WO1991018027A2 - Compounds - Google Patents
Compounds Download PDFInfo
- Publication number
- WO1991018027A2 WO1991018027A2 PCT/GB1991/000788 GB9100788W WO9118027A2 WO 1991018027 A2 WO1991018027 A2 WO 1991018027A2 GB 9100788 W GB9100788 W GB 9100788W WO 9118027 A2 WO9118027 A2 WO 9118027A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polymer
- alkyl
- substitute sheet
- polymers
- nitrogen atom
- Prior art date
Links
- 0 CC(C)(CN)*(O)=O Chemical compound CC(C)(CN)*(O)=O 0.000 description 2
- XDTMQSROBMDMFD-UHFFFAOYSA-N C1CCCCC1 Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/30—Introducing nitrogen atoms or nitrogen-containing groups
- C08F8/32—Introducing nitrogen atoms or nitrogen-containing groups by reaction with amines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/74—Synthetic polymeric materials
- A61K31/785—Polymers containing nitrogen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/44—Preparation of metal salts or ammonium salts
Definitions
- the present invention relates to novel anion exchange polymers, processes for their preparation, pharmaceutical compositions containing them and their use in the lowering of plasma cholesterol levels in humans.
- Coronary Heart Disease is one of the most serious health problems of contemporary society.
- Worldwide epidemiological studies have shown that the incidence of CHD is related to a number of independent risk factors, in particular, for example, high concentrations of serum cholesterol (hypercholesterolaemia) .
- serum cholesterol hypercholesterolaemia
- Such adverse factors lead to atherosclerosis, and ultimately, in severe cases, intermittent claudication, cerebrovascular insufficiency, thrombosis and cardiac arrest.
- ion exchange polymers can be used as sequestering agents to bind bile acids and salts in the intestinal tract, forming complexes which are then excreted in the faeces. This sequestering leads to a decrease in the amount of bile acids returning to the liver via enterohepatic circulation.
- the synthesis of replacement bile acids from hepatic cholesterol depletes hepatic cholesterol, regulates hepatic LDL receptors and consequently reduces plasma cholesterol levels.
- Such sequestering polymers have been recognised as useful for the treatment of hypercholesterolaemia and it is now proven that reducing serum cholesterol with bile acid sequestrants has a beneficial effect on protecting against the occurrence of coronary heart disease.
- SUBSTITUTE SHEET One particular agent which is currently used to lower serum cholesterol levels in humans by binding bile acids in the intestinal tract is cholestyramine.
- Cholestyramine is a cross-linked anion exchange polystyrene polymer bearing an ionisable trimethylammonium group bound to the polymer backbone.
- this agent is associated with a number of undesirable side-effects, for example, it is unpalatable and must be taken in high doses and causes, in some cases, bloating, constipation and other gut side- effects.
- its ability to bind bile acids is inefficient with respect to the amounts of resin which it is necessary to use (up to 36 g per person per day) .
