USRE39561E1 - Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts - Google Patents
Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts Download PDFInfo
- Publication number
- USRE39561E1 USRE39561E1 US08/895,950 US89595097A USRE39561E US RE39561 E1 USRE39561 E1 US RE39561E1 US 89595097 A US89595097 A US 89595097A US RE39561 E USRE39561 E US RE39561E
- Authority
- US
- United States
- Prior art keywords
- group
- different
- compound
- halogen atom
- alkyl group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 0 CC.CC.[1*]C([2*])(C)C.[3*]c1c([5*])c(C[7*]Cc2c([6*])c([4*])c3c2CCCC3)c2c1CCCC2 Chemical compound CC.CC.[1*]C([2*])(C)C.[3*]c1c([5*])c(C[7*]Cc2c([6*])c([4*])c3c2CCCC3)c2c1CCCC2 0.000 description 17
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F17/00—Metallocenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/0805—Compounds with Si-C or Si-Si linkages comprising only Si, C or H atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F10/00—Homopolymers and copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F110/00—Homopolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F110/04—Monomers containing three or four carbon atoms
- C08F110/06—Propene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/639—Component covered by group C08F4/62 containing a transition metal-carbon bond
- C08F4/63912—Component covered by group C08F4/62 containing a transition metal-carbon bond in combination with an organoaluminium compound
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/639—Component covered by group C08F4/62 containing a transition metal-carbon bond
- C08F4/6392—Component covered by group C08F4/62 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring
- C08F4/63922—Component covered by group C08F4/62 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not
- C08F4/63927—Component covered by group C08F4/62 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not two cyclopentadienyl rings being mutually bridged
Abstract
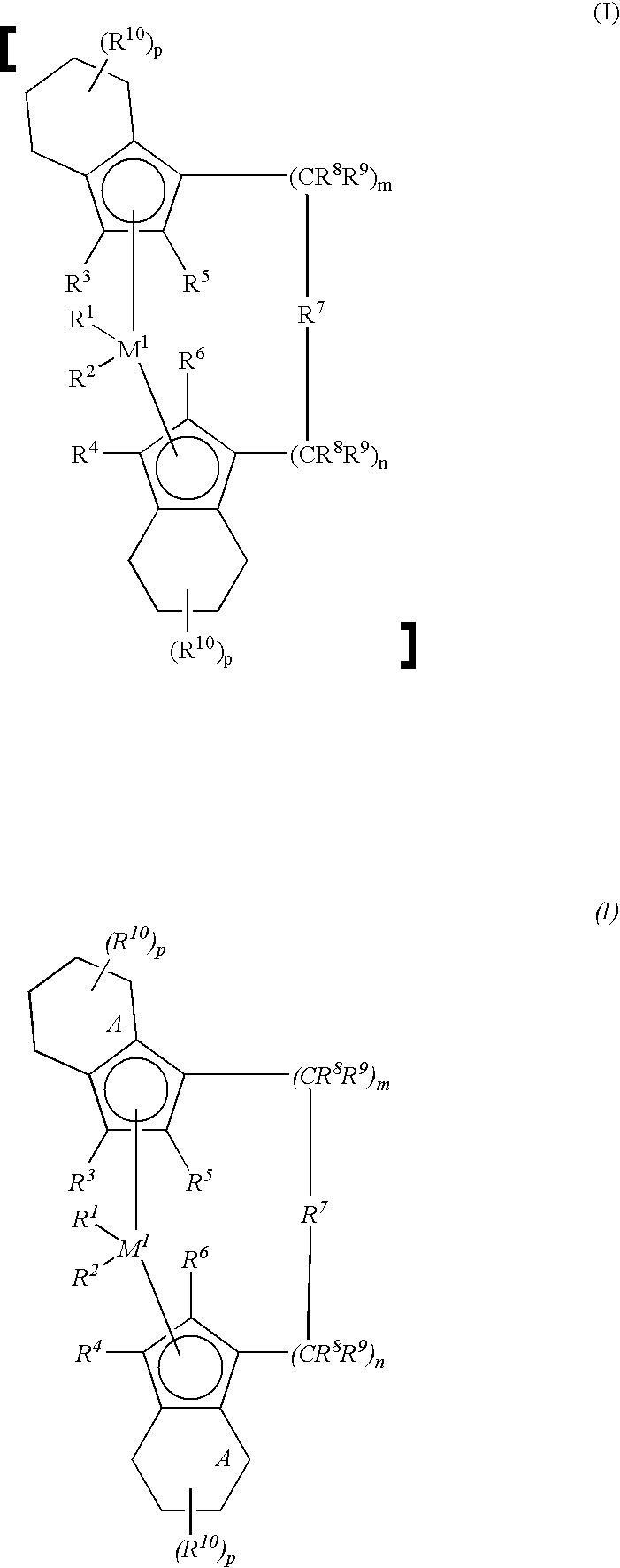
in which, preferably, M1 is Zr or Hf, R1 and R2 are alkyl or halogen, R3 and R4 are hydrogen, R5 and R6 are alkyl or haloalkyl, —(CR8R9)m—R7—(CR8R9)n— is a single- or multi-membered chain in which R7 may also be a (substituted) hetero atom, m+n is zero or 1, and R10 is hydrogen, form, together with aluminoxanes as cocatalysts, a very effective catalyst system for the preparation of polyolefins of high stereospecificity and high melting point.
