WO1996017681A1 - Absorbent material - Google Patents
Absorbent material Download PDFInfo
- Publication number
- WO1996017681A1 WO1996017681A1 PCT/US1995/015139 US9515139W WO9617681A1 WO 1996017681 A1 WO1996017681 A1 WO 1996017681A1 US 9515139 W US9515139 W US 9515139W WO 9617681 A1 WO9617681 A1 WO 9617681A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- superabsorbent
- superabsorbent material
- cationic
- groups
- polymer
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/60—Liquid-swellable gel-forming materials, e.g. super-absorbents
Definitions
- the present invention relates to an absorbent material, more particularly a material of the type commonly referred to as a "superabsorben " .
- superabsorbents are typically slightly cross-linked hydrophilic polymers.
- the polymers may differ in their chemical nature but they share the property of being capable of absorbing and retaining even under moderate pressure amounts of aqueous fluids equivalent to many times their own weight.
- superabsorbents can typically absorb up to 100 times their own weight or even more of distilled water.
- Superabsorbents have been suggested for use in many different industrial applications where advantage can be taken of their water absorbing and/or retaining properties and examples include agriculture, the building industry, the production of alkaline batteries and filters.
- the primary field of application for superabsorbents is in the production of hygienic and/or sanitary products such as disposable sanitary napkins and disposable diapers either for children or for incontinent adults.
- superabsorbents are used, generally in combination with cellulose fibres, to absorb body fluids such as menses or urine.
- body fluids such as menses or urine.
- the absorbent capacity of superabsorbents for body fluids is dramatically lower than for deionised water. It is generally believed that this effect results from the electrolyte content of body fluids and the effect is often referred to as "salt poisoning" .
- the water absorption and water retention characteristics of superabsorbents are due to the presence in the polymer structure of ionisable functional groups. These groups are usually carboxyl groups, a high proportion of which are in the salt form when the polymer is dry but which undergo dissociation and solvation upon contact with water. In the dissociated state, the polymer chain will have a series of functional groups attached to it which groups have the same electric charge and thus repel one another. This leads to expansion of the polymer structure which, in turn, permits further absorption of water molecules although this expansion is subject to the constraints provided by the cross-links in the polymer structure which must be sufficient to prevent dissolution of the polymer.
- Japanese Patent Application OPI No. 57-45,057 discloses an absorbent which comprises a mixture of a superabsorbent such as a cross-linked polyacrylate with an ion exchange resin in powder or granular form.
- EP-A-0210756 relates to an absorbent structure comprising a superabsorbent and an anion exchanger, optionally together with a cation exchanger, wherein both ion exchangers are in fibrous form.
- Combining a superabsorbent with an ion exchanger attempts to alleviate the salt poisoning effect by using the ion exchanger to reduce the salt content of the liquid.
- the ion exchanger has no direct effect on the performance of the superabsorbent and it may not be possible to reduce the salt content sufficiently to have the desired effect on the overall absorption capacity of the combination.
- the ion exchanger has no absorbing effect itself and thus acts as a diluent to the superabsorben .
- EP-A-0487975 relates to a cross-linked ampholytic copolymer said to be highly absorbent to aqueous electrolyte solutions and formed from an ampholytic ion pair monomer, a co-monomer and a cross-linking agent. It is assumed that when the ampholytic ion pair monomer is incorporated into the polymer backbone the ion pairs act as ionic cross-links which remain intact in deionised water but are broken in salt solution. Accordingly the copolymer is sensitive to the ionic strength of the solution in the sense that the effective degree of cross-linking is reduced as the ionic strength increases.
- EP-A-0161762 relates to a water swellable, water insoluble polymer produced by inverse suspension polymerisation of a diallylic ammonium salt monomer, an acrylic monomer and a cross-linking agent.
- the product is an acrylic acid polymer containing both cationic and anionic groups in the chain which is intended for use as a superabsorbent in salt form. It is claimed that the material can absorb the same quantity of water irrespective of the salt content of the water but absorption is at a low level and the material does not show any significant improvement in its water absorption in the presence of salt as compared to conventional superabsorbents.
- EP-A-01S1763 relates to a similar superabsorbent made by polymerising a diallyl ammonium compound and a cross-linking agent by suspension polymerisation.
- WO 92/20735 relates to a superabsorbent which is said to be substantially tolerant to salt solutions and which comprises a swellable hydrophobic polymer and an ionizable surfactant.
- the specification also discloses (but does not claim ) an alternative embodiment which uses a cationic superabsorbent which exchanges Cl " with OH" and an anionic superabsorbent which exchanges Na* with H*. No working examples of such a system are given and the superabsorbent gels disclosed are generally acrylamide derivatives.
- Acrylamide derivatives include the amide bond which is subject to hydrolysis at low alkaline pH (about pH 8) with release of toxic hydrolysis products. Hydrolysis problems will be exacerbated if the polymer is prepared and used in base form. An alkaline pH of about 8 may well arise in baby urine if fermentation of urea to ammonia takes place so that tissue hydrolysis products would be liable to be formed from acrylamide derivatives in contact with urine at this pH.
- An object of the present invention is to provide a superabsorbent with improved performance in the presence of electrolyte, for example in the case of menses or urine.
