US9498633B2 - Percutaneous tibial nerve stimulator - Google Patents
Percutaneous tibial nerve stimulator Download PDFInfo
- Publication number
- US9498633B2 US9498633B2 US14/298,207 US201414298207A US9498633B2 US 9498633 B2 US9498633 B2 US 9498633B2 US 201414298207 A US201414298207 A US 201414298207A US 9498633 B2 US9498633 B2 US 9498633B2
- Authority
- US
- United States
- Prior art keywords
- unit
- neurostimulator unit
- neurostimulator
- electrode
- transcutaneous
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/372—Arrangements in connection with the implantation of stimulators
- A61N1/37211—Means for communicating with stimulators
- A61N1/37235—Aspects of the external programmer
- A61N1/37247—User interfaces, e.g. input or presentation means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/0404—Electrodes for external use
- A61N1/0408—Use-related aspects
- A61N1/0456—Specially adapted for transcutaneous electrical nerve stimulation [TENS]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/05—Electrodes for implantation or insertion into the body, e.g. heart electrode
- A61N1/0502—Skin piercing electrodes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/36014—External stimulators, e.g. with patch electrodes
- A61N1/36017—External stimulators, e.g. with patch electrodes with leads or electrodes penetrating the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/3605—Implantable neurostimulators for stimulating central or peripheral nerve system
- A61N1/3606—Implantable neurostimulators for stimulating central or peripheral nerve system adapted for a particular treatment
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/372—Arrangements in connection with the implantation of stimulators
- A61N1/37211—Means for communicating with stimulators
- A61N1/37252—Details of algorithms or data aspects of communication system, e.g. handshaking, transmitting specific data or segmenting data
-
- G—PHYSICS
- G06—COMPUTING; CALCULATING OR COUNTING
- G06Q—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR ADMINISTRATIVE, COMMERCIAL, FINANCIAL, MANAGERIAL OR SUPERVISORY PURPOSES; SYSTEMS OR METHODS SPECIALLY ADAPTED FOR ADMINISTRATIVE, COMMERCIAL, FINANCIAL, MANAGERIAL OR SUPERVISORY PURPOSES, NOT OTHERWISE PROVIDED FOR
- G06Q20/00—Payment architectures, schemes or protocols
- G06Q20/22—Payment schemes or models
-
- G—PHYSICS
- G06—COMPUTING; CALCULATING OR COUNTING
- G06Q—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR ADMINISTRATIVE, COMMERCIAL, FINANCIAL, MANAGERIAL OR SUPERVISORY PURPOSES; SYSTEMS OR METHODS SPECIALLY ADAPTED FOR ADMINISTRATIVE, COMMERCIAL, FINANCIAL, MANAGERIAL OR SUPERVISORY PURPOSES, NOT OTHERWISE PROVIDED FOR
- G06Q50/00—Systems or methods specially adapted for specific business sectors, e.g. utilities or tourism
- G06Q50/10—Services
- G06Q50/22—Social work
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16H—HEALTHCARE INFORMATICS, i.e. INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR THE HANDLING OR PROCESSING OF MEDICAL OR HEALTHCARE DATA
- G16H20/00—ICT specially adapted for therapies or health-improving plans, e.g. for handling prescriptions, for steering therapy or for monitoring patient compliance
- G16H20/30—ICT specially adapted for therapies or health-improving plans, e.g. for handling prescriptions, for steering therapy or for monitoring patient compliance relating to physical therapies or activities, e.g. physiotherapy, acupressure or exercising
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/02—Details
- A61N1/04—Electrodes
- A61N1/05—Electrodes for implantation or insertion into the body, e.g. heart electrode
- A61N1/0551—Spinal or peripheral nerve electrodes
Definitions
- This disclosure relates to the field of nerve stimulators. More particularly, this disclosure relates to a percutaneous tibial nerve stimulator device for treating urinary symptoms and a system for providing payment for use of the device on a per-treatment session basis.
- Nerve and muscle stimulation is used to treat a variety of medical conditions such as to reduce pain and to assist muscle and joint rehabilitation. Nerve stimulation has also been used for decades to control bladder function. Various approaches to bladder neuromodulation have been studied with stimulation of the pudendal, sacral, and tibial nerves found to be effective in controlling bladder function.
- Percutaneous tibial nerve stimulation is a uniquely effective therapy for treating overactive bladder (OAB) and the associated symptoms of frequency, urgency, and urinary urge incontinence. It is a minimally invasive neuromodulation delivery method performed by placing a percutaneous needle electrode in the proximity of the tibial nerve, i.e., the stimulation site. A pulse generator and a transcutaneous electrode are electrically coupled to the needle electrode for providing current pulses to the stimulation site by passing current from the transcutaneous electrode to the percutaneous electrode.
- OAB overactive bladder
- the system described in the '588 patent includes a pulse generator, a first lead wire electrically coupled to the pulse generator, a transcutaneous electrode electrically coupled to the first lead wire, a second lead wire electrically coupled to the pulse generator, and a percutaneous electrode needle electrically coupled to the second lead wire.
- the transcutaneous electrode is positioned distal from the selected stimulation site on the surface of the skin.
- the percutaneous electrode needle is inserted through the skin in proximity of the tibial nerve. Current pulses traverse the internal stimulation site by passing from the transcutaneous electrode to the internal percutaneous electrode needle.
- Uroplasty devices Another problem that exists with the Uroplasty devices is that the unit is bulky and requires two lengthy leads for connecting the electrodes to a hand-held controller unit.
- a nerve stimulation system including a neurostimulator unit having a pulse generator for electrically coupling to a percutaneous needle electrode and a transcutaneous electrode.
- the percutaneous needle electrode has a leading end for inserting adjacent a stimulation site of a patient and the transcutaneous electrode is configured to be applied to skin adjacent the stimulation site of the patient.
- a microcontroller is in communication with the pulse generator for monitoring a number of available treatment credits and activating the pulse generator, each available treatment credit corresponding to a treatment session and the pulse generator operable to be activated for performing the treatment session when the number of available treatment credits is at least one.
- the pulse generator generates current pulses that pass between the transcutaneous electrode and the percutaneous needle electrode for providing stimulation to the stimulation site.
- a computer system is operable to communicate with the microcontroller of the neurostimulator unit.
- the computer system is operable to receive a treatment credit request having purchase information including a number of treatment credits purchased, each treatment credit purchased corresponding to an available treatment credit, and transmitting the number of treatment credits purchased to the microcontroller of the neurostimulator unit.
- the computer system includes a customer interface for submitting the treatment credit request.
- the neurostimulator unit is preferably dimensioned and configured to be removeably attached to the transcutaneous electrode.
- each treatment credit purchased includes a unique serial number
- the microcontroller being operable to transmit usage information to the computer system for monitoring usage of the purchased treatment credits by the neurostimulator unit based on the unique serial numbers.
- the neurostimulator unit may be linked to a unique identification number and the purchase information of the treatment credit request includes the unique identification number for authorizing the transmission of the number of treatment credits purchased to the microcontroller of the neurostimulator unit.
- the treatment credit request may be transferred from the computer system to a portable digital storage device, the portable storage device connectable to the neurostimulator unit for transmitting the number of treatment credits purchased to the microcontroller of the neurostimulator unit.
- the computer system communicates with the microcontroller of the neurostimulator unit wirelessly through one of radio frequency, Bluetooth, infrared, and a wireless internet connection.
- the computer system may also communicate with the microcontroller of the neurostimulator unit through a universal serial bus connection.
- a nerve stimulation system includes a plurality of neurostimulator units.
- Each neurostimulator unit includes a unique identification number for identifying the neurostimulator unit, a pulse generator operable to be electrically coupled to a plurality of electrodes configured to be applied adjacent a stimulation site of a patient, and a microcontroller in communication with the pulse generator for monitoring a number of available treatment credits and activating the pulse generator, each available treatment credit corresponding to a treatment session and the pulse generator operable to be activated for performing the treatment session when the number of treatment credits available is at least one, the pulse generator generating current pulses during the treatment session that pass between the plurality of electrodes for providing stimulation to the stimulation site.
- a computer system receives a treatment credit request having purchase information including the unique identification number of one of the plurality of neurostimulator units and a number of treatment credits purchased, each treatment credit purchased corresponding to an available treatment credit, the computer system being operable to communicate with the microcontroller of the neurostimulator units for transmitting the purchase information to an appropriate neurostimulator unit based on the unique identification number included in the purchase information.
- each unique identification number is linked to at least one customer account and the computer system includes a customer interface for accessing the customer account and submitting the treatment credit request.
- a method for providing a reusable nerve stimulation system to treat incontinence includes the following steps: providing a computer system having a customer interface; providing a neurostimulator unit including: a pulse generator for electrically coupling to a transcutaneous electrode configured to be applied to skin adjacent the stimulation site of a patient and a percutaneous electrode having a leading end for inserting adjacent a stimulation site of a patient, and a microcontroller in communication with the pulse generator for monitoring a number of available treatment credits to the neurostimulator unit and for activating the pulse generator during a treatment session when the number of available treatment credits is at least one; receiving at the computer system a treatment credit request transmitted through the customer interface, each treatment credit request having purchase information including a number of treatment credits purchased, each treatment credit purchased corresponding to an available treatment credit; transmitting the number of treatment credits purchased to the microcontroller of the neurostimulator unit; and adjusting the number of treatment credits available to the neurostimulator unit based on the number of treatment credits purchased transmitted to
- the method further includes determining whether the number of available treatment credits is at least one; and activating the pulse generator based on a treatment session request when it is determined that the number of available treatment credits is at least one.