Description
- M1 is a metal from group IVb, Vb or VIb of the Periodic Table,
- R1 and R2 are identical or different and are a hydrogen atom, a C1-C10-alkyl group, a C1-C10-alkoxy group, a C6-C10-aryl group, a C6-C10-aryloxy group, a C2-C10-alkenyl group, a C7-C40-arylalkyl group, a C7-C40-alkylaryl group, a C8-C40-arylalkenyl group or a halogen atom,
- R3 and R4 are identical or different and are a hydrogen atom, a halogen atom, a C1-C10-alkyl group, which may be halogenated, a C6-C10-aryl group, an —NR2 15, —SR15, —OSiR3 15, —SiR3 15 or —PR2 15 radical in which R15 is a halogen atom, a C1-C10-alkyl group or a C6-C10-aryl group,
- R5 and R6 are identical or different and are as defined for R3 and R4, with the proviso that R5 and R6 are not hydrogen,
- R7 is
where - R11, R12 and R13 are identical or different and are a hydrogen atom, a halogen atom, a C1-C10-alkyl group, a C1-C10-fluoroalkyl group, a C6-C10-aryl group, a C6-C10-fluoroaryl group, a C1-C10-alkoxy group, a C2-C10-alkenyl group, a C7-C40-arylalkyl group, a C8-C40-arylalkenyl group or a C7-C40-alkylaryl group, or R11 and R12 or R11 and R13, in each case with the atoms connecting them, form a ring,
- M2 is silicon, germanium or tin,
- R8 and R9 are identical or different and are as defined for R11,
- m and n are identical or different and are zero, 1 or 2, m plus n being zero, 1 or 2, and,
- the radicals R10 are identical or different and are as defined for R11, R12 and R13.
- Alkyl is straight-chain or branched alkyl. Halogen (halogenated) is fluorine, chlorine, bromine or iodine, preferably fluorine or chlorine.
- R1 and R2 are identical or different and are a hydrogen atom, a C1-C10-, preferably C1-C3-alkyl group, a C1-C10-, preferably C1-C3-alkoxy group, a C6-C10-, preferably C6-C8-aryl group, a C6-C10-, preferably C6-C8-aryloxy group, a C2-C10-, preferably C2-C4-alkenyl group, a C7-C40-, preferably C7-C10-arylalkyl group, a C7-C40-, preferably C7-C12-alkylaryl group, a C8-C40-, preferably C8-C12-arylalkenyl group or a halogen atom, preferably chlorine.
- R3 and R4 are identical or different and are a hydrogen atom, a halogen atom, preferably a fluorine, chlorine or bromine atom, a C1-C10-, preferably C1-C4-alkyl group, which may be halogenated, a C6-C10-, preferably C6-C8-aryl group, an —NR2 15, —SR15, —O—SiR3 15, —SiR3 15 or —PR2 15 radical in which R15 is a halogen atom, preferably a chlorine atom, or a C1-C10-, preferably C1-C3-alkyl group or a C6-C10-, preferably C6-C8-aryl group. R3 and R4 are particularly preferably hydrogen.
- R5 and R6 are identical or different, preferably identical, and are as defined for R3 and R4, with the proviso that R5 and R6 cannot be hydrogen. R5 and R6 are preferably (C1-C4)-alkyl, which may be halogenated, such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl or trifluoromethyl, in particular methyl.