- the present invention provides a superabsorbent material which comprises a combination of
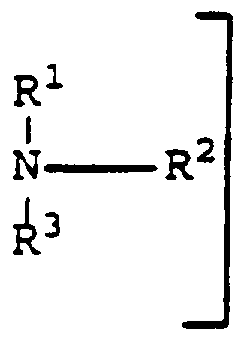
- a cationic superabsorbent in which from 20 to 1 00% of the functional groups are in basic form, the cationic superabsorbent being based on a polysaccharide or a polymer of units of a monomer of formula (I) :
- R and R 2 which may be the same or different, are each organic radicals which do not adversely affect the properties of the polymer and X is a suitable anion.
- the anionic superabsorbent preferably has 50 to 100% and more preferably has substantially 100% of the functional groups in free acid form.
- the cationic superabsorbent preferably has 50 to 100% and more preferably has substantially 100% of the functional groups in basic form.
- both anionic and cationic superabsorbents have to have functional groups in salt form before they act as superabsorbents .
- Commercially available superabsorbents are usually available in salt form. It has now surprisingly been found according to the present invention that a combination of an anionic superabsorbent in free acid form with a cationic superabsorbent as defined above in basic form is particularly effective as a superabsorbent in the case of electrolyte containing solutions, for example menses and urine.
- the anionic and the cationic superabsorbent are both converted from a non-absorbing form into the salt forms in which they act as superabsorbents;
- anionic superabsorbent does not behave as an ion exchanger in the sense that contacting the material alone in acid form with an electrolyte containing solution does not result in conversion to the salt form.
- the functional groups in anionic superabsorbents are typically carboxyl groups which act as a weak acid which does not dissociate when placed, for example, in a sodium chloride solution.
- presence of the cationic superabsorbent has the effect of attaching chloride ions from sodium chloride solution, thereby displacing the equilibrium in favour of conversion of the anionic superabsorbent into the salt form.
- the anionic superabsorbent can be any material having superabsorbent properties in which the functional groups are anionic, namely sulphonic groups, sulphate groups, phosphate groups or carboxyl groups.
- the functional groups are carboxyl groups.
- the functional groups are attached to a slightly cross-linked acrylic base polymer.
- the base polymer may be a polyacrylamide, polyvinyl alcohol, ethylene maleic anhydride copolymer, polyvinylether, polyvinyl sulphonic acid, polyacrylic acid, polyvinylpyrrolidone and polyvinylmorpholine. Copolymers of these monomers can also be used.
- Starch and cellulose based polymers can also be used including hydroxypropyl cellulose, carboxymethyl cellulose and acrylic grafted starches.
- Particular base polymers include cross-linked polyacrylates, hydrolysed acryloni rile grafted starch, starch polyacrylates, and isobutylene maleic anhydride copolymers.
- Particularly preferred base polymers are starch polyacrylates and cross-linked polyacrylates.
- the functional groups will generally be carboxyl grou DS .
- anionic superabsorbents are available commercially, for example Dow 2090 (Dow) , Favor 922 (Stockhausen) , Sanwet IM 1500 (Sanyo) , Aqualon AON D3236 (Aqualon Company) .
- Commercially available anionic superabsorbents are generally sold in salt form and need to be converted to the free acid form for use according to the invention, for example, Favor 922 may be swelled in water, acidified with HCl (0.01m), washed with water to remove excess HCl and dried in an air ventilated oven to obtain Favor 922 in acid form (FAVOR H) as follows:
- the anionic superabsorbent may be directly synthesized in acid form by the radical polymerization of the acrylic acid monomer with a crosslinking agent, namely in the same manner as commercially available superabsorbents are synthesized.
- the cationic superabsorbent can also be a material formed from a polysaccharide based polymer as described above for the anionic superabsorbent but with cationic functional groups.
- the cationic superabsorbent may be based on a polymer of units of a monomer of formula (I) :
- R 1 and R 2 wwhhiicchh mmaayy bbee the same or different, are each organic radicals which do not adversely affect the properties of the polymer and X is a suitable anion.
- R 1 and R 2 are each independently an optionally substituted saturated hydrocarbon group or aryl group.
- the saturated hydrocarbon group may be an alkyl group which may be straight or branched chain or cyclic.
- the aryl group also includes arylalkyl groups.
- the groups R 1 and R 2 have from l to 20 carbon atoms, more preferably from 1 to 6 carbon atoms.
- the saturated hydrocarbon groups or the aryl groups may be substituted by one or more suitable substituents selected from carboxyl, ester, hydroxyl, ether, sulphate, sulphonate, primary, secondary or tertiary amines or quaternary ammonium groups.
- R group is a hydrocarbon radical having from l to 20, preferably from 1 to 6 carbon atoms, more preferably the R group is methyl.
- suitable substituents include saturated hydrocarbon groups as defined above.
- the preferred groups for R 1 and R 2 are methyl groups.
- X may be any suitable anion which may be inorganic or organic.
- suitable inorganic anions include halide (in particular fluoride, chloride, bromide and iodide) , nitrate, phosphate, nitrite, carbonate, bicarbonate, borate, sulphate and hydroxide.
- Suitable organic anions include carboxylate such as acetate, citrate, salicilate and propionate.
- the anion is a chloride or hydroxide ion.
- Preferred monomers are diallyl dimethyl ammonium chloride and dimethyl diallyl ammonium hydroxide.
- cationic superabsorbents used according to the present inventions are resistant to hydrolysis at low alkaline pH and thus are not subject to the problems with release of toxic hydrolysis products referred to above in the context of the acrylamide derivatives suggested by WO 92/20735.
- suitable cationic functional groups include primary, secondary or tertiary amine groups or quaternary ammonium groups which should be present in base form. Preferably quaternary ammonium groups are used.
