类似的步骤由2.28克(0.01摩尔)5,7-二氯-2-甲基-8-羟基喹啉和1.165克(0.005摩尔)四氯化锆制备1.0克对比配合物双[8-(2-甲基-5,7-二氯喹啉)氧基]二氯化锆(Ⅴ)(环戊二烯基)(8-喹啉氧基)二氯化锆(Ⅵ)和(环戊二烯基)[8-(2-甲基-5,7-二氯喹啉)氧基]二氯化锆(Ⅶ)的制备是用由1.45克(0.01摩尔)8-羟基喹啉或1.15克(0.005摩尔)5,7-二氯-2-甲基-8-羟基喹啉制得的锂盐在-78℃下与等摩尔量的环戊二烯基三氯化锆在甲苯中进行反应。在搅拌过夜和过滤后,从甲苯溶液分离到产物(0.62克Ⅵ和1.7克Ⅶ)。
实施例4
聚合反应
在研究中所有的聚合反应均在一个1.7升的反应器中进行。在进行聚合反应前反应器在用氮气吹扫下加热至130℃并保持在该温度30分钟进行“烘烤”。将乙烯、氢、己烯、丁烯和氮通过含13X分子筛的柱进行处理。在一典型的聚合反应中,反应器中加入0.850升己烷或甲苯,并用注射器加入要求体积的稀PMAO(AKZO公司)。监测盛氢的1升高压不锈钢容器的压力降(Δp)将所需的氢加入反应器。用加压氮将催化剂的甲苯溶液加入反应器。在整个实验过程中反应器保持在等温条件下。在反应器中通入乙烯并通过调压器在需要时加入乙烯将压力控制在150psi。在反应器的温度和压力稳定后,将催化剂浆液加入反应器,聚合反应开始。用布洛克斯质流计(Brooks mass flow meter)监测乙烯流量。
使反应器放气终止聚合反应,聚合物经过滤而回收。加入约1000ppm丁基化羟甲苯/己烷(BHT)以稳定聚合物,并在80℃于真空炉中进一步脱挥发物2小时。按ASTM D-1238测定聚合物的熔体流动性。按ASTM D-150585在密度梯度柱中测压塑模制品的聚合物密度。
反应条件记录在下表中。
表中“Al/M”为催化剂的PMAO中的铝对金属(钛或锆)的摩尔比*对比实施例下表为聚合反应结果
| 实验号 | 催化剂 | 温度(℃) | 共聚单体 | 共聚单体(克) | 催化剂(毫摩尔) | 摩尔Al/M | H2,ΔP | 反应时间(分钟) |
| 1 | Ⅰ | 80 | 无 | 0 | 9.5E-3 | 1897 | 0 | 15 |
| 2 | Ⅰ | 80 | 无 | 0 | 4.7E-3 | 3795 | 0 | 15 |
| 3 | Ⅰ | 80 | 无 | 0 | 4.7E-3 | 1897 | 20 | 15 |
| 4 | Ⅰ | 80 | 无 | 0 | 4.7E-3 | 1897 | 50 | 10 |
| 5 | Ⅰ | 80 | 丁烯 | 20 | 4.7E-3 | 1897 | 20 | 15 |
| 6 | Ⅱ | 80 | 无 | 0 | 9.0E-3 | 1001 | 0 | 60 |
| 7 | Ⅲ | 80 | 丁烯 | 10 | 8.4E-3 | 1074 | 10 | 30 |
| 8 | Ⅲ | 80 | 无 | 10 | 8.4E-3 | 1074 | 10 | 30 |
| 9 | Ⅳ | 80 | 丁烯 | 0 | 6.6E-3 | 1324 | 0 | 30 |
| 10 | Ⅳ | 80 | 丁烯 | 10 | 6.6E-3 | 1324 | 10 | 30 |
| 11* | Ⅴ | 80 | 丁烯 | 10 | 1.14E-2 | 1175 | 0 | 10 |
| 12* | Ⅴ | 80 | 丁烯 | 10 | 4.06E-3 | 1645 | 0 | 10 |
| 13* | Ⅴ | 80 | 丁烯 | 10 | 8.12E-3 | 1645 | 0 | 10 |
| 14 | Ⅵ | 80 | 丁烯 | 10 | 6.74E-3 | 991 | 0 | 15 |
| 15 | Ⅵ | 80 | 丁烯 | 10 | 1.35E-2 | 991 | 0 | 15 |
| 16 | Ⅵ | 80 | 丁烯 | 10 | 1.35E-2 | 1288 | 5 | 15 |
| 17 | Ⅵ | 80 | 丁烯 | 10 | 1.89E-2 | 1132 | 15 | 15 |
| 18 | Ⅵ | 110 | 丁烯 | 10 | 1.89E-2 | 1132 | 0 | 15 |
| 19 | Ⅶ | 80 | 丁烯 | 10 | 1.10E-2 | 1212 | 0 | 15 |
| 20 | Ⅶ | 80 | 丁烯 | 10 | 1.10E-2 | 1212 | 5 | 15 |
| 21 | Ⅶ | 110 | 丁烯 | 10 | 1.54E-2 | 1126 | 15 | 15 |
| 22 | Ⅶ | 80 | 丁烯 | 10 | 1.54E-2 | 1126 | 0 | 15 |
| 23 | Ⅶ | 80 | 丁烯 | 10 | 1.54E-2 | 2078 | 0 | 15 |
| 实验号 | 催化剂产率(kg/gm/h) | MI2 | MI20 | MFR | 密度 | Mw/Mn |
| 1 | 179.0 | <0.01 | <0.01 | - | ||
| 2 | 153.2 | <0.01 | <0.01 | - | ||
| 3 | 165.5 | <0.01 | 1.8 | - | ||
| 4 | 133.1 | <0.01 | 2.47 | - | ||
| 5 | 272.9 | <0.01 | 0.964 | - | ||
| 6 | 62.9 | <0.01 | <0.01 | - | ||
| 7 | 99.2 | 0.90 | 16.9 | 18.9 | 0.9513 | 2.51 |
| 8 | 167.9 | 0.41 | 4.2 | 10.2 | 3.67 | |
| 9 | 103.2 | <0.01 | - | - | ||
| 10 | 24.8 | <0.01 | - | - | ||
| 11* | 低 | - | - | - | ||
| 12* | 无 | - | - | - | ||
| 13* | 无 | - | - | - | ||
| 14 | 无 | - | - | - | ||
| 15 | 98.1 | <0.01 | 1.15 | - | ||
| 16 | 177.7 | 0.93 | 21.4 | 23.1 | ||
| 17 | 137.6 | 1.31 | 34.5 | 26.3 | ||
| 18 | 159.0 | 0.81 | 16.1 | 19.8 | ||
| 19 | 119.8 | <0.01 | .83 | - | ||
| 20 | 198.7 | 0.46 | 11.4 | 24.7 | ||
| 21 | 157.8 | 0.63 | 17.8 | 28.2 | ||
| 22 | 160.1 | 0.06 | 12.8 | - | ||
| 23 | 112.1 | 0.06 | 1.84 | 29.7 |
*对比实施例表中的Kg/gM/h为每克催化剂每小时生产的聚合物千克数。聚合物的熔体指数根据ASTM D-1238条件E和条件F测定。MI2是用2.16公斤重量(条件E)测定的熔体指数。MI20是用21.6公斤重量(条件F)测定的熔体指数。MFR是MI20/MI2比。聚合物密度根据ASTM D-1505测定。聚合物分子量分布的测定使用Wa-ters 150C凝胶渗透色谱仪,温度为135℃,溶剂为1,2,4-三氯苯。用重均分子量(Mw)和Mw/Mn(数均分子量)比表示分子量分布特征。
用本发明的催化剂可得到好的生产率和高分子量聚合物,由其极低的MI值可证实,甚至在较高温度(110℃)下催化剂Ⅵ和Ⅶ也是如此。
Claims (6)
1.一种催化剂体系,包括:
(a)一种助催化剂,和
(b)一种催化剂,包括选自锆、钛和铪的过渡金属和配位到金属上的双齿配位体,其中该配位体是8-喹啉氧基或2-吡啶氧基配位体。
2.权利要求1的催化剂体系,其中助催化剂选自甲基铝氧烷(MAO)、聚甲基铝氧烷(PMAO)和非配位惰性阴离子的酸式盐。
3.权利要求1的催化剂体系,其中该配位体是被C1-6烷基、C1-6烷氧基、卤素或CF3取代的2-吡啶氧基和8-喹啉氧基。
4.载体化的权利要求1的催化剂体系。
5.在权利要求1的催化剂体系存在下聚合乙烯的方法。
6.在权利要求1的催化剂体系存在下聚合一种或多种线性或枝化α-烯烃的方法。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/423,232 US5637660A (en) | 1995-04-17 | 1995-04-17 | Polymerization of α-olefins with transition metal catalysts based on bidentate ligands containing pyridine or quinoline moiety |
| US08/423,232 | 1995-04-17 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1188481A CN1188481A (zh) | 1998-07-22 |
| CN1068331C true CN1068331C (zh) | 2001-07-11 |
Family
ID=23678124
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN96194004A Expired - Fee Related CN1068331C (zh) | 1995-04-17 | 1996-03-18 | 含喹啉氧基或吡啶氧基配位体的过渡金属催化剂体系 |
Country Status (12)
| Country | Link |
|---|---|
| US (3) | US5637660A (zh) |
| EP (2) | EP1059310B1 (zh) |
| JP (1) | JPH11503785A (zh) |