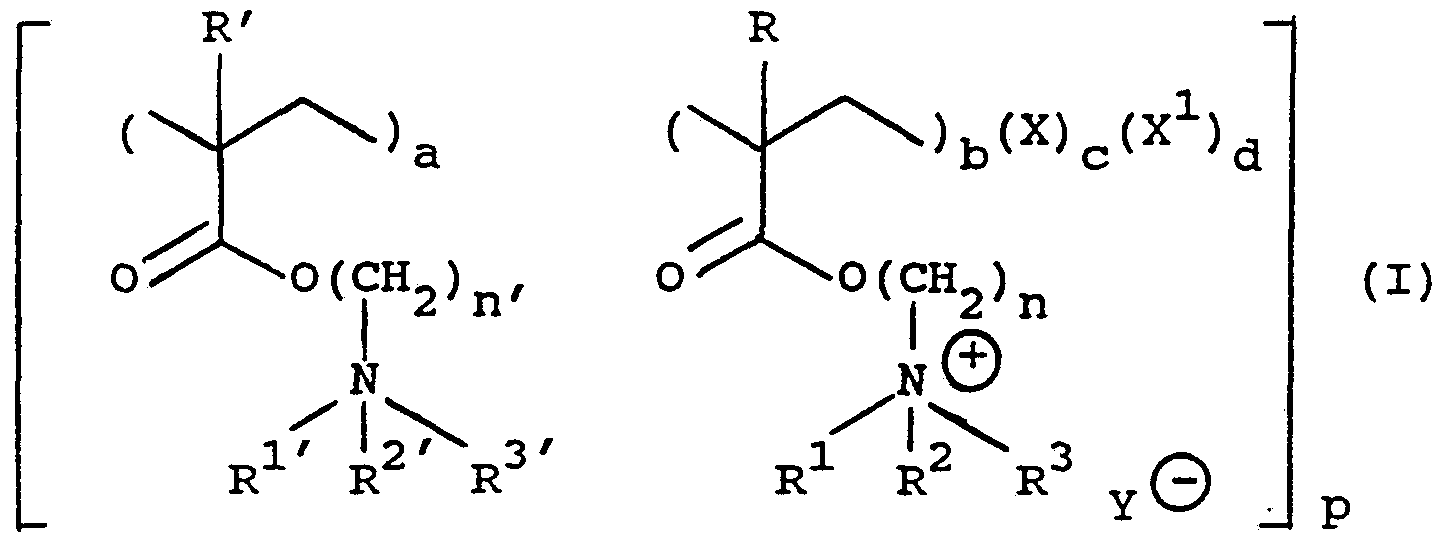
- the present invention therefore provides in a first aspect, cross-linked polymers of structure (I)
- a, b, c and d indicate the relative molar percentages of the units present in the polymer, (a) and (b) together being from about 25 to about 99.5 molar percent, and (c) being from about 0.5 to about 8 molar percent;
- X is a cross-linking unit
- X 1 is a comonomer unit
- R and R' are the same or different and are each hydrogen or C 1 _ alkyl
- R 1 and R 2 are the same or different and are each C 1 _ 4 alkyl, and R 3 is C 1 _ 20 alkyl or C- L _ 20 aralkyl; or R 1 is C _ 4 alkyl and R 2 and R 3 together with the nitrogen atom to which they are attached form a saturated ring, optionally containing one or more further heteroatoms; or R 1 to R 3 together with the nitrogen atom to which they are attached form an unsaturated ring, optionally containing one or more further heteroatoms;
- SUBSTITUTE SHEET R 1 ' and R 2 ' are the same or different and are each
- R 3 is C 1 _ 20 alkyl or C 1 _ 20 aralk y 1 or R 1 ' is C 1 _ 4 alkyl and R 2 ' and R 3 ' together with the nitrogen atom to which they are attached form a saturated ring, optionally containing one or more further heteroatoms; or R 1 ' to R 3 ' together with the nitrogen atom to which they are attached form an unsaturated ring, optionally containing one or more further heteroatoms;
- n and n' are the same or different and are each 1 to 20;
- p is a number indicating the degree of polymerisation of the polymer
- Y ⁇ is a physiologically acceptable counter ion.
- (a) plus (b) is from about 25 to about 99.5 molar percent; preferably from about 60 to about 99.5 molar percent.
- (c) is from about 0.5 to about 8 molar percent; preferably from about 0.5 to about 5.0 molar percent.
- X is a cross-linking unit i.e. a unit which provides a random distribution of cross-links between chains of polymers.
- SUBSTITUTE SHEET Preferred such units include, for example, divinylbenzene, alkylene glycol bis methacrylates of structure (i)
- z is l to 4, preferably z is 1.
- m is 2 to 6, preferably m is 2.
- X 1 is a comonomer unit.
- X 1 is styrene, an alkyl alkylate of structure (ii) or an alkylstyrene of structure (iii)
- R and d are as described for structure (I) and R 4 is C 1 _ 0 alkyl.