- Preferred base polymers include pblysaccharides and polymers based on dimethyldiallyl ammonium chloride.
- the cationic superabsorbent can be a polysaccharide superabsorbent obtained by reacting a fibrous polysaccharide such as cellulose with an excess of a quaternary ammonium compound containing at least one group capable of reacting with polysaccharide hydroxyl groups and having a degree of substitution of 0.5 to 1.1.
- the quaternary ammonium compound may have the general formula:
- n is an integer from l to 16;
- X is halogen;
- Z is an anion such as halide or hydroxyl; and
- R, R 1 , R 2 and R 3 which may be the same or different, are each hydrogen, alkyl, hydroxyalkyl, alkenyl or aryl and R 2 may additionally represent a residue of formula
- the cationic superabsorbent may be a cross-linked cellulose based superabsorbent, in particular a cationic polysaccharide, for example a fibrous polysaccharide, having superabsorbent characteristics, the polysaccharide being substituted by quaternary ammonium groups and having a ds of at least 0.5 and the polysaccharide being cross-linked to a sufficient extent that it remains insoluble in water.
- a cationic polysaccharide for example a fibrous polysaccharide, having superabsorbent characteristics, the polysaccharide being substituted by quaternary ammonium groups and having a ds of at least 0.5 and the polysaccharide being cross-linked to a sufficient extent that it remains insoluble in water.
- superabsorbents of this type are described in more detail in our co-pending patent application No (internal reference DR44) .
- the cationic superabsorbent may be a water-swellable, water-insoluble polymer comprising units derived from a diallylic quaternary ammonium salt monomer, cross-linked by a suitable polyfunctional vinyl compound, characterised in that the polymer has been produced by cationic polymerisation in an aqueous phase using a free radical catalyst.
- a suitable polyfunctional vinyl compound characterised in that the polymer has been produced by cationic polymerisation in an aqueous phase using a free radical catalyst.
- the functional groups on anionic superabsorbent are such that the superabsorbent is a weak acid and those on the cationic superabsorbent are such that the superabsorbent is a strong base.
- the ratio of anionic to cationic superabsorbent is in the range 3:1 to 1:5 based on monomer units, more preferably 2:1 to 1:2, each monomer unit having one functional group therein.
- the anionic and cationic superabsorbents are used such that they have equal exchange power so that pH extremes in the bodily fluids absorbed are not reached and the optimum desalting effect is achieved.
- Cationic and anionic exchange power of the superabsorbent may be experimentally determined by, for example, titration, or in the case of synthetic polymers by a therotical calculation.
- the absorbent material according to the ' invention is particularly suitable for use in applications where it is desired to absorb electrolyte containing aqueous liquids.
- liquids include in particular menses and urine and the absorbent material can be used as the filling in catamenials and diapers generally in admixture with a fibrous absorbent such as cellulose fluff.
- the absorbent according to the invention can be present as granules or fibres.
- the absorbent materials according to the invention show particularly good absorption of electrolyte containing aqueous liquids as is demonstrated below in the following examples by tests carried out using saline solution (1% NaCl) and synthetic urine.
- saline solution 1% NaCl
- synthetic urine Pre p aration - Cationic Superabsorbent based on
- CATIONIC POLYMER IN ACID FORM 219 grams of a 60% aqueous solution of dimethyldiallyammonium chloride (DMAC) available from Fluka were weighed into a 500ml flask. 0.4597 g of bisacrylamide (crosslinker agent) were weighed separately into a 5 ml test tube and was dissolved using 2 ml distilled water. 0.12 g of ammonium persulfate (radical initiator) were dissolved separately in a 5 ml test tube in 2 ml distilled water. The air was removed from the monomer solution by means of a vacuum pump.
- DMAC dimethyldiallyammonium chloride
- the crosslinker solution and the radical initiator solution were added to the monomer solution, the temperature was adjusted to 60 ⁇ C by placing the flask in a thermostatic bath for four hours.
- the solid product formed was cut using a spatula and transferred in a 5 litre beaker containing 4 litres of distilled water, after two hours the swelled gel which had formed was filtered by a nonwoven tissue fabric filter. The gel was dried in a ventilated oven at 60°C for 12 hours. lOOg of a dried polymer called Fai 9 Cl " were collected.
- the gel was filtered in a nonwoven fabric tissue filter, the step of acidification and filtering of the gel containing solution was repeated until the disappearance of sodium ions from the washing waters (sodium ion content of the solution can be measured by potentiometric method using a sodium sensitive electrode) .
- the gel is transferred in a tea-bag type envelope and is suspended for 10 min to remove unabsorbed water after which the envelope is weighed. Absorbency is measured as follows:
- Wwet weight of the envelope containing the wet AGMs in g
- Wdry weight of the envelope containing the dry AGMs in g
- Gl weight of the dry anionic AGM in g
- G2 weight of the dry cationic AGM in g
- Retention Absorbency after centrifugation
- samples A to D were put into a saline solution (1%) or solution of synthetic urine and into deionized water.
- Sample E was tested only in saline/synthetic urine.
- 1% NaCl represents a stringent test of the superabsorben .
- Studies in the literature show that the salt content of urine varies depending on a number of factors but 1% by weight represents the maximum likely to the encountered in practice.