| KR (1) | KR19990007920A (zh) |
| CN (1) | CN1068331C (zh) |
| AU (1) | AU5314496A (zh) |
| BR (1) | BR9608224A (zh) |
| DE (2) | DE69635719T2 (zh) |
| ES (2) | ES2255914T3 (zh) |
| MX (1) | MX9707982A (zh) |
| RU (1) | RU2169735C2 (zh) |
| WO (1) | WO1996033202A2 (zh) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102781981A (zh) * | 2010-02-19 | 2012-11-14 | 陶氏环球技术有限责任公司 | 烯烃单体聚合方法及其催化剂 |
Families Citing this family (178)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5637660A (en) * | 1995-04-17 | 1997-06-10 | Lyondell Petrochemical Company | Polymerization of α-olefins with transition metal catalysts based on bidentate ligands containing pyridine or quinoline moiety |
| KR100453414B1 (ko) * | 1995-08-31 | 2005-04-14 | 스미또모 가가꾸 고오교오 가부시끼가이샤 | 전이금속착체,올레핀중합용촉매및올레핀중합체제조방법 |
| US5852146A (en) * | 1996-06-27 | 1998-12-22 | Union Carbide Chemicals & Plastics Technology Corporation | Catalyst for the production of olefin polymers |
| US5856258A (en) * | 1996-10-15 | 1999-01-05 | Northwestern University | Phenolate constrained geometry polymerization catalyst and method for preparing |
| US6432862B1 (en) * | 1996-12-17 | 2002-08-13 | E. I. Du Pont De Nemours And Company | Cobalt catalysts for the polymerization of olefins |
| US6214761B1 (en) * | 1996-12-17 | 2001-04-10 | E. I. Du Pont De Nemours And Company | Iron catalyst for the polymerization of olefins |
| US6417305B2 (en) * | 1996-12-17 | 2002-07-09 | E. I. Du Pont De Nemours And Company | Oligomerization of ethylene |
| US6451938B1 (en) | 1997-02-25 | 2002-09-17 | Exxon Mobil Chemical Patents Inc. | Polymerization catalyst system comprising heterocyclic fused cyclopentadienide ligands |
| TW420693B (en) * | 1997-04-25 | 2001-02-01 | Mitsui Chemicals Inc | Olefin polymerization catalysts, transition metal compounds, and <alpha>-olefin/conjugated diene copolymers |
| US6136748A (en) * | 1997-07-02 | 2000-10-24 | Union Carbide Chemicals & Plastics Technology Corporation | Catalyst composition for the polymerization of olefins |
| US6096676A (en) * | 1997-07-02 | 2000-08-01 | Union Carbide Chemicals & Plastics Technology Corporation | Catalyst for the production of olefin polymers |
| US6103657A (en) * | 1997-07-02 | 2000-08-15 | Union Carbide Chemicals & Plastics Technology Corporation | Catalyst for the production of olefin polymers |
| US6268447B1 (en) * | 1998-12-18 | 2001-07-31 | Univation Technologies, L.L.C. | Olefin polymerization catalyst |
| CN1067690C (zh) * | 1998-03-04 | 2001-06-27 | 中国科学院化学研究所 | 一种间规聚合的催化剂体系及其制备方法和用途 |
| GB9809207D0 (en) * | 1998-04-29 | 1998-07-01 | Bp Chem Int Ltd | Novel catalysts for olefin polymerisation |
| DE69935815T2 (de) | 1998-07-01 | 2007-12-27 | Exxonmobil Chemical Patents Inc., Baytown | Elastische Mischung mit Kristallpolymeren und kristallisierbaren Polymeren des Propens |
| US6333389B2 (en) * | 1998-12-18 | 2001-12-25 | Univation Technologies, Llc | Olefin polymerization catalysts, their production and use |
| US6303719B1 (en) | 1998-12-18 | 2001-10-16 | Univation Technologies | Olefin polymerization catalyst system |
| US6180552B1 (en) | 1999-04-07 | 2001-01-30 | Equistar Chemicals, L.P. | Transition metal complexes containing neutral, multidentate azacyclic ligands |
| US6204216B1 (en) | 1999-04-15 | 2001-03-20 | Equistar Chemicals, L.P. | Olefin polymerization catalysts containing amine derivatives |
| US6201076B1 (en) | 1999-04-29 | 2001-03-13 | Equistar Chemicals, L.P. | Olefin polymerization process with fatty amine additives for improved activity and reduced fouling |
| US6020493A (en) * | 1999-05-06 | 2000-02-01 | Equistar Chemicals, Lp | Single-site catalyst preparation |
| US6271325B1 (en) | 1999-05-17 | 2001-08-07 | Univation Technologies, Llc | Method of polymerization |
| US6211311B1 (en) * | 1999-05-25 | 2001-04-03 | Equistar Chemicals, L.P. | Supported olefin polymerization catalysts |
| US6291386B1 (en) * | 1999-05-25 | 2001-09-18 | Equistar Chemicals, Lp | Process for the in-situ preparation of single-site transition metal catalysts and polymerization process |
| US6239062B1 (en) | 1999-09-02 | 2001-05-29 | Equistar Chemicals, L.P. | Olefin polymerization catalysts containing indolyl-amido ligands |
| US6194527B1 (en) | 1999-09-22 | 2001-02-27 | Equistar Chemicals, L.P. | Process for making polyolefins |