- R is preferably methyl and R 4 is preferably C 6 _ 12 alkyl.
- R 1 to R 3 or R 1 ' to R 3 ' together with the nitrogen atom to which they are attached form an unsaturated ring optionally containing one or more further heteroatoms.
- Suitable examples of such rings unsaturated 5 or 6 membered rings such as imidazolyl and pyridyl. More suitably, R 1 or R 1 is C 1 _ 4 alkyl and R 2 and R 3 or R 2 ' and R 3 together with the nitrogen atom to which they are attached form a saturated ring optionally containing one or more further heteroatoms.
- saturated rings include, for example, morpholino, piperidino and piperazino rings, and in addition, bicyclic rings i.e. those in which the R 1 or
- R J group forms a bridge between the two nitrogen atoms of a saturated ring e.g.
- R 1 and R 2 or R 1 and R 2 ' are the same or different and are each C 1 _ alkyl; more preferably R 1 and
- R 2 or R 1' and R are the same and are each C 1-4 alkyl, in particular methyl; and R 3 or R 3 ' is C 1 _ 20 alkyl or C 1 _ 0 aralkyl, preferably C 1 _ 0 alkyl, most preferably ⁇ l -" .2 --- ⁇ - r ⁇ ' - P rt i cu l ar C 12 alkyl.
- n is 1 to 20; preferably n is 8 to 20; most preferably n is 8 to 12.
- n' is 1 to 20; preferably n' is 8 to 20; most preferably n' is 8 to 12.
- p is a number indicating the degree of polymerisation of the polymer. Owing to the three dimensional cross- linkage, precise figures cannot be given for p, but in any case will be greater than 1,000.
- Y ⁇ is a physiologically acceptable counter ion such as a bicarbonate, carbonate, formate, acetate, sulphonate, propionate, malonate, succinate, maleate, tartrate, citrate, maleate, fumarate, ascorbate, sulphate, phosphate, halide or glucuronate; or the anion of an a ino acid such as aspartic or glutamic acid.
- Y ⁇ is a sulphate, phosphate or halide ion; more preferably a halide ion, in particular a chloride ion.
- C 1-4 alkyl and C 1 _ 20 alkyl groups as herein defined include both straight and branched alkyl groups.
- the polymers of the present invention are also characterised by their total exchange capacity i.e. the theoretical maximum capacity of the resin if each counter ion were to be exchanged with bile acid.
- the total exchange capacity is defined in terms of the number of milliequivalent ⁇ of counter ion per gram of dry weight of polymer.
- Suitable total exchange capacities are in the range of, for example where the counter ion Y ⁇ is chlorine,
- SUBSTITUTE SHEET from about 1.5 to about 5.0 meq Cl ⁇ per gram of resin.
- Preferred within this range are polymers having a total exchange capacity of between 2 and 3 meq Cl ⁇ /gram of resin.
- bile acid when used herein shall be taken to include bile acids, bile salts and conjugates thereof.
- the polymers of the present invention can be prepared by processes analogous to those known in the art.
- the present invention therefore provides, in a further aspect, a process for preparing the polymers of structure (I) which comprises reaction of a polymer of structure (II)
- R- are as described for structure (I)
- Suitable groups Z displaceable by an amine will be apparent to those skilled in the art and include for example halogen, such as bromine.
- the reaction between a polymer of structure (II) and a compound of structure R 1 R 2 R 3 N and a compound of structure R 1, R 2 'R 3 'N can be carried out in a suitable solvent at elevated temperature.
- suitable solvents include for example, a C j ⁇ al a ol such as ethanol, N-methyl- pyrrolidone, dimethylformamide, tetrahydrof ran, nitro- ethane or sulpholane.
- the reaction is carried out in methanol at a temperature of about 40° for a period of up to 24 hours or until the reaction is complete.
- the intermediate polymers of structure (II) can be prepared from readily available materials by methods known to those skilled in the art.