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002207081A CA2207081C (en) | 1994-12-06 | 1995-11-21 | Absorbent material |
| BR9509864A BR9509864A (en) | 1994-12-06 | 1995-11-21 | Superabsorbent material and use |
| EP95940778A EP0796144A4 (en) | 1994-12-06 | 1995-11-21 | Absorbent material |
| AU42417/96A AU4241796A (en) | 1994-12-06 | 1995-11-21 | Absorbent material |
| JP8517612A JPH10509915A (en) | 1994-12-06 | 1995-11-21 | Absorbable material |
| CZ971699A CZ169997A3 (en) | 1994-12-06 | 1995-11-21 | Superabsorptive material and the use thereof |
| NO972549A NO972549L (en) | 1994-12-06 | 1997-06-04 | Absorbent material |
| FI972385A FI972385A0 (en) | 1994-12-06 | 1997-06-05 | absorbent material |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IT94TO000991A IT1267184B1 (en) | 1994-12-06 | 1994-12-06 | ABSORBENT MATERIAL, FOR EXAMPLE OF THE SUPER ABSORBENT TYPE, AND RELATIVE USE. |
| ITTO94A000991 | 1994-12-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1996017681A1 true WO1996017681A1 (en) | 1996-06-13 |
Family
ID=11412952
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1995/015139 WO1996017681A1 (en) | 1994-12-06 | 1995-11-21 | Absorbent material |
Country Status (12)
| Country | Link |
|---|---|
| EP (1) | EP0796144A4 (en) |
| JP (1) | JPH10509915A (en) |
| CN (1) | CN1173146A (en) |
| AU (1) | AU4241796A (en) |
| BR (1) | BR9509864A (en) |
| CA (1) | CA2207081C (en) |
| CZ (1) | CZ169997A3 (en) |
| FI (1) | FI972385A0 (en) |
| HU (1) | HUT77680A (en) |
| IT (1) | IT1267184B1 (en) |
| NO (1) | NO972549L (en) |
| WO (1) | WO1996017681A1 (en) |
Cited By (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998024832A1 (en) * | 1996-12-02 | 1998-06-11 | Kimberly-Clark Worldwide, Inc. | Absorbent composition |
| WO1998037149A1 (en) * | 1997-02-19 | 1998-08-27 | The Procter & Gamble Company | Mixed-bed ion-exchange hydrogel-forming polymer compositions and absorbent members comprising relatively high concentrations of these compositions |
| WO1999025393A2 (en) * | 1997-11-19 | 1999-05-27 | Amcol International Corporation | Multicomponent superabsorbent gel particles |
| WO1999026610A1 (en) * | 1997-11-26 | 1999-06-03 | The Procter & Gamble Company | Skin care composition |
| WO1999034843A1 (en) * | 1998-01-07 | 1999-07-15 | The Procter & Gamble Company | Absorbent polymer compositions having high sorption capacities under an applied pressure |
| WO1999034841A1 (en) * | 1998-01-07 | 1999-07-15 | The Procter & Gamble Company | Absorbent polymer compositions with high sorption capacity and high fluid permeability under an applied pressure |
| WO1999034842A1 (en) * | 1998-01-07 | 1999-07-15 | The Procter & Gamble Company | Absorbent polymer compositions having high sorption capacities under an applied pressure and improved integrity in the swollen state |
| WO1999047093A1 (en) * | 1998-03-16 | 1999-09-23 | Sca Hygiene Products Ab | Absorbent structure and product based on raw materials having a high degree of renewability |
| US5962578A (en) * | 1997-11-19 | 1999-10-05 | Amcol International Corporation | Poly(dialkylaminoalkyl (meth)acrylamide)-based superabsorbent gels |
| WO2000009612A1 (en) * | 1998-08-13 | 2000-02-24 | Nippon Shokubai Co., Ltd. | Cross-linked polymer composition swelling in water and process for producing the same |
| US6072101A (en) * | 1997-11-19 | 2000-06-06 | Amcol International Corporation | Multicomponent superabsorbent gel particles |
| US6087448A (en) * | 1997-11-19 | 2000-07-11 | Amcol International Corporation | Solid superabsorbent material containing a poly(vinylguanidine) and an acidic water-absorbing resin |
| WO2000055258A1 (en) * | 1999-03-12 | 2000-09-21 | The Procter & Gamble Company | Absorbent polymer compositions having high sorption capacities under an applied pressure |
| EP1081178A2 (en) * | 1999-09-01 | 2001-03-07 | Nippon Shokubai Co., Ltd. | Water-swellable crosslinked polymer, its composition, and their production processes and uses |
| US6232520B1 (en) | 1997-02-19 | 2001-05-15 | The Procter & Gamble Company | Absorbent polymer compositions having high sorption capacities under an applied pressure |
| US6342298B1 (en) | 1997-11-19 | 2002-01-29 | Basf Aktiengesellschaft | Multicomponent superabsorbent fibers |
| US6372953B1 (en) | 1995-01-10 | 2002-04-16 | The Procter & Gamble Company | Absorbent members comprising a high surface area material for absorbing body liquids |
| AU746439B2 (en) * | 1997-12-12 | 2002-05-02 | Kimberly-Clark Worldwide, Inc. | Structure having balanced pH profile |
| US6426445B1 (en) | 1995-01-10 | 2002-07-30 | The Procter & Gamble Company | Absorbent members comprising an agglomerate of hydrogel-forming absorbent polymer and particulate hydrophilic foam |
| US6534554B1 (en) | 1999-10-27 | 2003-03-18 | Basf Aktiengesellschaft | Multicomponent ion exchange resins |