| US6265504B1 (en) | 1999-09-22 | 2001-07-24 | Equistar Chemicals, Lp | Preparation of ultra-high-molecular-weight polyethylene |
| US6232260B1 (en) | 1999-10-14 | 2001-05-15 | Equistar Chemicals, L.P. | Single-site catalysts for olefin polymerization |
| US6228959B1 (en) | 1999-10-15 | 2001-05-08 | Equistar Chemicals, L.P. | Single-site catalysts containing homoaromatic ligands |
| US6300439B1 (en) | 1999-11-08 | 2001-10-09 | Univation Technologies, Llc | Group 15 containing transition metal catalyst compounds, catalyst systems and their use in a polymerization process |
| US6271323B1 (en) | 1999-10-28 | 2001-08-07 | Univation Technologies, Llc | Mixed catalyst compounds, catalyst systems and their use in a polymerization process |
| US6300438B1 (en) | 1999-10-22 | 2001-10-09 | Univation Technolgies, Llc | Hafnium transition metal catalyst compounds, catalyst systems and their use in a polymerization process |
| US6274684B1 (en) * | 1999-10-22 | 2001-08-14 | Univation Technologies, Llc | Catalyst composition, method of polymerization, and polymer therefrom |
| US6624107B2 (en) | 1999-10-22 | 2003-09-23 | Univation Technologies, Llc | Transition metal catalyst compounds having deuterium substituted ligand and catalyst systems thereof |
| US6265505B1 (en) | 1999-11-18 | 2001-07-24 | Univation Technologies, Llc | Catalyst system and its use in a polymerization process |
| US6417304B1 (en) | 1999-11-18 | 2002-07-09 | Univation Technologies, Llc | Method of polymerization and polymer produced therefrom |
| US6380328B1 (en) | 1999-12-10 | 2002-04-30 | Univation Technologies, Llc | Catalyst systems and their use in a polymerization process |
| CA2389587C (en) * | 1999-11-01 | 2010-05-25 | W.R. Grace & Co.-Conn. | Active, heterogeneous supported bi-or tri-dentate olefin polymerisation catalyst |
| US6294626B1 (en) | 1999-11-15 | 2001-09-25 | Equistar Chemicals, Lp | Olefin polymerization catalysts containing modified boraaryl ligands |
| US6281155B1 (en) | 1999-11-19 | 2001-08-28 | Equistar Chemicals, L.P. | Supported olefin polymerization catalysts |
| US6281306B1 (en) * | 1999-12-16 | 2001-08-28 | Univation Technologies, Llc | Method of polymerization |
| US6239239B1 (en) * | 2000-02-17 | 2001-05-29 | Equistar Chemicals, L.P. | Quinolinoxy and pyridinoxy single-site catalysts containing benzyl ligands |
| US6255415B1 (en) * | 2000-02-29 | 2001-07-03 | Equistar Chemicals, L.P. | Ethylene polymerization process |
| US6476165B1 (en) | 2000-03-08 | 2002-11-05 | Equistar Chemicals, Lp | Olefin polymerization process using fatty amine additives and boron-modified supported catalyst |
| US6498221B1 (en) * | 2000-03-30 | 2002-12-24 | Equistar Chemicals, Lp | Single-site catalysts containing chelating N-oxide ligands |
| US6812304B2 (en) | 2000-06-14 | 2004-11-02 | Equistar Chemicals, Lp | Process for producing improved premixed supported boraaryl catalysts |
| JP2004504420A (ja) * | 2000-07-17 | 2004-02-12 | ユニベーション・テクノロジーズ・エルエルシー | 触媒系及びその重合方法への使用 |
| US6433088B1 (en) | 2000-08-04 | 2002-08-13 | Equistar Chemicals, Lp | Clear and printable polypropylene films |
| US6486270B1 (en) | 2000-08-25 | 2002-11-26 | Equistar Chemicals, Lp | High molecular weight, medium density polyethylene |
| US6355733B1 (en) | 2000-10-13 | 2002-03-12 | Equistar Chemicals, Lp | Polyethylene blends and films |
| US6414099B1 (en) | 2000-10-18 | 2002-07-02 | Equistar Chemicals, Lp | Single-site catalysts based on caged diimide ligands |
| US6713577B2 (en) | 2000-11-07 | 2004-03-30 | Symyx Technologies, Inc. | Substituted pyridyl amine catalysts and processes for polymerizing and polymers |
| US6660678B1 (en) | 2000-11-21 | 2003-12-09 | Equistar Chemicals, Lp | Single-site catalysts for olefin polymerization |
| DE60118026T2 (de) * | 2000-12-06 | 2006-11-09 | Omlidon Technologies Llc, Los Altos | Schmelzverarbeitbares, verschleissfestes polyethylen |
| EP1700870B9 (en) | 2000-12-06 | 2009-09-02 | Eidgenössische Technische Hochschule Zürich | Melt-processible, wear resistant polyethylene |
| US6878662B2 (en) | 2000-12-28 | 2005-04-12 | Univation Technologies, Llc | Polymerization catalyst system, polymerization process and polymer therefrom |