- polymers of structure (II) in which X is a cross-link of structure (i) in which 2 is 1 and is 2, and Z is bromine and R is methyl can be prepared by reaction of the appropriate bromo alkyl methacrylate, ethylene glycol bis methacrylate, and, optionally, for example, a C 1 _ 20 alkyl alkacrylate (if a comonomer unit X 1 is desired in the final polymer) in an aqueous suspension comprising polyvinyl alcohol in the presence of an initiator at elevated temperature.
- chloro or bromo alkyl methacrylates can be prepared by reaction of the corresponding chloro- or bromoalkanol and methacrylic anhydride in the presence of 4-dimethylaminopyridine (DMAP) in a suitable solvent such as pyridine, or by reaction of the corresponding chloro or bromo alkanol with methacryloyl chloride in the presence of a base in a suitable solvent -
- DMAP 4-dimethylaminopyridine
- SUBSTITUTE SHEET suitable combinations of bases and solvents include, for example sodium bicarbonate in petroleum spirit as a solvent and pyridine as a base in toluene as solvent (cf. method described in Polymer (1987) , _l__, 325-331, and Br.Polymer J. (1984) 16, 39-45) .
- the polymers of structure (I) have been found to bind bile acids both in in vitro and in in vivo models. As indicated earlier it is recognised that removal of bile acids from the intestinal tract in this way lowers serum cholesterol levels and also has a beneficial effect on protecting against atherosclerosis and its dependent clinical conditions.
- the present invention therefore provides in a further aspect, polymers of structure (I) for use in therapy, in particular for the lowering of serum cholesterol levels in mammals, including humans.
- the polymers of structure (I) are expected to be of use in protecting against atherosclerosis and its sequelae, and for example, in the treatment of pruritus and diarrhoea.
- polymers of structure (I) When used in therapy polymers of structure (I) are in general administered in a pharmaceutical composition.
- a pharmaceutical composition comprising a polymer of structure (I) in association with a pharmaceutically acceptable carrier.
- compositions of the present invention can be prepared by techniques well known to those skilled in the art of pharmacy.
- polymers are preferably administered as formulations in admixture with one or more conventional pharmaceutical excipients which are physically and chemically compatible with the polymer, which are non- toxic, are without deleterious side-effects but which confer appropriate properties on the dosage form.
- aqueous pharmaceutically acceptable carriers such as water itself or aqueous dilute ethanol, propylene glycol, polyethylene glycol or glycerol or sorbitol solutions are preferred.
- Such formulations can also include preservatives and flavouring and sweetening agents such as sucrose, fructose, invert sugar, cocoa, citric acid, ascorbic acid, fruit juices etc.
- preservatives and flavouring and sweetening agents such as sucrose, fructose, invert sugar, cocoa, citric acid, ascorbic acid, fruit juices etc.
- digestible oil or fat based carriers should be avoided or minimised as they contribute to the condition sought to be alleviated by use of the polymers. They are also subject to absorption by the polymers during prolonged contact, thus reducing the capacity of the polymer to absorb bile acids after administration.
- the polymers can also be prepared as 'concentrates' , for dilution prior to administration, and as formulations suitable for direct oral administration. They can be administered orally ad libitum, on a relatively continuous basis for example by dispersing the polymer in water, drinks or food, for example in a granule presentation suitable for admixture with water or other drink to provide a palatable drinking suspension.
- the polymers are administered in tablet form or in gelatin capsules containing solid particulate polymer or a non-aqueous suspension of solid polymer
- SUBSTITUTE SHEET containing a suitable suspending agent.
- suitable excipients for such formulations will be apparent to those skilled in the art and include, for example, for tablets and capsules, lactose, microcrystalline cellulose, magnesium, stearate, povidone, sodium starch, glycollate and starches; and for suspensions in capsules, polyethylene glycol, propylene glycol and colloidal silicone dioxide. If desired these dosage forms in addition optionally comprise suitable flavouring agents. Alternatively, a chewable tablet or granule presentation incorporating suitable flavouring and similar agents may be used.