- R7 is
═BR11, ═AlR11, —Ge—, —Sn—, —O—, —S—, ═SO, ═SO2, ═NR11, ═CO, ═PR11 or ═P(O)R11, where R11, R12 and R13 are identical or different and are a hydrogen atom, a halogen atom, a C1-C10-, preferably C1-C4-alkyl group, in particular a methyl group, a C1-C10-fluoroalkyl group, preferably a CF3 group, a C6-C10-, preferably C6-C8-aryl group, a C6-C10-fluoroaryl group, preferably a pentafluorophenyl group, a C1-C10-, preferably C1-C4-alkoxy group, in particular a methoxy group, a C2-C10-, preferably C2-C4-alkenyl group, a C7-C40-, preferably C7-C10-arylalkyl group, a C8-C40-, preferably C8-C12-arylalkenyl group or a C7-C40-, preferably C7-C12-alkylaryl group, or R11 and R12 or R11 and R13, in each case together with the atoms connecting them, form a ring. - M2 is silicon, germanium or tin, preferably silicon or germanium.
- R7 is preferably ═CR11R12, ═SiR11R12, ═GeR11R12, —O—, —S—, ═SO, ═PR11 or ═P(O)R11.
- R8 and R9 are identical or different and are as define as for R11.
- m and n are identical or different and are zero, 1 or 2, preferably zero or 1, where m plus n is zero, 1 or 2, preferably zero or 1.
radical, n plus m is zero or 1, and R10 is hydrogen; in particular the compounds I listed in the working examples.
- a) reacting a compound of the formula II
in which R3-R10, m and n are defined in the formula I and M3 is an alkali metal, preferably lithium, with a compound of the formula III
M1X4 (III)
in which M1 is a defined in the formula I, and X is a halogen atom, preferably chlorine, and catalytically hydrogenating the reaction product, or - b) reacting a compound of the formula IIa
with a compound of the formula III
M1X4 (III)
in which all the substituents are as defined under a), and, if desired, derivatizing the reaction product obtained under a) or b).
for the linear type and/or of the formula (V)
for the cyclic type, where, in the formulae (IV) and (V), the radicals R may be identical or different and are a C1-C6-alkyl group, a C6-C18-aryl group or hydrogen, and p is an integer from 2 to 50, preferably from 10 to 35.
| VN = | viscosity number in cm3/g | ||
| determined | |||
| Mw = | weight average molecular weight | by gel perme- | |
| Mw/Mn = | molecular weight dispersity | ation chroma- | |
| tography | |||
| II = | isotactic index (II = mm + 1/8 ms), determined by |
| 13C-NMR spectroscopy | |
| niso = | length of the isotactic blocks (in propylene units) |
| (niso = 1 + 2 mm/mr), determined by 13C-NMR | |
| spectroscopy | |
| Comp. | Polym. | M.p. | ΔHmeh | ||
| Ex. | Metallocene | temp. [° C.] | niso | [° C. ] | [J/g] |
| A | 1 | 70 | 38 | 145 | 86.6 |
| B | 1 | 50 | 48 | 148 | 88.1 |
| C | 1 | 30 | 48 | 152 | 90.2 |
| D | 2 | 70 | 34 | 141 | — |
| E | 2 | 50 | 38 | 143 | — |
| F | 3 | 70 | 32 | 140 | — |
| G | 3 | 50 | 34 | 142 | — |
| H | 3 | 30 | 37 | 145 | — |
| Comp. | Polym. | M.p. | ΔHmeh | ||
| Ex. | Metallocene | temp. [° C.] | niso | [° C. ] | [J/g] |
| I | 4 | 70 | 23 | 132 | 64.9 |
| K | 4 | 50 | 30 | 138 | 78.1 |
| L | 4 | 30 | 29 | 137 | 78.6 |
| M | 5 | 70 | 25 | 134 | 77.0 |
| N | 5 | 50 | 30 | 138 | 78.9 |
| O | 5 | 30 | 32 | 138 | 78.6 |
Claims (23)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/895,950 USRE39561E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE4035884 | 1990-11-12 | ||
| US07/789,361 US5276208A (en) | 1990-11-12 | 1991-11-08 | Metallocenes containing ligands of 2-substituted idenyl derivatives, process for their preparation, and their use as catalysts |
| US08/324,260 USRE37208E1 (en) | 1990-11-12 | 1994-10-17 | Polyolefins prepared with metallocene catalysts having 2-substituted indenyl type ligands |