| US6673882B2 (en) * | 2001-02-12 | 2004-01-06 | Equistar Chemicals, Lp | Supported single-site catalysts useful for olefin polymerization |
| US6664349B2 (en) | 2001-03-29 | 2003-12-16 | Equistar Chemicals, Lp | Ethylene polymerization process |
| US6579957B2 (en) | 2001-04-11 | 2003-06-17 | Equistar Chemicals, Lp | Single-site catalysts based on anionic thiopyran dioxide ligands |
| JP5156167B2 (ja) | 2001-04-12 | 2013-03-06 | エクソンモービル・ケミカル・パテンツ・インク | プロピレン−エチレンポリマー及び製造法 |
| US6391988B1 (en) | 2001-04-18 | 2002-05-21 | Equistar Chemicals L.P. | Tris(pyrazoyl) based anions |
| US6583240B2 (en) | 2001-05-23 | 2003-06-24 | Equistar Chemicals, Lp | Ethylene polymerization process |
| US6759361B2 (en) | 2001-06-04 | 2004-07-06 | Equistar Chemicals, Lp | Aluminoboronate activators for single-site olefin polymerization catalysts |
| US6489414B1 (en) | 2001-06-06 | 2002-12-03 | Equistar Chemicals, Lp | Chelating pyrimidines as ligands for single-site olefin polymerization catalysts |
| US6544918B1 (en) | 2001-07-17 | 2003-04-08 | Equistar Chemicals, Lp | Olefin polymerization catalysts containing chelating dianionic ligands |
| US6583242B2 (en) | 2001-08-02 | 2003-06-24 | Equistar Chemicals, Lp | Supported olefin polymerization catalysts |
| ATE412016T1 (de) * | 2001-09-14 | 2008-11-15 | Basell Polyolefine Gmbh | Verfahren zur polymerisation von olefinen |
| DE10145453A1 (de) * | 2001-09-14 | 2003-06-05 | Basell Polyolefine Gmbh | Monocyclopentadienylkomplexe mit einem kondensierten Heterocyclus |
| CN100357303C (zh) * | 2001-09-14 | 2007-12-26 | 住友化学工业株式会社 | 过渡金属络合物和烯烃聚合用催化剂以及用其制备烯烃聚合物的方法 |
| CN1160381C (zh) * | 2001-09-27 | 2004-08-04 | 中国石油化工股份有限公司 | 含双席夫碱配体的烯烃聚合催化剂及制备方法与应用 |
| US6960635B2 (en) * | 2001-11-06 | 2005-11-01 | Dow Global Technologies Inc. | Isotactic propylene copolymers, their preparation and use |
| US6906160B2 (en) | 2001-11-06 | 2005-06-14 | Dow Global Technologies Inc. | Isotactic propylene copolymer fibers, their preparation and use |
| EP1308450A3 (en) * | 2001-11-06 | 2003-10-01 | Symyx Technologies, Inc. | Titanium substituted pyridyl amine complexes, catalysts and processes for polymerizing ethylene and styrene |
| WO2003040201A1 (en) | 2001-11-06 | 2003-05-15 | Dow Global Technologies Inc. | Isotactic propylene copolymers, their preparation and use |
| US6927256B2 (en) | 2001-11-06 | 2005-08-09 | Dow Global Technologies Inc. | Crystallization of polypropylene using a semi-crystalline, branched or coupled nucleating agent |
| US6919467B2 (en) | 2001-12-18 | 2005-07-19 | Univation Technologies, Llc | Imino-amide catalyst compositions for the polymerization of olefins |
| US7001863B2 (en) | 2001-12-18 | 2006-02-21 | Univation Technologies, Llc | Monoamide based catalyst compositions for the polymerization of olefins |
| US6831187B2 (en) | 2001-12-18 | 2004-12-14 | Univation Technologies, Llc | Multimetallic catalyst compositions for the polymerization of olefins |
| US7199255B2 (en) | 2001-12-18 | 2007-04-03 | Univation Technologies, Llc | Imino-amide catalysts for olefin polymerization |
| US6864205B2 (en) | 2001-12-18 | 2005-03-08 | Univation Technologies, Llc | Heterocyclic-amide catalyst compositions for the polymerization of olefins |
| US6596826B1 (en) | 2001-12-20 | 2003-07-22 | Equistar Chemicals, Lp | Olefin polymerization catalysts containing 1,3-diboretanyl ligands |
| US6586545B1 (en) | 2001-12-20 | 2003-07-01 | Equistar Chemicals, Lp | Complexes based on four-membered cyclic anionic six-electron-donor ligands |
| US6635728B2 (en) * | 2002-01-10 | 2003-10-21 | Equistar Chemicals, Lp | Preparation of ultra-high-molecular-weight polyethylene |
| US6613841B2 (en) | 2002-01-28 | 2003-09-02 | Equistar Chemicals, Lp | Preparation of machine direction oriented polyethylene films |
| US6780807B2 (en) | 2002-02-04 | 2004-08-24 | Equistar Chemicals L.P. | Acyclic anionic six-electron-donor ancillary ligands |
| US6825296B2 (en) * | 2002-03-29 | 2004-11-30 | The University Of Hong Kong | Catalyst component for olefin polymerization |
| US6693157B2 (en) * | 2002-04-08 | 2004-02-17 | Equistar Chemicals, Lp | Olefin polymerization catalysts containing triquinane ligands |
| US6908972B2 (en) | 2002-04-16 | 2005-06-21 | Equistar Chemicals, Lp | Method for making polyolefins |