- the polymer is administered in unit dosage form, each dosage unit containing preferably from 0.5 g to 1.5 g of polymer.
- the daily dosage regimen for an adult patient may be, for example, a total daily oral dose of between 1 and 10 g, preferably 1-5 g, the compound being administered 1 to 4 times a day.
- the compound is administered for a period of continuous therapy of one month or more sufficient to achieve the required reduction in serum cholesterol levels.
- polymers of the present invention can be co-administered (together or sequentially) with further active ingredients such as HMGCoA reductase inhibitors and other hypocholesterolaemic agents, and other drugs for the treatment of cardiovascular diseases.
- SUBSTITUTE SHEET was determined by elemental analysis and/or potentiometric titration of chloride ion. Figures quoted are expressed as milli equivalents of exchangeable chloride ion per gram of dry resin weight; and the percent cross-linking values given are based on the ratios of the starting monomers used in the polymerisation stage.
- a chewable tablet composition can be prepared from the following : mg/tablet
- a food additive composition for example, a sachet for reconstitution or mixing with food, is prepared by incorporating into a powder formulation compound of structure (I) (250 mg) , sodium carboxymethylcellulose (50 mg) , sucrose (2400 mg) and flavours (50 mg) .
- the lower the % dissociation the more efficient the polymer can be expected to be in extracting bile acids in vivo.
- Test compound 150 mg is equilibrated with 5mM sodium glycocholate (30 ml) in Krebs' buffer. The compound is separated by centrifugation and the total bound determined by subtraction of the amount in the supernatant from the total bile acid used. Dissociation is measured by resuspending the compound in Krebs' buffer, shaking and sampling the mixture through a filter at several time points up to 20 minutes. Radioactivity and hence bile acid dissociated is determined in the filtrate.
Abstract
Description
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP91509109A JPH05506880A (en) | 1990-05-21 | 1991-05-20 | Compound |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB909011332A GB9011332D0 (en) | 1990-05-21 | 1990-05-21 | Compounds |
| GB9011332.5 | 1990-05-21 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO1991018027A2 true WO1991018027A2 (en) | 1991-11-28 |
| WO1991018027A3 WO1991018027A3 (en) | 1992-01-09 |
Family
ID=10676309
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/GB1991/000788 WO1991018027A2 (en) | 1990-05-21 | 1991-05-20 | Compounds |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP0530242A1 (en) |
| JP (1) | JPH05506880A (en) |
| AU (1) | AU7867491A (en) |
| GB (1) | GB9011332D0 (en) |
| WO (1) | WO1991018027A2 (en) |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995034588A1 (en) * | 1994-06-10 | 1995-12-21 | Geltex Pharmaceuticals, Inc. | Cross-linked polymers for removing bile salts from a patient |
| US5556619A (en) * | 1992-08-20 | 1996-09-17 | The Du Pont Merck Pharmaceutical Company | Crosslinked polymeric ammonium salts |
| US5607669A (en) * | 1994-06-10 | 1997-03-04 | Geltex Pharmaceuticals, Inc. | Amine polymer sequestrant and method of cholesterol depletion |
| US5618530A (en) * | 1994-06-10 | 1997-04-08 | Geltex Pharmaceuticals, Inc. | Hydrophobic amine polymer sequestrant and method of cholesterol depletion |
| US5665348A (en) * | 1992-01-14 | 1997-09-09 | Hisamitsu Pharmaceutical Co, Inc. | Cholesterol-lowering drug |
| US5679717A (en) * | 1994-06-10 | 1997-10-21 | Geltex Pharmaceuticals, Inc. | Method for removing bile salts from a patient with alkylated amine polymers |