| US08/895,950 USRE39561E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US07/789,361 Reissue US5276208A (en) | 1990-11-12 | 1991-11-08 | Metallocenes containing ligands of 2-substituted idenyl derivatives, process for their preparation, and their use as catalysts |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USRE39561E1 true USRE39561E1 (en) | 2007-04-10 |
Family
ID=27201879
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US08/895,909 Expired - Lifetime USRE39532E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
| US08/895,950 Expired - Lifetime USRE39561E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US08/895,909 Expired - Lifetime USRE39532E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
Country Status (1)
| Country | Link |
|---|---|
| US (2) | USRE39532E1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080200708A1 (en) * | 2002-10-25 | 2008-08-21 | Basell Polyolefine Gmbh | Preparation of Partially Hydrogenated Rac-Ansa-Metallocene Complexes |
Citations (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE284708C (en) | ||||
| EP0129368A1 (en) | 1983-06-06 | 1984-12-27 | Exxon Research And Engineering Company | Process and catalyst for polyolefin density and molecular weight control |
| EP0185918A2 (en) | 1984-11-27 | 1986-07-02 | Hoechst Aktiengesellschaft | Process for preparing polyolefins |
| JPS62121707A (en) * | 1985-11-22 | 1987-06-03 | Mitsui Petrochem Ind Ltd | Production of alpha-olefin random copolymer |
| DE3726067A1 (en) | 1987-08-06 | 1989-02-16 | Hoechst Ag | METHOD FOR PRODUCING 1-OLEFIN POLYMERS |
| EP0316155A2 (en) | 1987-11-09 | 1989-05-17 | Chisso Corporation | A transition-metal compound having a substituted, bridged bis-cyclopentadienyl ligand |
| EP0320762A2 (en) | 1987-12-18 | 1989-06-21 | Hoechst Aktiengesellschaft | Process for the preparation of a chiral, stereorigid metallocen compound |
| US4871705A (en) * | 1988-06-16 | 1989-10-03 | Exxon Chemical Patents Inc. | Process for production of a high molecular weight ethylene a-olefin elastomer with a metallocene alumoxane catalyst |
| EP0336128A2 (en) | 1988-03-12 | 1989-10-11 | Hoechst Aktiengesellschaft | Process for preparing an alpha-olefin polymer |
| EP0344887A2 (en) | 1988-03-21 | 1989-12-06 | Exxon Chemical Patents Inc. | Silicon bridged transition metal compounds |
| EP0347129A1 (en) | 1988-06-16 | 1989-12-20 | Exxon Chemical Patents Inc. | Process for production of high molecular weight EPDM elastomers using a metallocene-alumoxane catalyst system |
| US4892851A (en) | 1988-07-15 | 1990-01-09 | Fina Technology, Inc. | Process and catalyst for producing syndiotactic polyolefins |
| DE3826075A1 (en) | 1988-07-30 | 1990-02-01 | Hoechst Ag | 1-OLEFIN ISO BLOCK POLYMER AND METHOD FOR THE PRODUCTION THEREOF |
| EP0355289A1 (en) | 1988-07-01 | 1990-02-28 | ATOCHEM NORTH AMERICA, INC. (a Pennsylvania corp.) | Accelerometer |
| EP0366290A2 (en) | 1988-10-24 | 1990-05-02 | Chisso Corporation | Process for producing olefin polymers |
| EP0407870A2 (en) | 1989-07-08 | 1991-01-16 | Hoechst Aktiengesellschaft | Process for preparing cycloolefin polymers |
| EP0426643A1 (en) | 1989-10-30 | 1991-05-08 | Fina Research S.A. | Process for the preparation of metallocenes |
| US5017714A (en) | 1988-03-21 | 1991-05-21 | Exxon Chemical Patents Inc. | Silicon-bridged transition metal compounds |
| EP0433990A2 (en) | 1989-12-21 | 1991-06-26 | Hoechst Aktiengesellschaft | Process for preparing polypropylene molding compositions |
| EP0442725A2 (en) | 1990-02-13 | 1991-08-21 | Mitsui Petrochemical Industries, Ltd. | Olefin polymerization solid catalysts and process for the polymerization of olefins |
| EP0485821A1 (en) | 1990-11-12 | 1992-05-20 | Hoechst Aktiengesellschaft | Metallocenes with 2-substituted indenyl-derivates as ligands, process for their preparation and their use as catalysts |