| US7094723B2 (en) * | 2002-04-18 | 2006-08-22 | Equistar Chemicals Lp | Catalysts containing at least one heterocyclic ligand for improving the catalysts' performance of olefin polymerization |
| US6642326B1 (en) | 2002-05-03 | 2003-11-04 | Equistar Chemicals, Lp | Use of silanes to enhance activity in single-site polymerizations |
| US6649698B1 (en) | 2002-05-17 | 2003-11-18 | Equistar Chemicals, Lp | Polyethylene blends |
| US6762255B2 (en) | 2002-06-06 | 2004-07-13 | Equistar Chemicals L.P. | Prealkylated olefin polymerization catalysts and olefin polymerization employing such catalysts |
| US6630547B1 (en) | 2002-06-11 | 2003-10-07 | Equistar Chemicals, Lp | Use of silanes to control molecular weight in olefin polymerizations |
| US7105672B2 (en) * | 2002-08-19 | 2006-09-12 | The University Of Hong Kong | Cyclometallated catalysts |
| US6765074B2 (en) | 2002-09-27 | 2004-07-20 | Equistar Chemicals, Lp | Olefin polymerization process |
| AU2003302033A1 (en) | 2002-10-15 | 2004-06-15 | Exxonmobil Chemical Patents Inc. | Multiple catalyst system for olefin polymerization and polymers produced therefrom |
| US7223822B2 (en) | 2002-10-15 | 2007-05-29 | Exxonmobil Chemical Patents Inc. | Multiple catalyst and reactor system for olefin polymerization and polymers produced therefrom |
| US6884749B2 (en) | 2002-10-17 | 2005-04-26 | Equistar Chemicals L.P. | Supported catalysts which reduce sheeting in olefin polymerization, process for the preparation and the use thereof |
| US6630548B1 (en) | 2002-11-01 | 2003-10-07 | Equistar Chemicals, Lp | Static reduction |
| US7579407B2 (en) * | 2002-11-05 | 2009-08-25 | Dow Global Technologies Inc. | Thermoplastic elastomer compositions |
| US7459500B2 (en) * | 2002-11-05 | 2008-12-02 | Dow Global Technologies Inc. | Thermoplastic elastomer compositions |
| US6716936B1 (en) | 2002-12-16 | 2004-04-06 | Equistar Chemicals L.P. | Cascaded boiling pool slurry reactors for producing bimodal low to medium density polyethylene polymers |
| US6713576B1 (en) | 2003-02-25 | 2004-03-30 | Equistar Chemicals, Lp | Olefin polymerization catalysts based on convex, polcyclic ligands |
| EP1611169B1 (en) | 2003-03-21 | 2010-06-02 | Dow Global Technologies Inc. | Morphology controlled olefin polymerization process |
| US20070029528A1 (en) * | 2003-04-04 | 2007-02-08 | San Diego State University Foundation | Compositions and methods for facilitating reaction at room temperature |
| US6710005B1 (en) * | 2003-04-10 | 2004-03-23 | Equistar Chemicals, Lp | Aluminoxane modification |
| US6984599B2 (en) * | 2003-04-23 | 2006-01-10 | Equistar Chemicals, Lp | Olefin polymerization catalysts based on hydroxyl-depleted calixarene ligands |
| US6774078B1 (en) | 2003-04-23 | 2004-08-10 | Equistar Chemicals, Lp | Olefin polymerization catalysts based on annulated cyclopentadienyl ligands |
| US6953764B2 (en) * | 2003-05-02 | 2005-10-11 | Dow Global Technologies Inc. | High activity olefin polymerization catalyst and process |
| US6995216B2 (en) * | 2003-06-16 | 2006-02-07 | Equistar Chemicals, Lp | Process for manufacturing single-site polyolefins |
| US6767975B1 (en) * | 2003-07-14 | 2004-07-27 | Equistar Chemicals, Lp | Olefin polymerization with pyridine moiety-containing singe-site catalysts |
| US6930156B2 (en) * | 2003-10-22 | 2005-08-16 | Equistar Chemicals, Lp | Polymer bound single-site catalysts |
| US20050148742A1 (en) * | 2004-01-02 | 2005-07-07 | Hagerty Robert O. | Method for controlling sheeting in gas phase reactors |
| US20070073012A1 (en) * | 2005-09-28 | 2007-03-29 | Pannell Richard B | Method for seed bed treatment before a polymerization reaction |
| US7985811B2 (en) * | 2004-01-02 | 2011-07-26 | Univation Technologies, Llc | Method for controlling sheeting in gas phase reactors |
| US7011892B2 (en) * | 2004-01-29 | 2006-03-14 | Equistar Chemicals, Lp | Preparation of polyethylene films |
| US20050200046A1 (en) * | 2004-03-10 | 2005-09-15 | Breese D. R. | Machine-direction oriented multilayer films |
| US7037987B2 (en) * | 2004-03-26 | 2006-05-02 | Rohn And Haas Company | Olefin polymerization catalyst and polymerization process |
| EP1732756B1 (en) * | 2004-04-05 | 2010-11-17 | Leucadia Inc | Degradable netting |
| US7598328B2 (en) * | 2004-04-07 | 2009-10-06 | Dow Global Technologies, Inc. | Supported catalysts for manufacture of polymers |