| US6639120B1 (en) * | 1997-12-12 | 2003-10-28 | Kimberly-Clark Worldwide, Inc. | Structure having balanced pH profile |
| WO2003097116A1 (en) * | 2002-05-20 | 2003-11-27 | First Water Limited | Ionic hydrogels with controlled aqueous fluid absorption |
| AU777449B2 (en) * | 1999-12-28 | 2004-10-14 | Kimberly-Clark Worldwide, Inc. | Superabsorbent polymers |
| US6881792B2 (en) | 1999-12-27 | 2005-04-19 | Nippon Shokubai Co., Ltd. | Production processes for basic water-absorbent resin and water-absorbing agent, and use thereof |
| US6891080B2 (en) * | 1999-04-12 | 2005-05-10 | Kao Corporation | Disposable diaper and absorbent member used therein |
| US6958429B2 (en) | 1998-06-08 | 2005-10-25 | Stockhausen Gmbh & Co. Kg | Water-absorbing polymers with supramolecular hollow molecules, method for producing them and use of the same |
| EP1624002A1 (en) * | 2004-08-07 | 2006-02-08 | The Procter & Gamble Company | Superabsorbent polymer particles comprising functionalizers and method of making them |
| US6997327B2 (en) | 2003-09-24 | 2006-02-14 | Velcon Filters, Inc. | Salt-tolerant, water absorbing filter |
| US7012105B2 (en) | 1997-12-12 | 2006-03-14 | Kimberly-Clark Worldwide, Inc. | Structure having balanced pH profile |
| EP1637105A1 (en) * | 2003-06-13 | 2006-03-22 | Sumitomo Seika Chemicals Co., Ltd. | Absorbing material and absorptive article using the same |
| EP2123242A1 (en) | 2008-05-19 | 2009-11-25 | The Procter and Gamble Company | Absorbent core |
| US7696401B2 (en) | 2003-07-31 | 2010-04-13 | Evonik Stockhausen, Inc. | Absorbent materials and absorbent articles incorporating such absorbent materials |
| US7855315B2 (en) * | 1997-11-19 | 2010-12-21 | Basf Aktiengesellschaft | Continuous manufacturing of superabsorbent/ion exchange sheet material |
| EP2394669A1 (en) * | 2010-06-11 | 2011-12-14 | The Procter & Gamble Company | Absorbent product comprising a cationic polysaccharide in a hydrophilic carrier matrix |
| US11173078B2 (en) | 2015-11-04 | 2021-11-16 | The Procter & Gamble Company | Absorbent structure |
| US11376168B2 (en) | 2015-11-04 | 2022-07-05 | The Procter & Gamble Company | Absorbent article with absorbent structure having anisotropic rigidity |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4704559B2 (en) * | 1999-12-27 | 2011-06-15 | 株式会社日本触媒 | Manufacturing method of basic water-absorbing resin, manufacturing method of water-absorbing agent, and use thereof |
| EP1149593A1 (en) * | 2000-04-25 | 2001-10-31 | The Procter & Gamble Company | Articles comprising cationic polysaccharides and acidic pH buffering means |
| AU2003229743A1 (en) * | 2002-05-01 | 2003-11-17 | Basf Aktiengesellschaft | Plasticized superabsorbent polymer sheets and use thereof in hygienic articles |
| EP2221068A2 (en) * | 2003-06-30 | 2010-08-25 | The Procter & Gamble Company | Absorbent structures comprising coated super-absorbent polymer articles |
| JP5813515B2 (en) * | 2009-02-18 | 2015-11-17 | クイック−メッド テクノロジーズ、インク. | Superabsorbent material made of peroxide |
| CN104289190B (en) * | 2014-09-10 | 2016-06-29 | 济南大学 | A kind of preparation of dimethyl diallyl ammonium chloride modified fiber crops adsorbent |
| CN104475065B (en) * | 2014-12-16 | 2017-01-18 | 湖南科技大学 | Efficient heavy-metal-ion absorbent material and preparation method thereof |
| CN105646788B (en) * | 2016-01-07 | 2018-02-13 | 太原理工大学 | A kind of preparation method of peanut shell degradable nano dust suppressant |
| JP7151422B2 (en) * | 2018-11-28 | 2022-10-12 | セイコーエプソン株式会社 | Liquid absorber and method of controlling the liquid absorber |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4818598A (en) * | 1985-06-28 | 1989-04-04 | The Procter & Gamble Company | Absorbent structures |
| US5286827A (en) * | 1990-10-12 | 1994-02-15 | Phillips Petroleum Company | Superabsorbent crosslinked copolymers formed from an ampholytic ion pair (2-methacryloyloxyethyldimethylammonium cation (MEDMA)/sulfonate anion) copolymerized with other comonomers |
| US5354806A (en) * | 1990-10-01 | 1994-10-11 | Phillips Petroleum Company | Superabsorbent crosslinked ampholytic ion pair copolymers |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3968037A (en) * | 1972-09-01 | 1976-07-06 | Calgon Corporation | Emulsion polymerization of cationic monomers |
| JPS60248720A (en) * | 1984-03-26 | 1985-12-09 | アトランテイツク・リツチフイ−ルド・カンパニ− | Water-insoluble water-swellable high molecular compound and manufacture |
| IT1249309B (en) * | 1991-05-03 | 1995-02-22 | Faricerca Spa | CATIONIC TYPE POLYSACCHARIDES |
| US5330656A (en) * | 1993-03-05 | 1994-07-19 | Calgon Corporation | Polysalt compositions and the use thereof for treating an oil and water system |
-
1994
- 1994-12-06 IT IT94TO000991A patent/IT1267184B1/en active IP Right Grant
-
1995