| US5703188A (en) * | 1993-06-02 | 1997-12-30 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and compositions therefor |
| FR2757866A1 (en) * | 1996-12-30 | 1998-07-03 | Catalyse | POLYMERS COMPRISING QUATERNARY AMMONIUMS GROUPS, THEIR USE FOR THE MANUFACTURE OF ANTIBACTERIAL PROPERTY MATERIAL AND METHODS FOR THEIR PREPARATION |
| US5800809A (en) * | 1992-01-14 | 1998-09-01 | Hisamitsu Pharmaceutical Co, Inc | Non-crosslinked acrylic polymers and non-crosslinked anion exchange resins |
| US5874522A (en) * | 1992-08-20 | 1999-02-23 | Dupont Pharmaceuticals Company | Crosslinked polymeric ammonium salts |
| US5900475A (en) * | 1994-06-10 | 1999-05-04 | Geltex Pharmaceuticals, Inc. | Hydrophobic sequestrant for cholesterol depletion |
| US5925379A (en) * | 1997-03-27 | 1999-07-20 | Geltex Pharmaceuticals, Inc. | Interpenetrating polymer networks for sequestration of bile acids |
| US5929184A (en) * | 1993-06-02 | 1999-07-27 | Geltex Pharmaceuticals, Inc. | Hydrophilic nonamine-containing and amine-containing copolymers and their use as bile acid sequestrants |
| US6129910A (en) * | 1993-06-02 | 2000-10-10 | Geltex Pharmaceuticals, Inc. | Water-insoluble noncrosslinked bile acid sequestrants |
| US6203785B1 (en) | 1996-12-30 | 2001-03-20 | Geltex Pharmaceuticals, Inc. | Poly(diallylamine)-based bile acid sequestrants |
| US6423754B1 (en) | 1997-06-18 | 2002-07-23 | Geltex Pharmaceuticals, Inc. | Method for treating hypercholesterolemia with polyallylamine polymers |
| US6726905B1 (en) | 1997-11-05 | 2004-04-27 | Genzyme Corporation | Poly (diallylamines)-based phosphate binders |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006052236A (en) * | 2002-07-30 | 2006-02-23 | Yukio Nagasaki | New compound having cholesterol-reducing activity |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2843573A (en) * | 1955-03-21 | 1958-07-15 | Rohm & Haas | New quaternary ammonium compounds in which the nitrogen atom carries an alkoxymethyl group |
| US3787474A (en) * | 1969-09-12 | 1974-01-22 | Inveresk Res Int | Cross-linked polymers |
| US4393145A (en) * | 1979-05-01 | 1983-07-12 | Etablissement Texcontor | Anionic ion exchange resins with cholesterol-decreasing properties |
| FR2528435A1 (en) * | 1982-06-09 | 1983-12-16 | Inst Francais Du Petrole | Additive lowering middle distillate cloud pt. - is reaction prod. of polymerised unsaturated carboxylic acid or its ester with amine |
-
1990
- 1990-05-21 GB GB909011332A patent/GB9011332D0/en active Pending
-
1991
- 1991-05-20 WO PCT/GB1991/000788 patent/WO1991018027A2/en not_active Application Discontinuation
- 1991-05-20 JP JP91509109A patent/JPH05506880A/en active Pending
- 1991-05-20 EP EP91909433A patent/EP0530242A1/en not_active Withdrawn
- 1991-05-20 AU AU78674/91A patent/AU7867491A/en not_active Abandoned
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2843573A (en) * | 1955-03-21 | 1958-07-15 | Rohm & Haas | New quaternary ammonium compounds in which the nitrogen atom carries an alkoxymethyl group |
| US3787474A (en) * | 1969-09-12 | 1974-01-22 | Inveresk Res Int | Cross-linked polymers |
| US4393145A (en) * | 1979-05-01 | 1983-07-12 | Etablissement Texcontor | Anionic ion exchange resins with cholesterol-decreasing properties |
| FR2528435A1 (en) * | 1982-06-09 | 1983-12-16 | Inst Francais Du Petrole | Additive lowering middle distillate cloud pt. - is reaction prod. of polymerised unsaturated carboxylic acid or its ester with amine |