| US7175918B2 (en) * | 2004-04-27 | 2007-02-13 | Equistar Chemicals, Lp | Polyolefin compositions |
| WO2006009980A2 (en) * | 2004-06-21 | 2006-01-26 | Exxonmobil Chemical Patents Inc. | Olefin polymerisation process |
| US8440125B2 (en) * | 2004-06-28 | 2013-05-14 | Equistar Chemicals, Lp | Polyethylene films having high resistance to deformation or elongation |
| CA2473378A1 (en) * | 2004-07-08 | 2006-01-08 | Nova Chemicals Corporation | Novel borate activator |
| US7125939B2 (en) * | 2004-08-30 | 2006-10-24 | Equistar Chemicals, Lp | Olefin polymerization with polymer bound single-site catalysts |
| US7402546B2 (en) * | 2004-09-23 | 2008-07-22 | Equistar Chemicals, Lp | Magnesium chloride support |
| WO2006049699A1 (en) | 2004-10-29 | 2006-05-11 | Exxonmobil Chemical Patents Inc | Catalyst compound containing divalent tridentate ligand |
| WO2006066126A2 (en) * | 2004-12-16 | 2006-06-22 | Symyx Technologies, Inc. | Phenol-heterocyclic ligands, metal complexes, and their uses as catalysts |
| US8034461B2 (en) * | 2005-02-09 | 2011-10-11 | Equistar Chemicals, Lp | Preparation of multilayer polyethylene thin films |
| US20060177641A1 (en) * | 2005-02-09 | 2006-08-10 | Breese D R | Multilayer polyethylene thin films |
| WO2006099053A1 (en) * | 2005-03-09 | 2006-09-21 | Exxonmobil Chemical Patents Inc. | Methods for oligomerizing olefins |
| US7414006B2 (en) * | 2005-03-09 | 2008-08-19 | Exxonmobil Chemical Patents Inc. | Methods for oligomerizing olefins |
| US9469739B2 (en) | 2005-04-07 | 2016-10-18 | Aspen Aerogels, Inc. | Microporous polyolefin-based aerogels |
| US8461223B2 (en) * | 2005-04-07 | 2013-06-11 | Aspen Aerogels, Inc. | Microporous polycyclopentadiene-based aerogels |
| DE112006001733T5 (de) | 2005-07-01 | 2008-07-31 | Albemarle Corporation | Aluminoxanatsalzzusammensetzungen mit verbesserter Stabilität in aromatischen und aliphatischen Lösungsmitteln |
| US7232604B2 (en) * | 2005-07-28 | 2007-06-19 | Equistar Chemicals, Lp | Flame retardant crosslinkable compositions and articles |
| US7273914B2 (en) * | 2005-08-03 | 2007-09-25 | Equistar Chemicals, Lp | Olefin polymerization methods |
| US7091291B1 (en) | 2005-11-23 | 2006-08-15 | Equistar Chemicals, Lp | Olefin polymerization process |
| US7608327B2 (en) * | 2005-12-20 | 2009-10-27 | Equistar Chemicals, Lp | High tear strength film |
| EP1803747A1 (en) | 2005-12-30 | 2007-07-04 | Borealis Technology Oy | Surface-modified polymerization catalysts for the preparation of low-gel polyolefin films |
| WO2007092136A2 (en) * | 2006-02-03 | 2007-08-16 | Exxonmobil Chemical Patents, Inc. | Process for generating alpha olefin comonomers |
| US7982085B2 (en) * | 2006-02-03 | 2011-07-19 | Exxonmobil Chemical Patents Inc. | In-line process for generating comonomer |
| US8003839B2 (en) * | 2006-02-03 | 2011-08-23 | Exxonmobil Chemical Patents Inc. | Process for generating linear apha olefin comonomers |
| US8404915B2 (en) * | 2006-08-30 | 2013-03-26 | Exxonmobil Chemical Patents Inc. | Phosphine ligand-metal compositions, complexes, and catalysts for ethylene trimerizations |
| EP2104679A1 (en) * | 2007-01-08 | 2009-09-30 | ExxonMobil Chemical Patents Inc. | Methods for oligomerizing olefins with chromium pyridine mono-oxazoline catalysts |
| EP2114974A1 (en) * | 2007-01-08 | 2009-11-11 | ExxonMobil Chemical Patents Inc. | Methods for oligomerizing olefins with chromium pyridine ether catalysts |
| US20080182951A1 (en) * | 2007-01-08 | 2008-07-31 | Ackerman Lily J | Chromium Pyridine Bis(Oxazoline) And Related Catalysts For Ethylene Dimerization |
| EP2114973A1 (en) * | 2007-01-08 | 2009-11-11 | ExxonMobil Chemical Patents Inc. | Methods for oligomerizing olefins with chromium pyridine thioether catalysts |
| US20080163978A1 (en) * | 2007-01-09 | 2008-07-10 | Botros Maged G | Process for producing multi-layer structures having improved metal adhesion |
| US7794848B2 (en) * | 2007-01-25 | 2010-09-14 | Equistar Chemicals, Lp | MDO multilayer polyethylene film |
| BRPI0703586B1 (pt) | 2007-10-19 | 2018-02-06 | Braskem S.A | Catalisador metaloceno suportado, e, copolímeros de etileno com alfa-olefinas de alto e ultra alto peso molecular |
| US20090286944A1 (en) * | 2008-05-15 | 2009-11-19 | Symyx Technologies, Inc. | Select phenol-heterocycle ligands, metal complexes formed therefrom, and their uses as catalysts |