- 1995-11-21 HU HU9800670A patent/HUT77680A/en unknown
- 1995-11-21 CZ CZ971699A patent/CZ169997A3/en unknown
- 1995-11-21 JP JP8517612A patent/JPH10509915A/en active Pending
- 1995-11-21 AU AU42417/96A patent/AU4241796A/en not_active Abandoned
- 1995-11-21 CA CA002207081A patent/CA2207081C/en not_active Expired - Fee Related
- 1995-11-21 CN CN95197333A patent/CN1173146A/en active Pending
- 1995-11-21 WO PCT/US1995/015139 patent/WO1996017681A1/en not_active Application Discontinuation
- 1995-11-21 BR BR9509864A patent/BR9509864A/en not_active Application Discontinuation
- 1995-11-21 EP EP95940778A patent/EP0796144A4/en not_active Withdrawn
-
1997
- 1997-06-04 NO NO972549A patent/NO972549L/en not_active Application Discontinuation
- 1997-06-05 FI FI972385A patent/FI972385A0/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4818598A (en) * | 1985-06-28 | 1989-04-04 | The Procter & Gamble Company | Absorbent structures |
| US5354806A (en) * | 1990-10-01 | 1994-10-11 | Phillips Petroleum Company | Superabsorbent crosslinked ampholytic ion pair copolymers |
| US5286827A (en) * | 1990-10-12 | 1994-02-15 | Phillips Petroleum Company | Superabsorbent crosslinked copolymers formed from an ampholytic ion pair (2-methacryloyloxyethyldimethylammonium cation (MEDMA)/sulfonate anion) copolymerized with other comonomers |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP0796144A4 * |
Cited By (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6426445B1 (en) | 1995-01-10 | 2002-07-30 | The Procter & Gamble Company | Absorbent members comprising an agglomerate of hydrogel-forming absorbent polymer and particulate hydrophilic foam |
| US6372953B1 (en) | 1995-01-10 | 2002-04-16 | The Procter & Gamble Company | Absorbent members comprising a high surface area material for absorbing body liquids |
| US6951895B1 (en) | 1996-12-02 | 2005-10-04 | Kimberly-Clark Worldwide, Inc. | Absorbent composition |
| WO1998024832A1 (en) * | 1996-12-02 | 1998-06-11 | Kimberly-Clark Worldwide, Inc. | Absorbent composition |
| US6232520B1 (en) | 1997-02-19 | 2001-05-15 | The Procter & Gamble Company | Absorbent polymer compositions having high sorption capacities under an applied pressure |
| WO1998037149A1 (en) * | 1997-02-19 | 1998-08-27 | The Procter & Gamble Company | Mixed-bed ion-exchange hydrogel-forming polymer compositions and absorbent members comprising relatively high concentrations of these compositions |
| US6258996B1 (en) * | 1997-02-19 | 2001-07-10 | The Procter & Gamble Company | Mixed-bed ion-exchange hydrogel-forming polymer compositions and absorbent members comprising relatively high concentrations of these compositions |
| US6590137B2 (en) | 1997-11-19 | 2003-07-08 | Bask Aktiengesellschaft | Multicomponent superabsorbent gel particles |
| US7855315B2 (en) * | 1997-11-19 | 2010-12-21 | Basf Aktiengesellschaft | Continuous manufacturing of superabsorbent/ion exchange sheet material |
| WO1999025393A3 (en) * | 1997-11-19 | 1999-09-02 | Amcol International Corp | Multicomponent superabsorbent gel particles |
| US5962578A (en) * | 1997-11-19 | 1999-10-05 | Amcol International Corporation | Poly(dialkylaminoalkyl (meth)acrylamide)-based superabsorbent gels |
| WO1999025393A2 (en) * | 1997-11-19 | 1999-05-27 | Amcol International Corporation | Multicomponent superabsorbent gel particles |
| US6072101A (en) * | 1997-11-19 | 2000-06-06 | Amcol International Corporation | Multicomponent superabsorbent gel particles |
| US6087448A (en) * | 1997-11-19 | 2000-07-11 | Amcol International Corporation | Solid superabsorbent material containing a poly(vinylguanidine) and an acidic water-absorbing resin |
| US6603056B2 (en) | 1997-11-19 | 2003-08-05 | Basf Aktiengesellschaft | Multicomponent superabsorbent gel particles |
| EP2011462A1 (en) | 1997-11-19 | 2009-01-07 | Basf Se | Multicomponent Superabsorbent Gel Particles |
| US6159591A (en) * | 1997-11-19 | 2000-12-12 | Amcol International Corporation | Multicomponent superabsorbent gel particles |
| US6596922B2 (en) | 1997-11-19 | 2003-07-22 | Basf Aktiengesellschaft | Multicomponent superabsorbent gel particles |
| US6222091B1 (en) | 1997-11-19 | 2001-04-24 | Basf Aktiengesellschaft | Multicomponent superabsorbent gel particles |
| US6596921B2 (en) | 1997-11-19 | 2003-07-22 | Basf Aktiengesellschaft | Multicomponent superabsorbent gel particles |
| US6235965B1 (en) | 1997-11-19 | 2001-05-22 | Basf Aktiengesellschaft | Multicomponent superabsorbent gel particles |
| US6555502B1 (en) | 1997-11-19 | 2003-04-29 | Basf Aktiengesellschaft | Multicomponent superabsorbent gel particles |
| US6509512B1 (en) | 1997-11-19 | 2003-01-21 | Basf Aktiengesellschaft | Multicomponent superabsorbent gel particles |
| US6392116B1 (en) | 1997-11-19 | 2002-05-21 | Basf Aktiengesellschaft | Diapers having improved acquisition rates |