- the method further includes transmitting usage information from the microcontroller to the computer system, the usage information including a number of treatment sessions performed; monitoring the usage information of the neurostimulator unit through the computer system; and providing the usage information to a customer through a customer interface of the computer system.
- method further includes providing each treatment credit purchased with a unique serial number; monitoring usage information of each treatment credit purchased at the neurostimulator unit; transmitting the usage information from the neurostimulator unit to the computer system for each treatment credit purchased based on the unique serial number.
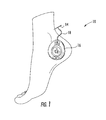
- FIG. 1 is a view of the tibial nerve stimulation device applied to the patient according to one embodiment of the disclosure
- FIG. 2 is a front view of a neurostimulator unit according to one embodiment of the disclosure
- FIG. 3 is a rear view of a neurostimulator unit according to one embodiment of the disclosure.
- FIG. 4 is a front view of a transcutaneous electrode patch according to one embodiment of the disclosure.
- FIG. 5 is a block diagram of the internal circuitry of a neurostimulator unit according to one embodiment of the disclosure.
- FIG. 6 is a view of a customer interface according to one embodiment of the disclosure.
- FIG. 7 is a flow chart of a method for performing treatment sessions using a neurostimulator unit according to one embodiment of the disclosure.
- a nerve stimulator device 10 includes a transcutaneous electrode pad (not shown), a percutaneous needle electrode 14 , a battery operated neurostimulator unit 16 having a pulse generator, and a single lead wire 18 for electrically coupling the needle electrode 14 to the neurostimulator unit 16 .
- the neurostimulator unit 16 includes dimensions of approximately 54 mm in length, approximately 46 mm in width, and a depth of approximately 14 mm and is configured to be removeably attached to the electrode pad which is adhesively applied to a patient's skin.
- the neurostimulator unit 16 is sized and configured for comfortable placement of the unit adjacent the heel or ankle area of the patient.
- the transcutaneous electrode pad 12 includes an attachment mechanism 20 on the top surface 13 of the pad 12 and the neurostimulator unit 16 includes a corresponding attachment mechanism 22 on the bottom surface 17 of the neurostimulator unit 16 for removeably attaching and electrically coupling the neurostimulator unit 16 with the electrode pad 12 .
- the neurostimulator unit 16 includes a female snap connector 22 disposed on the bottom surface 17 that connects to a corresponding male connector 20 disposed on the top surface 13 of the electrode pad 12 .
- a neurostimulator unit 16 having an integrated pulse generator and electrode pad 12 during operation of the stimulator device 10 , only a single lead 18 is needed to be included in the device 10 to connect the needle electrode 14 and provide stimulation to the stimulation site. No lead wire is needed to electrically couple the neurostimulator unit 16 and electrode pad 12 .
- the transcutaneous electrode pad 12 is positioned near a selected stimulation site on the surface of the skin.
- the percutaneous needle electrode 14 is then inserted through the skin adjacent the nerve or nerves to be stimulated, i.e., preferably adjacent the tibial nerve when treating incontinence.
- the neurostimulator unit 16 is attached to the transcutaneous electrode pad 12 and the lead wire 18 is connected to the neurostimulator unit 16 so that the pulse generator is electrically coupled to both the electrode pad 12 and the needle electrode 14 .
- the pulse generator is activated to perform a treatment session, current pulses traverse the stimulation site by passing from the transcutaneous electrode 12 to the internal percutaneous electrode needle 14 .
- the neurostimulator unit 16 includes a LCD display or other type of graphic display 30 for displaying operating information of the unit 16 such as the battery level, time remaining for a treatment session, signal strength of the pulse generator, treatment credits remaining for the unit 16 , and a network/computer connection status. Further aspects of the neurostimulator unit may include, for example, operating utilities such as a power button 32 for turning the unit 16 on and off, a toggle control 34 for adjusting the signal strength of the pulse generator, a start button 36 for beginning a treatment session, and a receiver 38 for receiving the lead wire 18 and electrically coupling the percutaneous electrode 14 to the neurostimulator unit 16 .
- operating utilities such as a power button 32 for turning the unit 16 on and off, a toggle control 34 for adjusting the signal strength of the pulse generator, a start button 36 for beginning a treatment session, and a receiver 38 for receiving the lead wire 18 and electrically coupling the percutaneous electrode 14 to the neurostimulator unit 16 .
- the graphic display 30 and/or some of the operating utilities may be either removable or permanently separate from the neurostimulator unit 16 to allow for handheld control of the device 10 .
- Such a design would provide for further compactness of the neurostimulator unit 16 .
- the handheld unit would then preferably communicate with the neurostimulator unit 16 wirelessly using an infrared, Bluetooth, wireless internet, or other means of wireless connection.
- the handheld unit may include a wired connection with the neurostimulator unit 16 .
- Other features of the handheld unit may include the ability to control multiple neurostimulator units 16 and the ability to time each treatment session and notify the patient or administrator with an audio alert at the end of the treatment session.
- the neurostimulator unit 16 is preferably connectable to a computer system for providing a treatment credit purchasing system and for monitoring the status and usage of the device 10 .
- the neurostimulator unit 16 is preferably operable to communicate with the computer system through a universal serial bus (USB) connector or wirelessly by using an infrared, Bluetooth, wireless internet, or other means of wireless connection.
- USB universal serial bus
- information may be transferred between the neurostimulator unit 16 and the computer system using a portable digital storage device such as a USB flash drive.
- the neurostimulator unit 16 includes a microcontroller or processor for storing information relating to the current status and past usage of the unit 16 and controlling operation of the device 10 .
- the status and usage information may be transferred from the unit 16 to the computer system when the unit 16 is connected to the computer system.
- the microcontroller monitors a number of treatment credits the neurostimulator unit has available to it. Each available treatment credit corresponds to a treatment session. After a treatment session is completed, the number of available treatment credits is decreased. If there are no more treatment credits available to the neurostimulator unit 16 , the microcontroller prevents the neurostimulator unit 16 from performing a treatment session by preventing activation of the pulse generator. In order for the unit 16 to be used in further treatment sessions, additional treatment credits must be purchased and transferred to the neurostimulator unit 16 .
- the neurostimulator unit 16 includes the graphic display 30 , power button 32 , toggle control 34 , start button 36 , pulse generator 40 , battery 42 , USB port 44 , and microcontroller 46 .
- the microcontroller 46 controls operation of the high voltage supply (DC to DC converter 54 ) and display of the operational characteristics on the graphic display 30 using a series of switches 48 , digital-to-analog converters 50 , analog-to-digital converters 52 , and a DC to DC converter 54 .
- a warning mechanism 56 such as a buzzer or light may be provided for notifying a patient or administrator that a treatment session is completed or a problem has occurred with the neurostimulator unit 16 .
- the USB port 44 is electrically isolated from the remaining components of unit 16 using an isolator 58 and isolated DC to DC converter 60 .
- Other aspects of the unit 16 include a power management chip 62 disposed between the battery 42 and microcontroller 46 , which uses power from the USB port 44 to charge the battery 44 connected through a fuse 64 disposed between the battery 42 and microcontroller 46 .
- a regulator 66 is provided for maintaining a constant supply voltage to the USB interface 68 when the USB port 44 is connected.
- the computer system includes a customer interface 70 for purchasing treatment credits to be used with an appropriate neurostimulator unit 16 .
- the customer could include the patient or a health care professional or office that administers treatment sessions to patients, and the terms user, customer, patient, and administrator may be used interchangeably herein.
- Each customer is set up with a customer account and the customer's neurostimulator unit or units 16 are then linked to this account.
- the customer interface 70 is preferably available to the customer through a web based application accessible from a desktop computer, laptop, tablet computer, smartphone, or the like.
- the customer has several options to choose from through the customer interface 70 such as downloading and purchasing treatment credits 72 , requesting a refund of treatment credits 74 , ordering supplies 76 such as electrode pads 12 and needle electrodes 14 , accessing account information 78 , requesting technical support 80 , and one or more links 82 that redirect the user to additional information on the web such as the manufacturer website.
- the microcontroller When the neurostimulator unit 16 is connected to the computer system, the microcontroller is updated, preferably automatically, based on the actions of the customer through the customer interface 70 . For example, if the user purchases a treatment credit using the customer interface 70 , the purchased treatment credit is transferred to the microcontroller of the neurostimulator unit 16 for providing the user with an additional treatment session when the unit 16 is connected to the computer system or a network connected to the computer system.
- a method for performing tibial nerve stimulation treatment sessions using the nerve stimulation device 10 is provided. It should be understood that the following steps are generally performed in no particular order and some steps may be omitted.
- the user or customer is provided with a nerve stimulation device 10 having a neurostimulator unit 16 as described substantially above.
- the nerve stimulation device 10 is provided to the customer at a reduced cost which is to be recouped by having the customer pay for using the device 10 on a per-treatment session basis.
- the customer signs up for a customer account on the computer system and links the customer's one or more neurostimulator units 16 with the customer account.
- each neurostimulator unit 16 includes a unique identification number and the neurostimulator unit 16 is linked to the customer account by providing the appropriate unique identification number.