| KR101142117B1 (ko) * | 2008-09-25 | 2012-05-09 | 에스케이이노베이션 주식회사 | 전이금속 촉매계 및 이를 이용한 에틸렌 단독중합체 또는 에틸렌과 α-올레핀의 공중합체 제조방법 |
| KR101142122B1 (ko) * | 2008-09-30 | 2012-05-09 | 에스케이이노베이션 주식회사 | 새로운 전이금속 촉매계 및 이를 이용한 에틸렌 단독중합체또는 에틸렌과 α-올레핀의 공중합체 제조방법 |
| US20100120308A1 (en) * | 2008-11-13 | 2010-05-13 | Conwed Plastics Llc | Oxo-biodegradable netting |
| WO2010106105A2 (de) * | 2009-03-20 | 2010-09-23 | Basf Se | Biologisch abbaubares material aus einem polymer enthaltend poröses metallorganisches gerüstmaterial |
| WO2010110801A1 (en) | 2009-03-27 | 2010-09-30 | Exxonmobil Chemical Patents Inc. | Olefin oligomerization reaction processes exhibiting reduced fouling |
| US8153544B2 (en) * | 2009-07-22 | 2012-04-10 | Equistar Chemicals, Lp | Method for preparing non-metallocene catalysts |
| US8158733B2 (en) | 2009-07-22 | 2012-04-17 | Equistar Chemicals, Lp | Catalysts based on 2-(2-aryloxy)quinoline or 2-(2-aryloxy)dihydroquinoline ligands |
| US7858718B1 (en) * | 2009-07-22 | 2010-12-28 | Equistar Chemicals, Lp | Catalysts based on 2-aryl-8-anilinoquinoline ligands |
| CA2688217C (en) * | 2009-12-11 | 2016-07-12 | Nova Chemicals Corporation | Multi reactor process |
| EP2526129B1 (en) * | 2010-01-21 | 2017-09-27 | Saudi Basic Industries Corporation (Sabic) | Ethylene polymerisation process |
| CN101857651B (zh) * | 2010-06-12 | 2012-06-20 | 上海化工研究院 | 具有单活性中心的超高分子量聚乙烯催化剂的制备方法 |
| US20120016092A1 (en) | 2010-07-14 | 2012-01-19 | Sandor Nagy | Catalysts based on quinoline precursors |
| US20130202860A1 (en) | 2010-08-18 | 2013-08-08 | Huanyu Wei | Chlorine-free ink and coating compositions & a method for printing on untreated polyolefin films with improved adhesion |
| CN103380154B (zh) | 2010-11-22 | 2016-08-24 | 阿尔比马尔公司 | 活化剂组合物、其制备及其在催化中的用途 |
| EP2841471B1 (en) | 2012-04-27 | 2020-05-06 | W. R. Grace & Co. - Conn. | Activator compositions, their preparation, and their use in catalysts |
| WO2016042014A1 (en) * | 2014-09-17 | 2016-03-24 | Versalis S.P.A. | Pyridine complex of zirconium, catalytic system comprising said pyridine complex of zirconium and process of (co)polymerization of conjugated dienes |
| CA2864573C (en) | 2014-09-22 | 2021-07-06 | Nova Chemicals Corporation | Shrink film from single site catalyzed polyethylene |
| US10399052B2 (en) | 2014-11-25 | 2019-09-03 | Univation Technologies, Llc | Methods of controlling polyolefin melt index |
| CA2874895C (en) | 2014-12-16 | 2022-02-15 | Nova Chemicals Corporation | High modulus single-site lldpe |
| WO2017138783A1 (ko) * | 2016-02-12 | 2017-08-17 | 주식회사 엘지화학 | 신규한 리간드 화합물 및 전이금속 화합물 |
| KR101931234B1 (ko) | 2016-02-12 | 2018-12-21 | 주식회사 엘지화학 | 신규한 리간드 화합물 및 전이금속 화합물 |
| CN112646065B (zh) * | 2019-10-11 | 2022-11-18 | 中国石油化工股份有限公司 | 一种新型单茂过渡金属化合物、包含其的催化剂组合物与应用 |
| CN111747995B (zh) * | 2020-07-30 | 2023-02-21 | 上海化工研究院有限公司 | 一种含氮芳氧基茂钛化合物及其制备方法和应用 |
| WO2022232760A1 (en) | 2021-04-30 | 2022-11-03 | Exxonmobil Chemical Patents Inc. | Processes for transitioning between different polymerization catalysts in a polymerization reactor |
| CN116410224B (zh) * | 2023-06-05 | 2023-11-10 | 研峰科技(北京)有限公司 | 一种环戊二烯三氯化钛的合成工艺 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3900452A (en) * | 1963-06-07 | 1975-08-19 | Montedison Spa | Olefinic copolymers and process for the preparation thereof |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5149880A (en) * | 1990-01-16 | 1992-09-22 | The Texas A & M University System | Nitrogen-containing aromatic heterocyclic ligand-metal complexes and their use for the activation of hydrogen peroxide and dioxygen in the reaction of organic compounds |
| US5637660A (en) * | 1995-04-17 | 1997-06-10 | Lyondell Petrochemical Company | Polymerization of α-olefins with transition metal catalysts based on bidentate ligands containing pyridine or quinoline moiety |
| US5852146A (en) * | 1996-06-27 | 1998-12-22 | Union Carbide Chemicals & Plastics Technology Corporation | Catalyst for the production of olefin polymers |
-
1995
- 1995-04-17 US US08/423,232 patent/US5637660A/en not_active Expired - Lifetime
-
1996
- 1996-03-18 KR KR1019970707443A patent/KR19990007920A/ko not_active Application Discontinuation
- 1996-03-18 EP EP00110565A patent/EP1059310B1/en not_active Expired - Lifetime