| US6376072B2 (en) | 1997-11-19 | 2002-04-23 | Basf Aktiengesellschaft | Multicomponent superabsorbent fibers |
| US6342298B1 (en) | 1997-11-19 | 2002-01-29 | Basf Aktiengesellschaft | Multicomponent superabsorbent fibers |
| WO1999026610A1 (en) * | 1997-11-26 | 1999-06-03 | The Procter & Gamble Company | Skin care composition |
| EP0922452A1 (en) * | 1997-11-26 | 1999-06-16 | The Procter & Gamble Company | Skin care composition |
| AU746439B2 (en) * | 1997-12-12 | 2002-05-02 | Kimberly-Clark Worldwide, Inc. | Structure having balanced pH profile |
| US7012105B2 (en) | 1997-12-12 | 2006-03-14 | Kimberly-Clark Worldwide, Inc. | Structure having balanced pH profile |
| US6639120B1 (en) * | 1997-12-12 | 2003-10-28 | Kimberly-Clark Worldwide, Inc. | Structure having balanced pH profile |
| KR100546551B1 (en) * | 1997-12-12 | 2006-01-26 | 킴벌리-클라크 월드와이드, 인크. | Structure Having Balanced pH Profile |
| US7317135B2 (en) | 1997-12-12 | 2008-01-08 | Kimberly-Clark Worldwide, Inc. | Structure having balanced pH profile |
| WO1999034841A1 (en) * | 1998-01-07 | 1999-07-15 | The Procter & Gamble Company | Absorbent polymer compositions with high sorption capacity and high fluid permeability under an applied pressure |
| WO1999034843A1 (en) * | 1998-01-07 | 1999-07-15 | The Procter & Gamble Company | Absorbent polymer compositions having high sorption capacities under an applied pressure |
| WO1999034842A1 (en) * | 1998-01-07 | 1999-07-15 | The Procter & Gamble Company | Absorbent polymer compositions having high sorption capacities under an applied pressure and improved integrity in the swollen state |
| US6121509A (en) * | 1998-01-07 | 2000-09-19 | The Procter & Gamble Company | Absorbent polymer compositions having high sorption capacities under an applied pressure and improved integrity when wet |
| WO1999047093A1 (en) * | 1998-03-16 | 1999-09-23 | Sca Hygiene Products Ab | Absorbent structure and product based on raw materials having a high degree of renewability |
| US6958429B2 (en) | 1998-06-08 | 2005-10-25 | Stockhausen Gmbh & Co. Kg | Water-absorbing polymers with supramolecular hollow molecules, method for producing them and use of the same |
| US6333109B1 (en) | 1998-08-13 | 2001-12-25 | Nippon Shokubai Co., Ltd. | Water-swellable crosslinked polymer composition and production |
| WO2000009612A1 (en) * | 1998-08-13 | 2000-02-24 | Nippon Shokubai Co., Ltd. | Cross-linked polymer composition swelling in water and process for producing the same |
| WO2000055258A1 (en) * | 1999-03-12 | 2000-09-21 | The Procter & Gamble Company | Absorbent polymer compositions having high sorption capacities under an applied pressure |
| US6891080B2 (en) * | 1999-04-12 | 2005-05-10 | Kao Corporation | Disposable diaper and absorbent member used therein |
| EP1081178A2 (en) * | 1999-09-01 | 2001-03-07 | Nippon Shokubai Co., Ltd. | Water-swellable crosslinked polymer, its composition, and their production processes and uses |
| EP1081178A3 (en) * | 1999-09-01 | 2001-07-04 | Nippon Shokubai Co., Ltd. | Water-swellable crosslinked polymer, its composition, and their production processes and uses |
| US6342652B1 (en) | 1999-09-01 | 2002-01-29 | Nippon Shokubai Co., Ltd. | Water-swellable crosslinked polymer, its composition, and their production processes and uses |
| US6534554B1 (en) | 1999-10-27 | 2003-03-18 | Basf Aktiengesellschaft | Multicomponent ion exchange resins |
| US6881792B2 (en) | 1999-12-27 | 2005-04-19 | Nippon Shokubai Co., Ltd. | Production processes for basic water-absorbent resin and water-absorbing agent, and use thereof |
| AU777449C (en) * | 1999-12-28 | 2007-03-15 | Kimberly-Clark Worldwide, Inc. | Superabsorbent polymers |
| KR100732073B1 (en) * | 1999-12-28 | 2007-06-27 | 킴벌리-클라크 월드와이드, 인크. | Superabsorbent Polymers |
| US7820873B2 (en) | 1999-12-28 | 2010-10-26 | Kimberly-Clark Worldwide, Inc. | Absorbent structure comprising synergistic components for superabsorbent polymer |
| EP1242128B1 (en) * | 1999-12-28 | 2006-08-09 | Kimberly-Clark Worldwide, Inc. | Superabsorbent polymers |
| AU777449B2 (en) * | 1999-12-28 | 2004-10-14 | Kimberly-Clark Worldwide, Inc. | Superabsorbent polymers |
| WO2003097116A1 (en) * | 2002-05-20 | 2003-11-27 | First Water Limited | Ionic hydrogels with controlled aqueous fluid absorption |
| EP1637105A4 (en) * | 2003-06-13 | 2009-03-25 | Sumitomo Seika Chemicals | Absorbing material and absorptive article using the same |
| EP1637105A1 (en) * | 2003-06-13 | 2006-03-22 | Sumitomo Seika Chemicals Co., Ltd. | Absorbing material and absorptive article using the same |
| US8269060B2 (en) | 2003-07-31 | 2012-09-18 | Evonik Stockhausen, Llc | Absorbent materials and absorbent articles incorporating such absorbent materials |
| US7696401B2 (en) | 2003-07-31 | 2010-04-13 | Evonik Stockhausen, Inc. | Absorbent materials and absorbent articles incorporating such absorbent materials |