- the customer After signing into the customer account, the customer is directed to the customer interface 70 and purchases one or more treatment credits in step 104 .
- Each purchased treatment credit preferably includes a unique serial number for assisting in tracking usage of the treatment credit.
- the neurostimulator unit 16 is connected to the computer system through a wireless or hardwire connection as described above for transferring information between the neurostimulator unit 16 and the computer system.
- the neurostimulator unit 16 transmits its unique identification number to the computer system and the computer system verifies that the unit 16 is linked to the customer account.
- the computer system transmits the one or more treatment credits purchased in step 104 to the neurostimulator unit 16 in step 110 .
- the information could be transferred between the computer system and the neurostimulator unit using a digital storage device such as a flash drive.
- information transferred to the computer system from the neurostimulator unit 16 could include a number of remaining treatment credits, if any, available to the unit 16 , the total number of treatment sessions provided by the unit 16 , the status of incomplete treatment sessions performed by the unit 16 , usage information of particular treatment credits based on the treatment credit's unique serial number, battery status, etc. This information can then be accessed from the customer interface 70 for monitoring the status and usage of the neurostimulator unit 16 when desired. For example, the usage information would be helpful when preparing bills to patients and processing insurance claims.
- step 112 the patient or administrator requests a treatment session to be performed using the neurostimulator unit 16 .
- the unit may or may not be connected to the computer system.
- step 114 the neurostimulator unit 16 determines whether a treatment credit is available. If it is determined in step 114 that there the unit 16 has no more available treatment credits, the patient or user must return to step 104 to purchase additional treatment credits before another treatment session may be performed. If a treatment credit is available, the nerve stimulation device 10 performs a treatment session using one of the treatment credits purchased in step 104 and transferred to the neurostimulator unit in step 116 .
- the unit 16 activates the pulse generator so that current pulses traverse the stimulation site during the treatment session by passing from the transcutaneous electrode 12 to the internal percutaneous electrode needle 14 .
- the number of available treatment credits is reduced by one. Step 112 is then repeated when another treatment session is requested.
Abstract
A method for treating incontinence includes positioning a transcutaneous electrode on skin of a patient adjacent a stimulation site; inserting a leading end of a percutaneous needle electrode through the skin adjacent the stimulation site, the percutaneous needle electrode including a lead wire disposed opposite the leading end; securing a neurostimulator unit to the transcutaneous electrode such that the neurostimulator unit is electrically coupled to the transcutaneous electrode with no lead wire disposed between the neurostimulator unit and the transcutaneous electrode, the neurostimulator unit including a pulse generator; connecting the lead wire of the percutaneous needle electrode to the neurostimulator unit for electrically coupling the needle electrode to the neurostimulator unit; and activating the pulse generator such that current pulses traverse the stimulation site by passing between the transcutaneous electrode pad and the percutaneous needle electrode.
Description
This application is a continuation of U.S. patent application Ser. No. 14/149,310 filed Jan. 7, 2014, which is a divisional to U.S. patent application Ser. No. 13/523,965 filed Jun. 15, 2012, which is now U.S. Pat. No. 8,660,646, which claimed priority to U.S. Provisional Application Ser. No. 61/497,570 filed Jun. 16, 2011, all of which being entitled “Percutaneous Tibial Nerve Stimulator” and each incorporated by reference herein in their entireties.
This disclosure relates to the field of nerve stimulators. More particularly, this disclosure relates to a percutaneous tibial nerve stimulator device for treating urinary symptoms and a system for providing payment for use of the device on a per-treatment session basis.
Nerve and muscle stimulation is used to treat a variety of medical conditions such as to reduce pain and to assist muscle and joint rehabilitation. Nerve stimulation has also been used for decades to control bladder function. Various approaches to bladder neuromodulation have been studied with stimulation of the pudendal, sacral, and tibial nerves found to be effective in controlling bladder function.
Percutaneous tibial nerve stimulation is a uniquely effective therapy for treating overactive bladder (OAB) and the associated symptoms of frequency, urgency, and urinary urge incontinence. It is a minimally invasive neuromodulation delivery method performed by placing a percutaneous needle electrode in the proximity of the tibial nerve, i.e., the stimulation site. A pulse generator and a transcutaneous electrode are electrically coupled to the needle electrode for providing current pulses to the stimulation site by passing current from the transcutaneous electrode to the percutaneous electrode.
One type of percutaneous tibial nerve stimulation system is described in U.S. Pat. No. 6,493,588 and is incorporated herein by reference. The system described in the '588 patent includes a pulse generator, a first lead wire electrically coupled to the pulse generator, a transcutaneous electrode electrically coupled to the first lead wire, a second lead wire electrically coupled to the pulse generator, and a percutaneous electrode needle electrically coupled to the second lead wire. During treatment, the transcutaneous electrode is positioned distal from the selected stimulation site on the surface of the skin. The percutaneous electrode needle is inserted through the skin in proximity of the tibial nerve. Current pulses traverse the internal stimulation site by passing from the transcutaneous electrode to the internal percutaneous electrode needle.
One problem with the system of the '588 patent is that the owners have had trouble implementing the system into a financially viable and profitable business model. In an attempt to solve this problem, the reusable lead wire system as described in the '588 patent was converted into a single use lead system as described, for example, in U.S. Pat. Nos. 7,536,226 and 8,046,082. While the advent of the single use lead system provides the ability to charge a patient/health care professional on a “per-treatment session” basis by requiring a new lead set to be used during each treatment session, the system requires perfectly operating leads to be wasted which leads to additional costs for the patient and manufacturer of the system. Further, the single use lead system is unable to determine whether a treatment session has been completed. Thus, if the treatment session was interrupted for any reason, the single use lead set would have to be replaced with a new lead set.
Another problem that exists with the Uroplasty devices is that the unit is bulky and requires two lengthy leads for connecting the electrodes to a hand-held controller unit.
What is needed therefore is a percutaneous tibial nerve stimulator system having reusable leads which provides the ability to charge a patient for each treatment session. In addition, a more compact and user friendly percutaneous tibial nerve stimulator system is desired.
A nerve stimulation system is disclosed including a neurostimulator unit having a pulse generator for electrically coupling to a percutaneous needle electrode and a transcutaneous electrode. The percutaneous needle electrode has a leading end for inserting adjacent a stimulation site of a patient and the transcutaneous electrode is configured to be applied to skin adjacent the stimulation site of the patient. A microcontroller is in communication with the pulse generator for monitoring a number of available treatment credits and activating the pulse generator, each available treatment credit corresponding to a treatment session and the pulse generator operable to be activated for performing the treatment session when the number of available treatment credits is at least one. During the treatment session, the pulse generator generates current pulses that pass between the transcutaneous electrode and the percutaneous needle electrode for providing stimulation to the stimulation site. A computer system is operable to communicate with the microcontroller of the neurostimulator unit. The computer system is operable to receive a treatment credit request having purchase information including a number of treatment credits purchased, each treatment credit purchased corresponding to an available treatment credit, and transmitting the number of treatment credits purchased to the microcontroller of the neurostimulator unit.
According to some embodiments, the computer system includes a customer interface for submitting the treatment credit request. The neurostimulator unit is preferably dimensioned and configured to be removeably attached to the transcutaneous electrode.
In certain embodiments, each treatment credit purchased includes a unique serial number, the microcontroller being operable to transmit usage information to the computer system for monitoring usage of the purchased treatment credits by the neurostimulator unit based on the unique serial numbers. The neurostimulator unit may be linked to a unique identification number and the purchase information of the treatment credit request includes the unique identification number for authorizing the transmission of the number of treatment credits purchased to the microcontroller of the neurostimulator unit.
According to some embodiments, the treatment credit request may be transferred from the computer system to a portable digital storage device, the portable storage device connectable to the neurostimulator unit for transmitting the number of treatment credits purchased to the microcontroller of the neurostimulator unit. In other embodiments, the computer system communicates with the microcontroller of the neurostimulator unit wirelessly through one of radio frequency, Bluetooth, infrared, and a wireless internet connection. The computer system may also communicate with the microcontroller of the neurostimulator unit through a universal serial bus connection.
According to another embodiment of the disclosure, a nerve stimulation system includes a plurality of neurostimulator units. Each neurostimulator unit includes a unique identification number for identifying the neurostimulator unit, a pulse generator operable to be electrically coupled to a plurality of electrodes configured to be applied adjacent a stimulation site of a patient, and a microcontroller in communication with the pulse generator for monitoring a number of available treatment credits and activating the pulse generator, each available treatment credit corresponding to a treatment session and the pulse generator operable to be activated for performing the treatment session when the number of treatment credits available is at least one, the pulse generator generating current pulses during the treatment session that pass between the plurality of electrodes for providing stimulation to the stimulation site. A computer system receives a treatment credit request having purchase information including the unique identification number of one of the plurality of neurostimulator units and a number of treatment credits purchased, each treatment credit purchased corresponding to an available treatment credit, the computer system being operable to communicate with the microcontroller of the neurostimulator units for transmitting the purchase information to an appropriate neurostimulator unit based on the unique identification number included in the purchase information.
According to some embodiments, each unique identification number is linked to at least one customer account and the computer system includes a customer interface for accessing the customer account and submitting the treatment credit request.