| EP0485823A1 (en) | 1990-11-12 | 1992-05-20 | Hoechst Aktiengesellschaft | 2-Substituted bisindenyl-metallocenes, process for their preparation and their use as catalysts for the polymerization of olefins |
| EP0500005A1 (en) | 1991-02-18 | 1992-08-26 | Hoechst Aktiengesellschaft | Process for the preparation of substituted indenes |
| EP0529908A1 (en) | 1991-08-20 | 1993-03-03 | Mitsubishi Chemical Corporation | Catalyst useful for the polymerization of olefin |
| US5243001A (en) | 1990-11-12 | 1993-09-07 | Hoechst Aktiengesellschaft | Process for the preparation of a high molecular weight olefin polymer |
| US5278264A (en) | 1991-08-26 | 1994-01-11 | Hoechst Ag | Process for the preparation of an olefin polymer |
| US5296434A (en) | 1991-06-18 | 1994-03-22 | Basf Aktiengesellschaft | Soluble catalyst systems for the preparation of polyalk-1-enes having high molecular weights |
| US5324800A (en) | 1983-06-06 | 1994-06-28 | Exxon Chemical Patents Inc. | Process and catalyst for polyolefin density and molecular weight control |
| US5455365A (en) | 1992-08-03 | 1995-10-03 | Hoechst Ag | Process for the preparation of an olefin polymer using metallocenes containing specifically substituted indenyl ligands |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3192276A (en) * | 1962-09-10 | 1965-06-29 | Union Carbide Corp | Alkylation of indene |
-
1997
- 1997-07-17 US US08/895,909 patent/USRE39532E1/en not_active Expired - Lifetime
- 1997-07-17 US US08/895,950 patent/USRE39561E1/en not_active Expired - Lifetime
Patent Citations (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE284708C (en) | ||||
| EP0129368A1 (en) | 1983-06-06 | 1984-12-27 | Exxon Research And Engineering Company | Process and catalyst for polyolefin density and molecular weight control |
| US5324800A (en) | 1983-06-06 | 1994-06-28 | Exxon Chemical Patents Inc. | Process and catalyst for polyolefin density and molecular weight control |
| EP0185918A2 (en) | 1984-11-27 | 1986-07-02 | Hoechst Aktiengesellschaft | Process for preparing polyolefins |
| US4769510A (en) * | 1984-11-27 | 1988-09-06 | Hoechst Aktiengesellschaft | Process for the preparation of polyolefins |
| JPS62121707A (en) * | 1985-11-22 | 1987-06-03 | Mitsui Petrochem Ind Ltd | Production of alpha-olefin random copolymer |
| DE3726067A1 (en) | 1987-08-06 | 1989-02-16 | Hoechst Ag | METHOD FOR PRODUCING 1-OLEFIN POLYMERS |
| US4931417A (en) | 1987-11-09 | 1990-06-05 | Chisso Corporation | Transition-metal compound having a bis-substituted-cyclopentadienyl ligand of bridged structure |
| EP0316155A2 (en) | 1987-11-09 | 1989-05-17 | Chisso Corporation | A transition-metal compound having a substituted, bridged bis-cyclopentadienyl ligand |
| EP0320762A2 (en) | 1987-12-18 | 1989-06-21 | Hoechst Aktiengesellschaft | Process for the preparation of a chiral, stereorigid metallocen compound |
| US5103030A (en) | 1987-12-18 | 1992-04-07 | Hoechst Aktiengesellschaft | Process for the preparation of a chiral stereorigid metallocene |
| EP0336128A2 (en) | 1988-03-12 | 1989-10-11 | Hoechst Aktiengesellschaft | Process for preparing an alpha-olefin polymer |
| EP0344887A2 (en) | 1988-03-21 | 1989-12-06 | Exxon Chemical Patents Inc. | Silicon bridged transition metal compounds |
| US5017714A (en) | 1988-03-21 | 1991-05-21 | Exxon Chemical Patents Inc. | Silicon-bridged transition metal compounds |
| EP0347129A1 (en) | 1988-06-16 | 1989-12-20 | Exxon Chemical Patents Inc. | Process for production of high molecular weight EPDM elastomers using a metallocene-alumoxane catalyst system |
| US4871705A (en) * | 1988-06-16 | 1989-10-03 | Exxon Chemical Patents Inc. | Process for production of a high molecular weight ethylene a-olefin elastomer with a metallocene alumoxane catalyst |