Cited By (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5800809A (en) * | 1992-01-14 | 1998-09-01 | Hisamitsu Pharmaceutical Co, Inc | Non-crosslinked acrylic polymers and non-crosslinked anion exchange resins |
| US5665348A (en) * | 1992-01-14 | 1997-09-09 | Hisamitsu Pharmaceutical Co, Inc. | Cholesterol-lowering drug |
| US5556619A (en) * | 1992-08-20 | 1996-09-17 | The Du Pont Merck Pharmaceutical Company | Crosslinked polymeric ammonium salts |
| US5874522A (en) * | 1992-08-20 | 1999-02-23 | Dupont Pharmaceuticals Company | Crosslinked polymeric ammonium salts |
| US5726284A (en) * | 1992-08-20 | 1998-03-10 | The Dupont Merck Pharmaceutical Company | Crosslinked polymric ammonium salts |
| US6129910A (en) * | 1993-06-02 | 2000-10-10 | Geltex Pharmaceuticals, Inc. | Water-insoluble noncrosslinked bile acid sequestrants |
| US6060517A (en) * | 1993-06-02 | 2000-05-09 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and compositions therefor |
| US5624963A (en) * | 1993-06-02 | 1997-04-29 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and compositions therefor |
| US5929184A (en) * | 1993-06-02 | 1999-07-27 | Geltex Pharmaceuticals, Inc. | Hydrophilic nonamine-containing and amine-containing copolymers and their use as bile acid sequestrants |
| US5703188A (en) * | 1993-06-02 | 1997-12-30 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and compositions therefor |
| US5840766A (en) * | 1993-06-02 | 1998-11-24 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and compositions therefor |
| US5919832A (en) * | 1994-06-10 | 1999-07-06 | Geltex Pharmaceuticals Inc. | Amine polymer sequestrant and method of cholesterol depletion |
| US6066678A (en) * | 1994-06-10 | 2000-05-23 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and alkylated compositions therefor |
| US7399821B2 (en) | 1994-06-10 | 2008-07-15 | Genzyme Corporation | Alkylated poly(allylamine) polymers and methods of use |
| US7101960B2 (en) | 1994-06-10 | 2006-09-05 | Genzyme Corporation | Process for removing bile salts from a patient and alkylated compositions therefor |
| US5693675A (en) * | 1994-06-10 | 1997-12-02 | Geltex Pharmaceuticals Inc. | Alkylated amine polymers |
| US5900475A (en) * | 1994-06-10 | 1999-05-04 | Geltex Pharmaceuticals, Inc. | Hydrophobic sequestrant for cholesterol depletion |
| US5917007A (en) * | 1994-06-10 | 1999-06-29 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and alkylated compositions therefor |
| WO1995034588A1 (en) * | 1994-06-10 | 1995-12-21 | Geltex Pharmaceuticals, Inc. | Cross-linked polymers for removing bile salts from a patient |
| US6784254B2 (en) | 1994-06-10 | 2004-08-31 | Genzyme Corporation | Process for removing bile salts from a patient and alkylated compositions therefor |
| US5679717A (en) * | 1994-06-10 | 1997-10-21 | Geltex Pharmaceuticals, Inc. | Method for removing bile salts from a patient with alkylated amine polymers |
| US5969090A (en) * | 1994-06-10 | 1999-10-19 | Geltex Pharmaceuticals, Inc. | Hydrophobic sequestrant for cholesterol depletion |
| US5981693A (en) * | 1994-06-10 | 1999-11-09 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and alkylated compositions therefor |
| US5618530A (en) * | 1994-06-10 | 1997-04-08 | Geltex Pharmaceuticals, Inc. | Hydrophobic amine polymer sequestrant and method of cholesterol depletion |
| AU694777B2 (en) * | 1994-06-10 | 1998-07-30 | Genzyme Corporation | Cross-linked polymers for removing bile salts from a patient |
| US5607669A (en) * | 1994-06-10 | 1997-03-04 | Geltex Pharmaceuticals, Inc. | Amine polymer sequestrant and method of cholesterol depletion |
| US6433026B2 (en) | 1994-06-10 | 2002-08-13 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and alkylated compositions therefor |