According to yet another embodiment of the disclosure, a method for providing a reusable nerve stimulation system to treat incontinence is disclosed. The method includes the following steps: providing a computer system having a customer interface; providing a neurostimulator unit including: a pulse generator for electrically coupling to a transcutaneous electrode configured to be applied to skin adjacent the stimulation site of a patient and a percutaneous electrode having a leading end for inserting adjacent a stimulation site of a patient, and a microcontroller in communication with the pulse generator for monitoring a number of available treatment credits to the neurostimulator unit and for activating the pulse generator during a treatment session when the number of available treatment credits is at least one; receiving at the computer system a treatment credit request transmitted through the customer interface, each treatment credit request having purchase information including a number of treatment credits purchased, each treatment credit purchased corresponding to an available treatment credit; transmitting the number of treatment credits purchased to the microcontroller of the neurostimulator unit; and adjusting the number of treatment credits available to the neurostimulator unit based on the number of treatment credits purchased transmitted to the microcontroller of the neurostimulator unit.
According to some embodiments, the method further includes determining whether the number of available treatment credits is at least one; and activating the pulse generator based on a treatment session request when it is determined that the number of available treatment credits is at least one.
In certain embodiments, the method further includes transmitting usage information from the microcontroller to the computer system, the usage information including a number of treatment sessions performed; monitoring the usage information of the neurostimulator unit through the computer system; and providing the usage information to a customer through a customer interface of the computer system.
According to some embodiments, method further includes providing each treatment credit purchased with a unique serial number; monitoring usage information of each treatment credit purchased at the neurostimulator unit; transmitting the usage information from the neurostimulator unit to the computer system for each treatment credit purchased based on the unique serial number.
Further advantages of the disclosure are apparent by reference to the detailed description in conjunction with the figures, wherein elements are not to scale so as to more clearly show the details, wherein like reference numbers indicate like elements throughout the several views, and wherein:
Referring to FIG. 1 , a nerve stimulator device 10 includes a transcutaneous electrode pad (not shown), a percutaneous needle electrode 14, a battery operated neurostimulator unit 16 having a pulse generator, and a single lead wire 18 for electrically coupling the needle electrode 14 to the neurostimulator unit 16. In preferred embodiments, the neurostimulator unit 16 includes dimensions of approximately 54 mm in length, approximately 46 mm in width, and a depth of approximately 14 mm and is configured to be removeably attached to the electrode pad which is adhesively applied to a patient's skin. In other words, the neurostimulator unit 16 is sized and configured for comfortable placement of the unit adjacent the heel or ankle area of the patient.
Referring to FIGS. 3-4 , the transcutaneous electrode pad 12 includes an attachment mechanism 20 on the top surface 13 of the pad 12 and the neurostimulator unit 16 includes a corresponding attachment mechanism 22 on the bottom surface 17 of the neurostimulator unit 16 for removeably attaching and electrically coupling the neurostimulator unit 16 with the electrode pad 12. In a preferred embodiment, the neurostimulator unit 16 includes a female snap connector 22 disposed on the bottom surface 17 that connects to a corresponding male connector 20 disposed on the top surface 13 of the electrode pad 12.
By providing a neurostimulator unit 16 having an integrated pulse generator and electrode pad 12 during operation of the stimulator device 10, only a single lead 18 is needed to be included in the device 10 to connect the needle electrode 14 and provide stimulation to the stimulation site. No lead wire is needed to electrically couple the neurostimulator unit 16 and electrode pad 12. This results in a compact system having very few parts. The entirety of the system is also able to be connected to the patient during treatment which provides the patient greater flexibility in movement during a treatment session. Further, as a hand held pulse generator is not being used, the single lead 18 only needs to be as long as the distance from where the electrode pad 12 is applied to the skin of the patient to the insertion site of the needle electrode 14.
In operation, the transcutaneous electrode pad 12 is positioned near a selected stimulation site on the surface of the skin. The percutaneous needle electrode 14 is then inserted through the skin adjacent the nerve or nerves to be stimulated, i.e., preferably adjacent the tibial nerve when treating incontinence. The neurostimulator unit 16 is attached to the transcutaneous electrode pad 12 and the lead wire 18 is connected to the neurostimulator unit 16 so that the pulse generator is electrically coupled to both the electrode pad 12 and the needle electrode 14. When the pulse generator is activated to perform a treatment session, current pulses traverse the stimulation site by passing from the transcutaneous electrode 12 to the internal percutaneous electrode needle 14.
Referring to FIG. 2 , the neurostimulator unit 16 includes a LCD display or other type of graphic display 30 for displaying operating information of the unit 16 such as the battery level, time remaining for a treatment session, signal strength of the pulse generator, treatment credits remaining for the unit 16, and a network/computer connection status. Further aspects of the neurostimulator unit may include, for example, operating utilities such as a power button 32 for turning the unit 16 on and off, a toggle control 34 for adjusting the signal strength of the pulse generator, a start button 36 for beginning a treatment session, and a receiver 38 for receiving the lead wire 18 and electrically coupling the percutaneous electrode 14 to the neurostimulator unit 16.
In alternate embodiments, the graphic display 30 and/or some of the operating utilities may be either removable or permanently separate from the neurostimulator unit 16 to allow for handheld control of the device 10. Such a design would provide for further compactness of the neurostimulator unit 16. The handheld unit would then preferably communicate with the neurostimulator unit 16 wirelessly using an infrared, Bluetooth, wireless internet, or other means of wireless connection. Alternatively, the handheld unit may include a wired connection with the neurostimulator unit 16. Other features of the handheld unit may include the ability to control multiple neurostimulator units 16 and the ability to time each treatment session and notify the patient or administrator with an audio alert at the end of the treatment session.
In another aspect of the disclosure, the neurostimulator unit 16 is preferably connectable to a computer system for providing a treatment credit purchasing system and for monitoring the status and usage of the device 10. The neurostimulator unit 16 is preferably operable to communicate with the computer system through a universal serial bus (USB) connector or wirelessly by using an infrared, Bluetooth, wireless internet, or other means of wireless connection. Alternatively, information may be transferred between the neurostimulator unit 16 and the computer system using a portable digital storage device such as a USB flash drive.
In these embodiments, the neurostimulator unit 16 includes a microcontroller or processor for storing information relating to the current status and past usage of the unit 16 and controlling operation of the device 10. The status and usage information may be transferred from the unit 16 to the computer system when the unit 16 is connected to the computer system. In particular, the microcontroller monitors a number of treatment credits the neurostimulator unit has available to it. Each available treatment credit corresponds to a treatment session. After a treatment session is completed, the number of available treatment credits is decreased. If there are no more treatment credits available to the neurostimulator unit 16, the microcontroller prevents the neurostimulator unit 16 from performing a treatment session by preventing activation of the pulse generator. In order for the unit 16 to be used in further treatment sessions, additional treatment credits must be purchased and transferred to the neurostimulator unit 16.
With reference to the block diagram of FIG. 5 , one embodiment of the circuitry of the neurostimulator unit 16 is disclosed. As described above, the neurostimulator unit includes the graphic display 30, power button 32, toggle control 34, start button 36, pulse generator 40, battery 42, USB port 44, and microcontroller 46. The microcontroller 46 controls operation of the high voltage supply (DC to DC converter 54) and display of the operational characteristics on the graphic display 30 using a series of switches 48, digital-to-analog converters 50, analog-to-digital converters 52, and a DC to DC converter 54. A warning mechanism 56 such as a buzzer or light may be provided for notifying a patient or administrator that a treatment session is completed or a problem has occurred with the neurostimulator unit 16.
As shown, it is preferred that the USB port 44 is electrically isolated from the remaining components of unit 16 using an isolator 58 and isolated DC to DC converter 60. Other aspects of the unit 16 include a power management chip 62 disposed between the battery 42 and microcontroller 46, which uses power from the USB port 44 to charge the battery 44 connected through a fuse 64 disposed between the battery 42 and microcontroller 46. A regulator 66 is provided for maintaining a constant supply voltage to the USB interface 68 when the USB port 44 is connected.
Referring to FIG. 6 , the computer system includes a customer interface 70 for purchasing treatment credits to be used with an appropriate neurostimulator unit 16. It should be understood that the customer could include the patient or a health care professional or office that administers treatment sessions to patients, and the terms user, customer, patient, and administrator may be used interchangeably herein. Each customer is set up with a customer account and the customer's neurostimulator unit or units 16 are then linked to this account. The customer interface 70 is preferably available to the customer through a web based application accessible from a desktop computer, laptop, tablet computer, smartphone, or the like.
As shown in FIG. 6 , the customer has several options to choose from through the customer interface 70 such as downloading and purchasing treatment credits 72, requesting a refund of treatment credits 74, ordering supplies 76 such as electrode pads 12 and needle electrodes 14, accessing account information 78, requesting technical support 80, and one or more links 82 that redirect the user to additional information on the web such as the manufacturer website.
When the neurostimulator unit 16 is connected to the computer system, the microcontroller is updated, preferably automatically, based on the actions of the customer through the customer interface 70. For example, if the user purchases a treatment credit using the customer interface 70, the purchased treatment credit is transferred to the microcontroller of the neurostimulator unit 16 for providing the user with an additional treatment session when the unit 16 is connected to the computer system or a network connected to the computer system.