| EP0355289A1 (en) | 1988-07-01 | 1990-02-28 | ATOCHEM NORTH AMERICA, INC. (a Pennsylvania corp.) | Accelerometer |
| EP0351392A2 (en) | 1988-07-15 | 1990-01-17 | Fina Technology, Inc. | Process and catalyst for producing syndiotactic polyolefins |
| US4892851A (en) | 1988-07-15 | 1990-01-09 | Fina Technology, Inc. | Process and catalyst for producing syndiotactic polyolefins |
| DE3826075A1 (en) | 1988-07-30 | 1990-02-01 | Hoechst Ag | 1-OLEFIN ISO BLOCK POLYMER AND METHOD FOR THE PRODUCTION THEREOF |
| EP0366290A2 (en) | 1988-10-24 | 1990-05-02 | Chisso Corporation | Process for producing olefin polymers |
| EP0407870A2 (en) | 1989-07-08 | 1991-01-16 | Hoechst Aktiengesellschaft | Process for preparing cycloolefin polymers |
| US5087677A (en) | 1989-07-08 | 1992-02-11 | Hoechst Aktiengesellschaft | Process for the preparation of cycloolefin polymers |
| EP0426643A1 (en) | 1989-10-30 | 1991-05-08 | Fina Research S.A. | Process for the preparation of metallocenes |
| EP0433990A2 (en) | 1989-12-21 | 1991-06-26 | Hoechst Aktiengesellschaft | Process for preparing polypropylene molding compositions |
| EP0442725A2 (en) | 1990-02-13 | 1991-08-21 | Mitsui Petrochemical Industries, Ltd. | Olefin polymerization solid catalysts and process for the polymerization of olefins |
| EP0485823A1 (en) | 1990-11-12 | 1992-05-20 | Hoechst Aktiengesellschaft | 2-Substituted bisindenyl-metallocenes, process for their preparation and their use as catalysts for the polymerization of olefins |
| US5145819A (en) | 1990-11-12 | 1992-09-08 | Hoechst Aktiengesellschaft | 2-substituted disindenylmetallocenes, process for their preparation, and their use as catalysts in the polymerization of olefins |
| US5243001A (en) | 1990-11-12 | 1993-09-07 | Hoechst Aktiengesellschaft | Process for the preparation of a high molecular weight olefin polymer |
| US5276208A (en) | 1990-11-12 | 1994-01-04 | Hoechst Aktiengesellschaft | Metallocenes containing ligands of 2-substituted idenyl derivatives, process for their preparation, and their use as catalysts |
| EP0485821A1 (en) | 1990-11-12 | 1992-05-20 | Hoechst Aktiengesellschaft | Metallocenes with 2-substituted indenyl-derivates as ligands, process for their preparation and their use as catalysts |
| EP0500005A1 (en) | 1991-02-18 | 1992-08-26 | Hoechst Aktiengesellschaft | Process for the preparation of substituted indenes |
| US5296434A (en) | 1991-06-18 | 1994-03-22 | Basf Aktiengesellschaft | Soluble catalyst systems for the preparation of polyalk-1-enes having high molecular weights |
| EP0529908A1 (en) | 1991-08-20 | 1993-03-03 | Mitsubishi Chemical Corporation | Catalyst useful for the polymerization of olefin |
| US5561093A (en) | 1991-08-20 | 1996-10-01 | Mitsubishi Chemical Corporation | Catalyst useful for the polymerization of olefins |
| US6143911A (en) | 1991-08-20 | 2000-11-07 | Mitsubishi Petrochemical Company Limited | Catalyst useful for the polymerization of olefins |
| US5278264A (en) | 1991-08-26 | 1994-01-11 | Hoechst Ag | Process for the preparation of an olefin polymer |
| US5329033A (en) | 1991-08-26 | 1994-07-12 | Hoechst Aktiengesellschaft | Process for the preparation of an olefin polymer |
| US5455365A (en) | 1992-08-03 | 1995-10-03 | Hoechst Ag | Process for the preparation of an olefin polymer using metallocenes containing specifically substituted indenyl ligands |
Non-Patent Citations (20)
| Title |
|---|
| Adcock et al., Austr. J. Chem., vol. 29, "Substituent Effects by <SUP>19</SUP>F Nuclear Magnetic Resonance: Polar and pi-Electron Effects", pp. 2571-2581. |
| BULLETIN DE LA SOCIETE CHIMIQUE DE FRANCE, "Etude de monomeres halogenes et de leur polymerisation cationique", No. 11, pp. 3092-3095, (1973). |