| US6225355B1 (en) | 1994-06-10 | 2001-05-01 | Geltex Pharmaceuticals, Inc. | Process for removing bile salts from a patient and alkylated compositions therefor |
| US6251967B1 (en) | 1996-12-30 | 2001-06-26 | Catalyse (Sarl) Limited Company | Antimicrobial polymers comprising quaternary ammonium groups, their use for making a material with antimicrobial properties and methods for preparing them |
| US6203785B1 (en) | 1996-12-30 | 2001-03-20 | Geltex Pharmaceuticals, Inc. | Poly(diallylamine)-based bile acid sequestrants |
| US6610283B1 (en) | 1996-12-30 | 2003-08-26 | Genzyme Corporation | Poly(diallylamine)-based bile acid sequestrants |
| FR2757866A1 (en) * | 1996-12-30 | 1998-07-03 | Catalyse | POLYMERS COMPRISING QUATERNARY AMMONIUMS GROUPS, THEIR USE FOR THE MANUFACTURE OF ANTIBACTERIAL PROPERTY MATERIAL AND METHODS FOR THEIR PREPARATION |
| US7125547B2 (en) | 1996-12-30 | 2006-10-24 | Genzyme Corporation | Poly(diallylamine)-based bile acid sequestrants |
| WO1998029463A1 (en) * | 1996-12-30 | 1998-07-09 | Catalyse | Antimicrobial polymers comprising quaternary ammonium groups, their use for making a material with antimicrobial properties and methods for preparing them |
| US5925379A (en) * | 1997-03-27 | 1999-07-20 | Geltex Pharmaceuticals, Inc. | Interpenetrating polymer networks for sequestration of bile acids |
| US6423754B1 (en) | 1997-06-18 | 2002-07-23 | Geltex Pharmaceuticals, Inc. | Method for treating hypercholesterolemia with polyallylamine polymers |
| US6726905B1 (en) | 1997-11-05 | 2004-04-27 | Genzyme Corporation | Poly (diallylamines)-based phosphate binders |
Also Published As
| Publication number | Publication date |
|---|---|
| WO1991018027A3 (en) | 1992-01-09 |
| EP0530242A1 (en) | 1993-03-10 |
| AU7867491A (en) | 1991-12-10 |
| GB9011332D0 (en) | 1990-07-11 |
| JPH05506880A (en) | 1993-10-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0373852B1 (en) | Compounds | |
| WO1991018027A2 (en) | Compounds | |
| EP0432995B1 (en) | Crosslinked pyridinio-acrylate-polymers | |
| EP0375350B1 (en) | Cross-linked vinylpyridinium polymers | |
| JP4420143B2 (en) | Polyallylamine polymers for the treatment of hypercholesterolemia | |
| US5840766A (en) | Process for removing bile salts from a patient and compositions therefor | |
| EP0764177B1 (en) | Crosslinked polymer containing composition for removing bile salts from a patient | |
| WO1994027620A1 (en) | Compositions and process for removing bile salts | |
| US5273740A (en) | Polystyrene anion exchange polymers | |
| US4954339A (en) | Novel polystyrene anion exchange polymers | |
| EP0402062A2 (en) | Compounds | |
| US5110875A (en) | Polystyrene anion exchange polymers | |
| EP0403271A2 (en) | Polymer compounds | |
| US5230885A (en) | Polystyrene anion exchange polymer pharmaceutical composition | |
| US5112922A (en) | Polystyrene anion exchange polymers | |
| AU608114B2 (en) | Compounds | |
| EP0385686A2 (en) | Compounds |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AU CA JP KR US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): AT BE CH DE DK ES FR GB GR IT LU NL SE |
|
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AU CA JP KR US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): AT BE CH DE DK ES FR GB GR IT LU NL SE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1991909433 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1991909433 Country of ref document: EP |
|
| NENP | Non-entry into the national phase in: |
Ref country code: CA |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1991909433 Country of ref document: EP |