Referring to FIG. 7 , a method for performing tibial nerve stimulation treatment sessions using the nerve stimulation device 10 is provided. It should be understood that the following steps are generally performed in no particular order and some steps may be omitted. In step 100, the user or customer is provided with a nerve stimulation device 10 having a neurostimulator unit 16 as described substantially above. Preferably, the nerve stimulation device 10 is provided to the customer at a reduced cost which is to be recouped by having the customer pay for using the device 10 on a per-treatment session basis. In step 102, the customer signs up for a customer account on the computer system and links the customer's one or more neurostimulator units 16 with the customer account. Preferably, each neurostimulator unit 16 includes a unique identification number and the neurostimulator unit 16 is linked to the customer account by providing the appropriate unique identification number.
After signing into the customer account, the customer is directed to the customer interface 70 and purchases one or more treatment credits in step 104. Each purchased treatment credit preferably includes a unique serial number for assisting in tracking usage of the treatment credit. In step 106, the neurostimulator unit 16 is connected to the computer system through a wireless or hardwire connection as described above for transferring information between the neurostimulator unit 16 and the computer system. In step 108, the neurostimulator unit 16 transmits its unique identification number to the computer system and the computer system verifies that the unit 16 is linked to the customer account.
After verification that the unit 16 is an authorized device for the particular customer account, the computer system transmits the one or more treatment credits purchased in step 104 to the neurostimulator unit 16 in step 110. Alternatively, the information could be transferred between the computer system and the neurostimulator unit using a digital storage device such as a flash drive. In addition to transmitting the purchased treatment credits to the neurostimulator unit 16, information transferred to the computer system from the neurostimulator unit 16 could include a number of remaining treatment credits, if any, available to the unit 16, the total number of treatment sessions provided by the unit 16, the status of incomplete treatment sessions performed by the unit 16, usage information of particular treatment credits based on the treatment credit's unique serial number, battery status, etc. This information can then be accessed from the customer interface 70 for monitoring the status and usage of the neurostimulator unit 16 when desired. For example, the usage information would be helpful when preparing bills to patients and processing insurance claims.
In step 112, the patient or administrator requests a treatment session to be performed using the neurostimulator unit 16. At this point, the unit may or may not be connected to the computer system. In step 114, the neurostimulator unit 16 determines whether a treatment credit is available. If it is determined in step 114 that there the unit 16 has no more available treatment credits, the patient or user must return to step 104 to purchase additional treatment credits before another treatment session may be performed. If a treatment credit is available, the nerve stimulation device 10 performs a treatment session using one of the treatment credits purchased in step 104 and transferred to the neurostimulator unit in step 116. In performing the treatment session, the unit 16 activates the pulse generator so that current pulses traverse the stimulation site during the treatment session by passing from the transcutaneous electrode 12 to the internal percutaneous electrode needle 14. After a treatment session is performed, the number of available treatment credits is reduced by one. Step 112 is then repeated when another treatment session is requested.
The foregoing description of preferred embodiments for this disclosure have been presented for purposes of illustration and description. They are not intended to be exhaustive or to limit the invention to the precise form disclosed. Obvious modifications or variations are possible in light of the above teachings. The embodiments are chosen and described in an effort to provide the best illustrations of the principles of the invention and its practical application, and to thereby enable one of ordinary skill in the art to utilize the invention in various embodiments and with various modifications as are suited to the particular use contemplated. All such modifications and variations are within the scope of the invention as determined by the appended claims when interpreted in accordance with the breadth to which they are fairly, legally, and equitably entitled.
Claims (16)
1. A method comprising:
applying an integrated neurostimulator unit and transcutaneous electrode pad to skin adjacent a stimulation site of a patient such that the electrode pad is disposed on an outer surface of the skin and there is no lead wire disposed between the neurostimulator unit and transcutaneous electrode pad, the neurostimulator unit including a pulse generator;
electrically coupling a percutaneous electrode having a leading end for inserting adjacent a tibial nerve of the patient to the neurostimulator unit;
inserting the leading end of the percutaneous electrode into the skin adjacent the tibial nerve of the patient; and
activating the pulse generator such that current pulses traverse the tibial nerve by passing between the transcutaneous electrode pad and the percutaneous electrode, the current pulses configured to treat incontinence.
2. The method of claim 1 wherein the transcutaneous electrode pad includes an attachment mechanism and the neurostimulator unit includes a corresponding attachment mechanism, wherein the applying step further includes electrically coupling the neurostimulator unit to the transcutaneous electrode by securing the neurostimulator unit attachment mechanism to the electrode pad attachment mechanism.
3. The method of claim 1 wherein the integrated neurostimulator unit and transcutaneous electrode pad is applied adjacent an ankle of the patient.
4. The method of claim 1 wherein the neurostimulator unit includes a display for displaying operating information.
5. The method of claim 1 further comprising:
providing a handheld unit operable to communicate with the neurostimulator unit; and
controlling operation of the neurostimulator unit with the handheld unit.
6. The method of claim 5 wherein the handheld unit is operable to communicate wirelessly with the neurostimulator unit.
7. The method of claim 1 further comprising:
providing a computer system operable to communicate with the neurostimulator unit; and
controlling operation of the neurostimulator unit through the computer system.
8. The method of claim 7 wherein the computer system is operable to communicate wirelessly with the neurostimulator unit.
9. A method comprising:
positioning a transcutaneous electrode on skin of a patient adjacent a tibial nerve;
inserting a leading end of a percutaneous needle electrode through the skin adjacent the tibial nerve, the percutaneous needle electrode including a lead wire disposed opposite the leading end;
securing a neurostimulator unit to the transcutaneous electrode such that the neurostimulator unit is electrically coupled to the transcutaneous electrode with no lead wire disposed between the neurostimulator unit and the transcutaneous electrode, the neurostimulator unit including a pulse generator;
connecting the lead wire of the percutaneous needle electrode to the neurostimulator unit for electrically coupling the needle electrode to the neurostimulator unit; and
activating the pulse generator such that current pulses traverse the tibial nerve by passing between the transcutaneous electrode pad and the percutaneous needle electrode, the current pulses configured to treat incontinence.
10. The method of claim 9 wherein the transcutaneous electrode includes an attachment mechanism and the neurostimulator unit includes a corresponding attachment mechanism, wherein the securing step further includes electrically coupling the neurostimulator unit to the transcutaneous electrode by securing the neurostimulator unit attachment mechanism to the transcutaneous electrode attachment mechanism.
11. The method of claim 9 wherein the transcutaneous electrode is positioned adjacent an ankle of the patient.
12. The method of claim 9 wherein the neurostimulator unit includes a display for displaying operating information.
13. The method of claim 9 further comprising:
providing a handheld unit operable to communicate with the neurostimulator unit; and
controlling operation of the neurostimulator unit with the handheld unit.
14. The method of claim 13 wherein the handheld unit is operable to communicate wirelessly with the neurostimulator unit.
15. The method of claim 9 further comprising:
providing a computer system operable to communicate with the neurostimulator unit; and
controlling operation of the neurostimulator unit through the computer system.