| Chemical Abstracts 90:567 103691p; (1978). |
| Criegee et al., Chem. Ber., vol. 94, "Uber den Nickelkomplex C<SUB>18</SUB>H<SUB>22</SUB>Ni und den daraus gewonnenen Kohlenwasserstoff C<SUB>13</SUB>H<SUB>18</SUB>", pp. 3461-3468 (1964). |
| Esperas, S., ACTA CHEMICA SCANDINAVICA, "The Crystal and Molecular Structure of Cyano(methylisocyanide)gold(I)", A 30, No. 7, pp. 527-530 (1976). |
| Ewen, J.A., et al, J. Am. Chem. Soc., Crystal Structures and Sterospecific Propylene Polymerizations with Chiral Hafnium Metallocene Catalysts, vol. 109, 1987, pp. 6544-6545. |
| Hart et al., NOTES, J. Am. Chem. Soc., vol. 72, "Acylation-Akylation Studies", pp. 3286-3287 (1950). |
| J. ORG. CHEM., "Friedel-Crafts Chemistry, A Mechanistic Study of the Reaction of 3-Chloro-4'-fluoro-2-methylpropiophenone with AlCl<SUB>3</SUB>and AlCl<SUB>3</SUB>-CH<SUB>3</SUB>NO<SUB>2</SUB>", vol. 43, No. 16, pp. 3126-3131 (1978). |
| J. ORG. CHEM., "Friedel-Crafts Reactions of Ethyl Cyclopropanecarboxylate", vol. 46, pp. 3758-3760 (1981). |
| JACS (1967) 89(23) 5868-5876, Nov. 1967. * |
| Katz, Thomas J., J. Am. Chem. Soc., "Asymmetric Synthesis of Helical Metallocenes", vol. 108, 1986, pp. 179-181. |
| Marechal et al., Bull. Soc. Chim. Fr. 6, "Homopolymerisation cationlique des dimethyl-4,7,dimethyl-4,6 et dimethyl-5,6 indenes", No. 348, pp. 1981-2039, (1969). |
| Miyamota, T.K., et al., CHEMISTRY LETTERS, The Chemical Society of Japan, "A Bulky Ligand and its Organometallic Compound: Synthesis of Heptamethylidene and a Ferrocene-Type Complex, Fe(n<SUP>5</SUP>-C<SUB>9</SUB>Me<SUB>7</SUB>)<SUB>2</SUB>", pp. 729-730 (1981). |
| Piccoliovazzi, N. et al., ORGANOMETALLICS, "Electronic Effects in Homogeneous Indenylzirconium Ziegler-Natta Catalysts", vol. 9, pp. 3098-3105 (1990). |
| Röll, V.W., et al., ANGEW. CHEM., "Stereo- und Regioselektivitat von chiralen, alkylsubstituierten ansa-Zirconocen-Katalysatoren bei der Methylalumoxan-aktivierten Propen-Polymerization", vol. 102, No. 3, pp. 339-341 (1990). |
| Soga, K. et al., MACROMOLECULES, "Perfect Conversion of Aspecific Sites into Isopecific Sites in Ziegler-Natta Catalysts", vol. 22, pp. 3824-3826 (1989). |
| Spaleci et al., New J. Chem., "Stereorigid Metallocenes: Correlations Between Structure and Behavior in Homopolymerizations of Propylene", vol. 14, pp. 499-503, 1990. * |
| Spaleck et al., NEW J. CHEM., "Stereorigid Metallocenes: Correlations Between Structure and Behaviour in Homopolymerizations of Propylene", vol. 14, pp. 499-503 (1990). |
| Winter v. Fujita, 53 U.S.P.Q.2d 1234 (Bd of Appeals 1999). |
| Winter v. Fujita, 53 U.S.P.Q.2d 1478 (Bd of Appeals 2000). |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080200708A1 (en) * | 2002-10-25 | 2008-08-21 | Basell Polyolefine Gmbh | Preparation of Partially Hydrogenated Rac-Ansa-Metallocene Complexes |
| US7619106B2 (en) * | 2002-10-25 | 2009-11-17 | Basell Polyolefine Gmbh | Preparation of partially hydrogenated rac-ansa-metallocene complexes |
Also Published As
| Publication number | Publication date |
|---|---|
| USRE39532E1 (en) | 2007-03-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| USRE37208E1 (en) | Polyolefins prepared with metallocene catalysts having 2-substituted indenyl type ligands | |
| USRE37384E1 (en) | 2-substituted disindenylmetallocenes, process for their preparation, and their use as catalysts in the polymerization of olefins | |
| US5239022A (en) | Process for the preparation of a syndiotactic polyolefin | |
| US5243001A (en) | Process for the preparation of a high molecular weight olefin polymer | |
| JP3143174B2 (en) | Method for producing olefin polymer | |
| AU640286B2 (en) | Process for the preparation of a high molecular weight olefin polymer | |