- 1996-03-18 AU AU53144/96A patent/AU5314496A/en not_active Abandoned
- 1996-03-18 WO PCT/US1996/003656 patent/WO1996033202A2/en not_active Application Discontinuation
- 1996-03-18 ES ES00110565T patent/ES2255914T3/es not_active Expired - Lifetime
- 1996-03-18 BR BR9608224-0A patent/BR9608224A/pt not_active Application Discontinuation
- 1996-03-18 JP JP8531730A patent/JPH11503785A/ja not_active Ceased
- 1996-03-18 RU RU97117175/04A patent/RU2169735C2/ru active
- 1996-03-18 DE DE69635719T patent/DE69635719T2/de not_active Expired - Lifetime
- 1996-03-18 ES ES96909748T patent/ES2164878T3/es not_active Expired - Lifetime
- 1996-03-18 MX MX9707982A patent/MX9707982A/es not_active IP Right Cessation
- 1996-03-18 CN CN96194004A patent/CN1068331C/zh not_active Expired - Fee Related
- 1996-03-18 EP EP96909748A patent/EP0832089B1/en not_active Expired - Lifetime
- 1996-03-18 DE DE69615554T patent/DE69615554T2/de not_active Expired - Lifetime
-
1997
- 1997-06-10 US US08/872,659 patent/US6759493B1/en not_active Expired - Fee Related
-
2003
- 2003-06-30 US US10/610,212 patent/US6790918B2/en not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3900452A (en) * | 1963-06-07 | 1975-08-19 | Montedison Spa | Olefinic copolymers and process for the preparation thereof |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102781981A (zh) * | 2010-02-19 | 2012-11-14 | 陶氏环球技术有限责任公司 | 烯烃单体聚合方法及其催化剂 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPH11503785A (ja) | 1999-03-30 |
| EP0832089A2 (en) | 1998-04-01 |
| CN1188481A (zh) | 1998-07-22 |
| ES2255914T3 (es) | 2006-07-16 |
| WO1996033202A2 (en) | 1996-10-24 |
| DE69635719T2 (de) | 2006-09-28 |
| DE69615554T2 (de) | 2002-05-08 |
| EP1059310A3 (en) | 2004-08-04 |
| EP0832089B1 (en) | 2001-09-26 |
| US6759493B1 (en) | 2004-07-06 |
| RU2169735C2 (ru) | 2001-06-27 |
| EP1059310B1 (en) | 2006-01-11 |
| US6790918B2 (en) | 2004-09-14 |
| DE69635719D1 (de) | 2006-04-06 |
| EP1059310A2 (en) | 2000-12-13 |
| US20040097670A1 (en) | 2004-05-20 |
| WO1996033202A3 (en) | 1996-11-28 |
| DE69615554D1 (de) | 2001-10-31 |
| AU5314496A (en) | 1996-11-07 |
| US5637660A (en) | 1997-06-10 |
| ES2164878T3 (es) | 2002-03-01 |
| MX9707982A (es) | 1998-02-28 |
| BR9608224A (pt) | 1999-11-30 |
| KR19990007920A (ko) | 1999-01-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1068331C (zh) | 含喹啉氧基或吡啶氧基配位体的过渡金属催化剂体系 | |
| US6528670B1 (en) | Bridged metallocenes for olefin copolymerization | |
| EP0491842B1 (en) | Monocyclopentadienyl transition metal olefin polymerization catalysts | |
| US20010020074A1 (en) | Polymerization catalyst systems and their use | |
| CN1185787A (zh) | 作为烯烃聚合催化剂的氮杂硼啉基金属配合物 | |
| WO1998034964A1 (en) | Group 5 transition metal compounds | |
| WO1998041530A1 (en) | Transition metal metallacyclopentadienyl compounds | |
| EP1417213A1 (en) | Method for making late transition metal catalysts for olefin polymerization | |
| CN115667325A (zh) | 杂化催化剂组合物、包含其的催化剂及它们的制备方法 | |
| US6100414A (en) | Cyclopentadienyl transition metal compounds useful as polymerization catalysts | |
| KR20130027316A (ko) | 담지 메탈로센 촉매의 제조방법 및 이를 이용하여 제조된 담지 메탈로센 촉매 | |
| CN105308059A (zh) | 催化剂组合物、其制备方法及使用其制备聚烯烃的方法 | |
| US5990033A (en) | Syndiotactic prochiral olefin polymerization using non-aromatic, anionic, dienyl group containing complexes | |
| CN107459592A (zh) | 改性硅胶载体和负载化茂金属催化剂及制备方法和茂金属催化剂体系 | |
| US6228959B1 (en) | Single-site catalysts containing homoaromatic ligands | |
| CA2218638C (en) | Polymerization of alpha-olefins with transition metal catalysts based on bidentate ligands containing pyridine or quinoline moiety | |
| US6228958B1 (en) | Azaborolinyl metal complexes as olefin polymerization catalysts | |
| US6294626B1 (en) | Olefin polymerization catalysts containing modified boraaryl ligands | |
| CA2219816C (en) | Azaborolinyl metal complexes as olefin polymerization catalysts | |
| MXPA00009362A (en) | Cyclopentadienyl transition metal compounds, as polymerization catalysts | |
| MXPA00002637A (en) | Cyclopentaphenanthreneyl metal complexes and polymerization process |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C17 | Cessation of patent right | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20010711 Termination date: 20110318 |