16. The method of claim 15 wherein the computer system is operable to communicate wirelessly with the neurostimulator unit.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/298,207 US9498633B2 (en) | 2011-06-16 | 2014-06-06 | Percutaneous tibial nerve stimulator |
| US14/453,844 US9076187B1 (en) | 2011-06-16 | 2014-08-07 | Medical device payment system |
| US14/694,704 US9245265B2 (en) | 2011-06-16 | 2015-04-23 | Medical device payment system |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201161497570P | 2011-06-16 | 2011-06-16 | |
| US13/523,965 US8660646B2 (en) | 2011-06-16 | 2012-06-15 | Percutaneous tibial nerve stimulator |
| US14/149,310 US8818520B2 (en) | 2011-06-16 | 2014-01-07 | Percutaneous tibial nerve stimulator |
| US14/298,207 US9498633B2 (en) | 2011-06-16 | 2014-06-06 | Percutaneous tibial nerve stimulator |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/149,310 Continuation US8818520B2 (en) | 2011-06-16 | 2014-01-07 | Percutaneous tibial nerve stimulator |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/453,844 Continuation-In-Part US9076187B1 (en) | 2011-06-16 | 2014-08-07 | Medical device payment system |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20140288613A1 US20140288613A1 (en) | 2014-09-25 |
| US9498633B2 true US9498633B2 (en) | 2016-11-22 |
Family
ID=47354292
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/523,965 Active US8660646B2 (en) | 2011-06-16 | 2012-06-15 | Percutaneous tibial nerve stimulator |
| US14/149,310 Active US8818520B2 (en) | 2011-06-16 | 2014-01-07 | Percutaneous tibial nerve stimulator |
| US14/298,207 Active US9498633B2 (en) | 2011-06-16 | 2014-06-06 | Percutaneous tibial nerve stimulator |
Family Applications Before (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/523,965 Active US8660646B2 (en) | 2011-06-16 | 2012-06-15 | Percutaneous tibial nerve stimulator |
| US14/149,310 Active US8818520B2 (en) | 2011-06-16 | 2014-01-07 | Percutaneous tibial nerve stimulator |
Country Status (6)
| Country | Link |
|---|---|
| US (3) | US8660646B2 (en) |
| EP (1) | EP2720749B1 (en) |
| JP (1) | JP2014527416A (en) |
| AU (1) | AU2012271480B2 (en) |
| CA (1) | CA2839532A1 (en) |
| WO (1) | WO2012174330A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10589089B2 (en) | 2017-10-25 | 2020-03-17 | Epineuron Technologies Inc. | Systems and methods for delivering neuroregenerative therapy |
| US11033737B2 (en) | 2014-08-17 | 2021-06-15 | Coloplast A/S | Miniature implantable neurostimulator system for sciatic nerves and their branches |
| US11247043B2 (en) | 2019-10-01 | 2022-02-15 | Epineuron Technologies Inc. | Electrode interface devices for delivery of neuroregenerative therapy |
| US11247045B2 (en) | 2017-10-25 | 2022-02-15 | Epineuron Technologies Inc. | Systems and methods for delivering neuroregenerative therapy |
| US11376429B2 (en) | 2012-04-26 | 2022-07-05 | Medtronic, Inc. | Trial stimulation systems |
Families Citing this family (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9037247B2 (en) | 2005-11-10 | 2015-05-19 | ElectroCore, LLC | Non-invasive treatment of bronchial constriction |
| US11351363B2 (en) | 2005-11-10 | 2022-06-07 | Electrocore, Inc. | Nerve stimulation devices and methods for treating cardiac arrhythmias |
| US11297445B2 (en) | 2005-11-10 | 2022-04-05 | Electrocore, Inc. | Methods and devices for treating primary headache |
| US11224742B2 (en) | 2006-10-02 | 2022-01-18 | Emkinetics, Inc. | Methods and devices for performing electrical stimulation to treat various conditions |
| US10786669B2 (en) | 2006-10-02 | 2020-09-29 | Emkinetics, Inc. | Method and apparatus for transdermal stimulation over the palmar and plantar surfaces |
| US11229790B2 (en) | 2013-01-15 | 2022-01-25 | Electrocore, Inc. | Mobile phone for treating a patient with seizures |
| US9375571B2 (en) | 2013-01-15 | 2016-06-28 | ElectroCore, LLC | Mobile phone using non-invasive nerve stimulation |
| US9254383B2 (en) | 2009-03-20 | 2016-02-09 | ElectroCore, LLC | Devices and methods for monitoring non-invasive vagus nerve stimulation |
| US10512769B2 (en) | 2009-03-20 | 2019-12-24 | Electrocore, Inc. | Non-invasive magnetic or electrical nerve stimulation to treat or prevent autism spectrum disorders and other disorders of psychological development |
| US9333347B2 (en) | 2010-08-19 | 2016-05-10 | ElectroCore, LLC | Devices and methods for non-invasive electrical stimulation and their use for vagal nerve stimulation on the neck of a patient |
| US10376696B2 (en) | 2009-03-20 | 2019-08-13 | Electrocore, Inc. | Medical self-treatment using non-invasive vagus nerve stimulation |
| US11400288B2 (en) | 2010-08-19 | 2022-08-02 | Electrocore, Inc | Devices and methods for electrical stimulation and their use for vagus nerve stimulation on the neck of a patient |
| US11191953B2 (en) | 2010-08-19 | 2021-12-07 | Electrocore, Inc. | Systems and methods for vagal nerve stimulation |
| US20200086108A1 (en) | 2010-08-19 | 2020-03-19 | Electrocore, Inc. | Vagal nerve stimulation to reduce inflammation associated with an aneurysm |
| US11432760B2 (en) | 2011-01-12 | 2022-09-06 | Electrocore, Inc. | Devices and methods for remote therapy and patient monitoring |
| US11511109B2 (en) | 2011-03-10 | 2022-11-29 | Electrocore, Inc. | Non-invasive magnetic or electrical nerve stimulation to treat gastroparesis, functional dyspepsia, and other functional gastrointestinal disorders |
| US11458297B2 (en) | 2011-03-10 | 2022-10-04 | Electrocore, Inc | Electrical and magnetic stimulators used to treat migraine/sinus headache, rhinitis, sinusitis, rhinosinusitis, and comorbid disorders |
| US9623253B2 (en) | 2012-07-17 | 2017-04-18 | Micron Devices, LLC | Devices and methods for treating urological disorders |
| WO2014153219A1 (en) * | 2013-03-14 | 2014-09-25 | Perryman Laura Tyler | Devices and methods for treating urological disorders |
| AU2013294692B2 (en) | 2012-07-26 | 2017-09-14 | Nyxoah SA | Insert tool for selectively powering an implant unit |
| WO2014040014A1 (en) * | 2012-09-10 | 2014-03-13 | Dermal Photonics Corporation | Systems and methods for usage replenishment |
| EP2928558B1 (en) | 2012-12-07 | 2017-09-06 | Medtronic Inc. | Minimally invasive implantable neurostimulation system |
| US10293160B2 (en) | 2013-01-15 | 2019-05-21 | Electrocore, Inc. | Mobile phone for treating a patient with dementia |
| US20160263376A1 (en) | 2013-11-27 | 2016-09-15 | The Governing Council Of The University Of Toronto | Systems and methods for improved treatment of overactive bladder |
| US11633593B2 (en) | 2013-11-27 | 2023-04-25 | Ebt Medical, Inc. | Treatment of pelvic floor disorders using targeted lower limb nerve stimulation |
| US10556107B2 (en) | 2013-11-27 | 2020-02-11 | Ebt Medical, Inc. | Systems, methods and kits for peripheral nerve stimulation |
| JP6594413B2 (en) * | 2014-05-20 | 2019-10-23 | エレクトロコア リミテッド ライアビリティ カンパニー | Non-invasive neural stimulation via portable devices |
| ES2936070T3 (en) | 2015-05-21 | 2023-03-14 | Ebt Medical Inc | Systems for the treatment of urinary dysfunction |
| CA3011993A1 (en) * | 2016-01-21 | 2017-08-03 | Cala Health, Inc. | Systems, methods and devices for peripheral neuromodulation for treating diseases related to overactive bladder |
| KR102583347B1 (en) * | 2016-05-31 | 2023-09-27 | 한국전자기술연구원 | Tibial Nerve Stimulation Signal Generation Device and Method |
| US11839762B2 (en) | 2016-10-05 | 2023-12-12 | Stimvia S.R.O. | Neuromodulation medical treatment device |
| PL3679979T3 (en) * | 2016-10-05 | 2021-12-27 | Tesla Medical S.R.O. | A device for neuromodulation treatment |
| US11896824B2 (en) | 2016-10-05 | 2024-02-13 | Stimvia S.R.O. | Method for neuromodulation treatment of low urinary tract dysfunction |
| US11638814B2 (en) | 2016-10-05 | 2023-05-02 | Tesla Medical S.R.O. | Method for neuromodulation treatment of neurodegenerative disease |
| US11590341B2 (en) | 2017-12-22 | 2023-02-28 | Electrocore, Inc | Vagal nerve stimulation therapy |
| US11446490B2 (en) | 2019-04-05 | 2022-09-20 | Tesla Medical S.R.O. | Method for a neuromodulation treatment |
| JP6573268B1 (en) * | 2019-05-15 | 2019-09-11 | 昌治 平野 | Compound wave therapy device |
| US11672991B2 (en) | 2019-07-17 | 2023-06-13 | Medtronic, Inc. | Minimally invasive neurostimulation device |
Citations (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5258906A (en) | 1988-07-13 | 1993-11-02 | Vital Heart Systems, Inc. | System for remotely authorizing operation of a device and for automatically generating an invoice based on device usage |
| US5458124A (en) | 1994-02-08 | 1995-10-17 | Stanko; Bruce E. | Electrocardiographic signal monitoring system |
| WO1999047204A1 (en) | 1998-03-18 | 1999-09-23 | Urosurge, Inc. | Electro-nerve stimulator systems and methods |
| US6049789A (en) | 1998-06-24 | 2000-04-11 | Mentor Graphics Corporation | Software pay per use licensing system |
| US20020120467A1 (en) | 2000-07-14 | 2002-08-29 | Asbjorn Buanes | Data processing system and method for managing the use of a medical device |