| JP3234876B2 (en) | Bridged chiral metallocenes, process for their preparation and their use as catalysts | |
| JP3394997B2 (en) | Process for producing olefin polymers using metallocenes having specifically substituted indenyl ligands | |
| JP3371118B2 (en) | Metallocenes having benzene-fused indenyl derivatives as ligands, their preparation and their use as catalysts | |
| KR100261362B1 (en) | Process for preparing high molecular weight polyolefins | |
| US5504232A (en) | Process for the preparation of an olefin polymer using specific metallocenes | |
| US5268495A (en) | Metallocenes having bicyclic cyclopentadiene derivatives as ligands, processes for their preparation and their use as catalysts | |
| US5830821A (en) | Process for olefin preparation using metallocenes having benzo-fused indenyl derivatives as ligands | |
| EP0914323A1 (en) | Metallocenes and catalysts for polymerization of olefins | |
| US5932669A (en) | Metallocenes having benzo-fused indenyl derivatives as ligands, processes for their preparation and their use as catalysts | |
| USRE39561E1 (en) | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts | |
| US20110306741A1 (en) | Synthesis of substituted tetrahydroindenyl complexes | |
| USRE37573E1 (en) | Process for the preparation of an olefin polymer using metallocenes containing specifically substituted indenyl ligands | |
| JP2001519436A (en) | Ferrocenyl-substituted bridged metallocenes used in olefin polymerization |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: CITIBANK, N.A., AS COLLATERAL AGENT,NEW YORK Free format text: GRANT OF SECURITY INTEREST IN UNITED STATES PATENTS AND PATENT APPLICATIONS;ASSIGNORS:BASELL POLYOLEFINE GMBH;ARCO CHEMICAL TECHNOLOGY L.P.;ARCO CHEMICAL TECHNOLOGY, INC.;AND OTHERS;REEL/FRAME:020704/0562 Effective date: 20071220 Owner name: CITIBANK, N.A., AS COLLATERAL AGENT, NEW YORK Free format text: GRANT OF SECURITY INTEREST IN UNITED STATES PATENTS AND PATENT APPLICATIONS;ASSIGNORS:BASELL POLYOLEFINE GMBH;ARCO CHEMICAL TECHNOLOGY L.P.;ARCO CHEMICAL TECHNOLOGY, INC.;AND OTHERS;REEL/FRAME:020704/0562 Effective date: 20071220 |
|
| AS | Assignment |
Owner name: CITIBANK, N.A., AS COLLATERAL AGENT,NEW YORK Free format text: SECURITY AGREEMENT;ASSIGNORS:BASELL POLYOLEFINE GMBH;ARCO CHEMICAL TECHNOLOGY L.P.;ARCO CHEMICAL TECHNOLOGY, INC.;AND OTHERS;REEL/FRAME:021354/0708 Effective date: 20071220 Owner name: CITIBANK, N.A., AS COLLATERAL AGENT, NEW YORK Free format text: SECURITY AGREEMENT;ASSIGNORS:BASELL POLYOLEFINE GMBH;ARCO CHEMICAL TECHNOLOGY L.P.;ARCO CHEMICAL TECHNOLOGY, INC.;AND OTHERS;REEL/FRAME:021354/0708 Effective date: 20071220 |
|
| CC | Certificate of correction | ||
| AS | Assignment |
Owner name: EQUISTAR CHEMICALS, LP, TEXAS Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0856 Effective date: 20100430 Owner name: LYONDELL CHEMICAL TECHNOLOGY, L.P., DELAWARE Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0856 Effective date: 20100430 Owner name: EQUISTAR CHEMICALS, LP, TEXAS Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0705 Effective date: 20100430 Owner name: LYONDELL CHEMICAL TECHNOLOGY, L.P., DELAWARE Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0705 Effective date: 20100430 Owner name: EQUISTAR CHEMICALS, LP,TEXAS Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0856 Effective date: 20100430 Owner name: LYONDELL CHEMICAL TECHNOLOGY, L.P.,DELAWARE Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0856 Effective date: 20100430 Owner name: EQUISTAR CHEMICALS, LP,TEXAS Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0705 Effective date: 20100430 Owner name: LYONDELL CHEMICAL TECHNOLOGY, L.P.,DELAWARE Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0705 Effective date: 20100430 |