| US6997327B2 (en) | 2003-09-24 | 2006-02-14 | Velcon Filters, Inc. | Salt-tolerant, water absorbing filter |
| WO2006017716A2 (en) * | 2004-08-07 | 2006-02-16 | The Procter & Gamble Company | Superabsorbent polymer particles comprising functionalizers and method of making them |
| EP1624002A1 (en) * | 2004-08-07 | 2006-02-08 | The Procter & Gamble Company | Superabsorbent polymer particles comprising functionalizers and method of making them |
| WO2006017716A3 (en) * | 2004-08-07 | 2006-05-26 | Procter & Gamble | Superabsorbent polymer particles comprising functionalizers and method of making them |
| EP2123242A1 (en) | 2008-05-19 | 2009-11-25 | The Procter and Gamble Company | Absorbent core |
| WO2009143118A1 (en) * | 2008-05-19 | 2009-11-26 | The Procter & Gamble Company | Absorbent core |
| EP2394669A1 (en) * | 2010-06-11 | 2011-12-14 | The Procter & Gamble Company | Absorbent product comprising a cationic polysaccharide in a hydrophilic carrier matrix |
| WO2011156197A1 (en) * | 2010-06-11 | 2011-12-15 | The Procter & Gamble Company | Absorbent product comprising a mixture of a cationic polysaccharide and a hydrophilic carrier matrix |
| US11173078B2 (en) | 2015-11-04 | 2021-11-16 | The Procter & Gamble Company | Absorbent structure |
| US11376168B2 (en) | 2015-11-04 | 2022-07-05 | The Procter & Gamble Company | Absorbent article with absorbent structure having anisotropic rigidity |
Also Published As
| Publication number | Publication date |
|---|---|
| ITTO940991A1 (en) | 1996-06-06 |
| EP0796144A4 (en) | 2000-03-29 |
| FI972385A (en) | 1997-06-05 |
| MX9704177A (en) | 1998-06-28 |
| AU4241796A (en) | 1996-06-26 |
| BR9509864A (en) | 1997-11-25 |
| NO972549D0 (en) | 1997-06-04 |
| CA2207081A1 (en) | 1996-06-13 |
| IT1267184B1 (en) | 1997-01-28 |
| EP0796144A1 (en) | 1997-09-24 |
| NO972549L (en) | 1997-08-06 |
| CN1173146A (en) | 1998-02-11 |
| JPH10509915A (en) | 1998-09-29 |
| FI972385A0 (en) | 1997-06-05 |
| CA2207081C (en) | 2001-04-17 |
| HUT77680A (en) | 1998-07-28 |
| CZ169997A3 (en) | 1997-10-15 |
| ITTO940991A0 (en) | 1994-12-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2207081C (en) | Absorbent material | |
| US7291674B2 (en) | Superabsorbent polymer | |
| JPS6024807B2 (en) | Method for producing super absorbent hydrogel | |
| CA2204888C (en) | Absorbent material | |
| US6068924A (en) | Absorbent material | |
| EP0791020A1 (en) | Absorbent material | |
| MXPA97003445A (en) | Absorbe material | |
| US5804605A (en) | Absorbent material | |
| CN1093139C (en) | Cationic polymer | |
| AU1672592A (en) | Cationic polysaccharides | |
| JPS60135432A (en) | Modified water-absorptive resin | |
| CA2205026C (en) | Cationic polymer | |
| US6084045A (en) | Cationic polymer | |
| US20050080389A1 (en) | Absorbent articles having increased absorbency of complex fluids | |
| MXPA97004177A (en) | Absorbe material | |
| CA2204890A1 (en) | Cationic polymer | |
| JPH05123573A (en) | Preparation of highly water absorptive cellulose material | |
| CA2204891A1 (en) | Absorbent material |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 95197333.9 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AL AM AT AU BB BG BR BY CA CH CN CZ DE DK EE ES FI GB GE HU IS JP KE KG KP KR KZ LK LR LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK TJ TM TT UA UG US UZ VN |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): KE LS MW SD SZ UG AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 1995940778 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 1997 849607 Country of ref document: US Date of ref document: 19970602 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PV1997-1699 Country of ref document: CZ |
|
| ENP | Entry into the national phase |
Ref document number: 2207081 Country of ref document: CA Ref document number: 2207081 Country of ref document: CA Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/1997/004177 Country of ref document: MX Ref document number: 1019970703775 Country of ref document: KR Ref document number: 972385 Country of ref document: FI |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1199700517 Country of ref document: VN |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: 1995940778 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: PV1997-1699 Country of ref document: CZ |
|
| WWP | Wipo information: published in national office |
Ref document number: 1019970703775 Country of ref document: KR |
|
| WWR | Wipo information: refused in national office |
Ref document number: PV1997-1699 Country of ref document: CZ |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1995940778 Country of ref document: EP |
|
| WWR | Wipo information: refused in national office |
Ref document number: 1019970703775 Country of ref document: KR |