| US6662052B1 (en) | 2001-04-19 | 2003-12-09 | Nac Technologies Inc. | Method and system for neuromodulation therapy using external stimulator with wireless communication capabilites |
| US20040102819A1 (en) | 2002-11-20 | 2004-05-27 | Shazhou Zou | Portable therapeutic device and method for pain relief |
| US6829704B2 (en) | 2001-04-13 | 2004-12-07 | General Electric Company | Method and system to automatically activate software options upon initialization of a device |
| US20050165693A1 (en) | 2004-01-23 | 2005-07-28 | Klaus Moritzen | Prepaid licensing system and method |
| US6966000B2 (en) | 2001-04-13 | 2005-11-15 | Ge Medical Technology Services, Inc. | Method and system to remotely grant limited access to software options resident on a device |
| US6983375B2 (en) | 2001-04-13 | 2006-01-03 | Ge Medical Technology Services, Inc. | Method and system to grant indefinite use of software options resident on a device |
| US6990434B2 (en) | 2003-10-28 | 2006-01-24 | General Electric Company | System and method for coordinated remote activation of multiple software-based options |
| US7093032B2 (en) | 2003-10-28 | 2006-08-15 | General Electric Company | System and method for multi-vendor authentication to remotely activate a software-based option |
| US20060190048A1 (en) | 2005-02-23 | 2006-08-24 | Medtronic, Inc. | Implantable neurostimulator supporting trial and chronic modes |
| US7219222B1 (en) | 2002-01-18 | 2007-05-15 | Ge Medical Technology Services, Inc. | Method and system to grant access to software options resident on a medical imaging device |
| US20070167990A1 (en) * | 2006-01-17 | 2007-07-19 | Theranova, Llc | Method and apparatus for low frequency induction therapy for the treatment of urinary incontinence and overactive bladder |
| US20070167992A1 (en) | 2006-01-18 | 2007-07-19 | Baylor Research Institute | Method and apparatus for reducing preterm labor using neuromodulation |
| US7349856B2 (en) | 2001-07-30 | 2008-03-25 | Siemens Aktiengesellschaft | Method for selectively enabling or blocking the use of medical equipment |
| US20080091256A1 (en) * | 2006-10-11 | 2008-04-17 | Cardiac Pacemakers | Transcutaneous neurostimulator for modulating cardiovascular function |
| US20080140451A1 (en) | 2005-01-10 | 2008-06-12 | Cytori Therapeutics, Inc. | Devices and Methods for Monitoring, Managing, and Servicing Medical Devices |
| US20080306325A1 (en) | 2006-10-02 | 2008-12-11 | Emkinetics | Method and apparatus for magnetic induction therapy |
| US7536226B2 (en) | 2001-10-18 | 2009-05-19 | Uroplasty, Inc. | Electro-nerve stimulator system and methods |
| US20090132018A1 (en) | 2007-11-16 | 2009-05-21 | Ethicon, Inc. | Nerve Stimulation Patches And Methods For Stimulating Selected Nerves |
| US7571002B2 (en) | 2004-02-12 | 2009-08-04 | Ndi Medical, Llc | Portable percutaneous assemblies, systems and methods for providing highly selective functional or therapeutic neuromuscular stimulation |
| US20090248578A1 (en) | 2008-03-20 | 2009-10-01 | Liposonix, Inc. | Methods and apparatus for medical device investment recovery |
| US7634645B2 (en) | 2001-04-13 | 2009-12-15 | Ge Medical Technology Services, Inc. | Method and system to request remotely enabled access to inactive software options resident on a device |
| WO2010037471A1 (en) | 2008-10-01 | 2010-04-08 | Cardiola Ltd. | An apparatus for use on a person's lap |
| US20100114259A1 (en) * | 2006-05-18 | 2010-05-06 | Uroplasty, Inc. | Method and Apparatus for Stimulating a Nerve of a Patient |
| US20100168501A1 (en) | 2006-10-02 | 2010-07-01 | Daniel Rogers Burnett | Method and apparatus for magnetic induction therapy |
| US7813809B2 (en) | 2004-06-10 | 2010-10-12 | Medtronic, Inc. | Implantable pulse generator for providing functional and/or therapeutic stimulation of muscles and/or nerves and/or central nervous system tissue |
| US7894913B2 (en) | 2004-06-10 | 2011-02-22 | Medtronic Urinary Solutions, Inc. | Systems and methods of neuromodulation stimulation for the restoration of sexual function |
| US20110082524A1 (en) | 2009-10-05 | 2011-04-07 | Arp Manufacturing Llc | Electro-Therapeutic Stimulator |
| WO2011041526A2 (en) | 2009-10-01 | 2011-04-07 | University Of Pittsburgh-Of The Commonwealth System Of Higher Education | Non-invasive method to treat urological and gastrointestinal disorders |
| WO2011053607A1 (en) | 2009-10-26 | 2011-05-05 | Emkinetics, Inc. | Method and apparatus for electromagnetic stimulation of nerve, muscle, and body tissues |
| US7962416B1 (en) | 2000-11-22 | 2011-06-14 | Ge Medical Technology Services, Inc. | Method and system to remotely enable software-based options for a trial period |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2002214696A1 (en) * | 2000-10-25 | 2002-05-06 | Ryan Byleveld | Apparatus for dispensing electrotherapy treatment programs |
| US7065412B2 (en) * | 2003-04-25 | 2006-06-20 | Medtronic, Inc. | Implantable trial neurostimulation device |
| US20050245840A1 (en) | 2004-04-28 | 2005-11-03 | Medtronic, Inc. | Implantable urinary tract monitor |
| US8068918B2 (en) * | 2007-03-09 | 2011-11-29 | Enteromedics Inc. | Remote monitoring and control of implantable devices |
| US8204597B2 (en) * | 2007-05-30 | 2012-06-19 | Medtronic, Inc. | Evaluating patient incontinence |
| US9320908B2 (en) * | 2009-01-15 | 2016-04-26 | Autonomic Technologies, Inc. | Approval per use implanted neurostimulator |
| EP2478487B1 (en) * | 2009-09-14 | 2019-12-18 | Masimo Laboratories, Inc. | Spot check monitor credit system |
-
2012
- 2012-06-15 CA CA2839532A patent/CA2839532A1/en not_active Abandoned
- 2012-06-15 US US13/523,965 patent/US8660646B2/en active Active
- 2012-06-15 WO PCT/US2012/042585 patent/WO2012174330A1/en unknown
- 2012-06-15 AU AU2012271480A patent/AU2012271480B2/en not_active Ceased
- 2012-06-15 JP JP2014516011A patent/JP2014527416A/en active Pending
- 2012-06-15 EP EP12799895.3A patent/EP2720749B1/en active Active
-
2014
- 2014-01-07 US US14/149,310 patent/US8818520B2/en active Active
- 2014-06-06 US US14/298,207 patent/US9498633B2/en active Active
Patent Citations (49)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5258906A (en) | 1988-07-13 | 1993-11-02 | Vital Heart Systems, Inc. | System for remotely authorizing operation of a device and for automatically generating an invoice based on device usage |
| US5458124A (en) | 1994-02-08 | 1995-10-17 | Stanko; Bruce E. | Electrocardiographic signal monitoring system |
| WO1999047204A1 (en) | 1998-03-18 | 1999-09-23 | Urosurge, Inc. | Electro-nerve stimulator systems and methods |
| EP1064047A1 (en) | 1998-03-18 | 2001-01-03 | Urosurge, Inc. | Electro-nerve stimulator systems and methods |
| US6493588B1 (en) | 1998-03-18 | 2002-12-10 | Mmc/Gatx Partnership No. 1 | Electro-nerve stimulator systems and methods |
| US6049789A (en) | 1998-06-24 | 2000-04-11 | Mentor Graphics Corporation | Software pay per use licensing system |
| US20020120467A1 (en) | 2000-07-14 | 2002-08-29 | Asbjorn Buanes | Data processing system and method for managing the use of a medical device |
| US7962416B1 (en) | 2000-11-22 | 2011-06-14 | Ge Medical Technology Services, Inc. | Method and system to remotely enable software-based options for a trial period |
| US6983375B2 (en) | 2001-04-13 | 2006-01-03 | Ge Medical Technology Services, Inc. | Method and system to grant indefinite use of software options resident on a device |
| US6829704B2 (en) | 2001-04-13 | 2004-12-07 | General Electric Company | Method and system to automatically activate software options upon initialization of a device |
| US6966000B2 (en) | 2001-04-13 | 2005-11-15 | Ge Medical Technology Services, Inc. | Method and system to remotely grant limited access to software options resident on a device |
| US7739725B1 (en) | 2001-04-13 | 2010-06-15 | Ge Medical Technology Services, Inc. | Method and system to grant indefinite use of software options resident on a device |
| US7634645B2 (en) | 2001-04-13 | 2009-12-15 | Ge Medical Technology Services, Inc. | Method and system to request remotely enabled access to inactive software options resident on a device |
| US6662052B1 (en) | 2001-04-19 | 2003-12-09 | Nac Technologies Inc. | Method and system for neuromodulation therapy using external stimulator with wireless communication capabilites |
| US7349856B2 (en) | 2001-07-30 | 2008-03-25 | Siemens Aktiengesellschaft | Method for selectively enabling or blocking the use of medical equipment |
| US7536226B2 (en) | 2001-10-18 | 2009-05-19 | Uroplasty, Inc. | Electro-nerve stimulator system and methods |
| US7219222B1 (en) | 2002-01-18 | 2007-05-15 | Ge Medical Technology Services, Inc. | Method and system to grant access to software options resident on a medical imaging device |
| US20040102819A1 (en) | 2002-11-20 | 2004-05-27 | Shazhou Zou | Portable therapeutic device and method for pain relief |
| US7421516B2 (en) | 2003-10-28 | 2008-09-02 | General Electric Company | System and method for multi-vendor authentication to remotely activate a software-based option |
| US7113894B2 (en) | 2003-10-28 | 2006-09-26 | General Electric Company | System and method for coordinated remote activation of multiple software-based options |
| US6990434B2 (en) | 2003-10-28 | 2006-01-24 | General Electric Company | System and method for coordinated remote activation of multiple software-based options |
| US7093032B2 (en) | 2003-10-28 | 2006-08-15 | General Electric Company | System and method for multi-vendor authentication to remotely activate a software-based option |
| US20050165693A1 (en) | 2004-01-23 | 2005-07-28 | Klaus Moritzen | Prepaid licensing system and method |
| US7571002B2 (en) | 2004-02-12 | 2009-08-04 | Ndi Medical, Llc | Portable percutaneous assemblies, systems and methods for providing highly selective functional or therapeutic neuromuscular stimulation |
| US7894913B2 (en) | 2004-06-10 | 2011-02-22 | Medtronic Urinary Solutions, Inc. | Systems and methods of neuromodulation stimulation for the restoration of sexual function |
| US7813809B2 (en) | 2004-06-10 | 2010-10-12 | Medtronic, Inc. | Implantable pulse generator for providing functional and/or therapeutic stimulation of muscles and/or nerves and/or central nervous system tissue |
| US20080140451A1 (en) | 2005-01-10 | 2008-06-12 | Cytori Therapeutics, Inc. | Devices and Methods for Monitoring, Managing, and Servicing Medical Devices |
| US20060190048A1 (en) | 2005-02-23 | 2006-08-24 | Medtronic, Inc. | Implantable neurostimulator supporting trial and chronic modes |
| WO2006091611A1 (en) | 2005-02-23 | 2006-08-31 | Medtronic, Inc. | Implantable neurostimulator supporting trial and chronic modes |
| US20070167990A1 (en) * | 2006-01-17 | 2007-07-19 | Theranova, Llc | Method and apparatus for low frequency induction therapy for the treatment of urinary incontinence and overactive bladder |
| US20150202454A1 (en) | 2006-01-17 | 2015-07-23 | Emkinetics, Inc. | Methods and devices for performing electrical stimulation to treat various conditions |
| US20140148870A1 (en) | 2006-01-17 | 2014-05-29 | Emkinetics, Inc. | Methods and devices for performing electrical stimulation to treat various conditions |
| US20070167992A1 (en) | 2006-01-18 | 2007-07-19 | Baylor Research Institute | Method and apparatus for reducing preterm labor using neuromodulation |
| US20100114259A1 (en) * | 2006-05-18 | 2010-05-06 | Uroplasty, Inc. | Method and Apparatus for Stimulating a Nerve of a Patient |
| US20080306325A1 (en) | 2006-10-02 | 2008-12-11 | Emkinetics | Method and apparatus for magnetic induction therapy |
| US20100168501A1 (en) | 2006-10-02 | 2010-07-01 | Daniel Rogers Burnett | Method and apparatus for magnetic induction therapy |
| US20100324621A1 (en) * | 2006-10-11 | 2010-12-23 | Imad Libbus | Transcutaneous neurostimulator for modulating cardiovascular function |
| US20080091256A1 (en) * | 2006-10-11 | 2008-04-17 | Cardiac Pacemakers | Transcutaneous neurostimulator for modulating cardiovascular function |
| US20090132018A1 (en) | 2007-11-16 | 2009-05-21 | Ethicon, Inc. | Nerve Stimulation Patches And Methods For Stimulating Selected Nerves |
| US20090248578A1 (en) | 2008-03-20 | 2009-10-01 | Liposonix, Inc. | Methods and apparatus for medical device investment recovery |
| EP2344241A1 (en) | 2008-10-01 | 2011-07-20 | Cardiola Ltd. | An apparatus for use on a person's lap |
| US20110264160A1 (en) | 2008-10-01 | 2011-10-27 | Cardiola Ltd. | Apparatus for use on a Person's Lap |
| WO2010037471A1 (en) | 2008-10-01 | 2010-04-08 | Cardiola Ltd. | An apparatus for use on a person's lap |
| WO2011041526A2 (en) | 2009-10-01 | 2011-04-07 | University Of Pittsburgh-Of The Commonwealth System Of Higher Education | Non-invasive method to treat urological and gastrointestinal disorders |
| WO2011044159A1 (en) | 2009-10-05 | 2011-04-14 | Arp Manufacturing Llc | Electro-therapeutic stimulator |
| EP2485796A1 (en) | 2009-10-05 | 2012-08-15 | Arp Manufacturing LLC | Electro-therapeutic stimulator |
| US20110082524A1 (en) | 2009-10-05 | 2011-04-07 | Arp Manufacturing Llc | Electro-Therapeutic Stimulator |
| WO2011053607A1 (en) | 2009-10-26 | 2011-05-05 | Emkinetics, Inc. | Method and apparatus for electromagnetic stimulation of nerve, muscle, and body tissues |
| US20120302821A1 (en) | 2009-10-26 | 2012-11-29 | Emkinetics, Inc. | Method and apparatus for electromagnetic stimulation of nerve, muscle, and body tissues |
Non-Patent Citations (9)
| Title |
|---|
| Interview Summary from U.S. Appl. No. 14/694,704, mailed Sep. 22, 2015, 3 pp. |
| Notice of Allowance from U.S. Appl. No. 14/694,704, mailed Oct. 1, 2015, 7 pp. |
| Notice of Reason for Rejection, and translation thereof, from counterpart Japanese Application No. 2014-516011, dated Mar. 3, 2016, 8 pp. |
| Office Action from U.S. Appl. No. 14/694,704, dated Jun. 10, 2015, 4 pp. |
| Patent Examination Report from counterpart Australian Application No. 2012271480, dated Jan. 11, 2016, 3 pp. |
| Protest by the Public Against Pending Application, for U.S. Appl. No. 14/298,207, dated Feb. 22, 2016, 6 pp. |
| Response to counterpart Australian Examination Report dated Jan. 11, 2016, from counterpart Australian Patent Application No. 2012271480, filed on Jun. 6, 2016, 3 pp. |
| Response to Office Action dated Jun. 10, 2015, from U.S. Appl. No. 14/694,704, filed Sep. 9, 2015, 8 pp. |
| Wrenn, et al.: Incorporating Percutaneous Tibial Nerve Stimulation (PTNS) Into a Community Based Urology Practice; www.urotoday.com/overactive-bladder, Jun. 14, 2011. |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11376429B2 (en) | 2012-04-26 | 2022-07-05 | Medtronic, Inc. | Trial stimulation systems |
| US11517752B2 (en) | 2012-04-26 | 2022-12-06 | Medtronic, Inc. | Trial stimulation systems |
| US11806527B2 (en) | 2012-04-26 | 2023-11-07 | Medtronic, Inc. | Trial stimulation systems |
| US11033737B2 (en) | 2014-08-17 | 2021-06-15 | Coloplast A/S | Miniature implantable neurostimulator system for sciatic nerves and their branches |
| US11679257B2 (en) | 2014-08-17 | 2023-06-20 | Coloplast A/S | Method of treating an overactive bladder condition |
| US10589089B2 (en) | 2017-10-25 | 2020-03-17 | Epineuron Technologies Inc. | Systems and methods for delivering neuroregenerative therapy |
| US11247044B2 (en) | 2017-10-25 | 2022-02-15 | Epineuron Technologies Inc. | Devices for delivering neuroregenerative therapy |
| US11247045B2 (en) | 2017-10-25 | 2022-02-15 | Epineuron Technologies Inc. | Systems and methods for delivering neuroregenerative therapy |
| US11247043B2 (en) | 2019-10-01 | 2022-02-15 | Epineuron Technologies Inc. | Electrode interface devices for delivery of neuroregenerative therapy |
| US11364381B2 (en) | 2019-10-01 | 2022-06-21 | Epineuron Technologies Inc. | Methods for delivering neuroregenerative therapy and reducing post-operative and chronic pain |
Also Published As
| Publication number | Publication date |
|---|---|
| US20140121726A1 (en) | 2014-05-01 |
| EP2720749A4 (en) | 2014-11-26 |
| WO2012174330A1 (en) | 2012-12-20 |
| AU2012271480B2 (en) | 2016-07-14 |
| EP2720749B1 (en) | 2018-09-19 |
| EP2720749A1 (en) | 2014-04-23 |
| US20120323294A1 (en) | 2012-12-20 |
| CA2839532A1 (en) | 2012-12-20 |
| US8660646B2 (en) | 2014-02-25 |
| US20140288613A1 (en) | 2014-09-25 |
| JP2014527416A (en) | 2014-10-16 |
| US8818520B2 (en) | 2014-08-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9498633B2 (en) | Percutaneous tibial nerve stimulator | |
| AU2012271480A1 (en) | Percutaneous tibial nerve stimulator | |
| US9245265B2 (en) | Medical device payment system | |
| US11426579B2 (en) | Systems, methods and kits for peripheral nerve stimulation | |
| US11872404B2 (en) | Systems and methods for wireless control of noninvasive electrotherapy | |
| US10080897B2 (en) | IPG configured to deliver different pulse regimes to different leads for pudendal nerve stimulation | |
| US20140330342A1 (en) | Smart phone application for providing neuro/muscular electro stimulation | |
| US10946194B2 (en) | System for improving neurostimulation compliance | |
| US11229788B1 (en) | Systems for improving neurostimulation compliance using a patient interface module | |
| WO2005122740A2 (en) | Interferential and neuromuscular electrical stimulation system and apparatus | |
| US20220008723A1 (en) | System, Method, and Apparatus for Neurostimulation | |
| CA3034123A1 (en) | Systems, methods, and kits for peripheral nerve stimulation | |
| US20220176118A1 (en) | System for improving neurostimulation treatment compliance with gamification | |
| US20220409896A1 (en) | System for display of neurostimulation usage and compliance |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: ADVANCED URO-SOLUTIONS, LLC, TENNESSEE Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:LAING, BRENT, DR.;GREEN, JOHN DAVID, DR.;REEL/FRAME:033283/0150 Effective date: 20140321 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1551); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 4 |