WHAT IS CLAIMED IS:
1. A biomaterial modified with at least one non- proteinaceous catalyst for the dismutation of superoxide or a precursor ligand of a non-proteinaceous catalyst for the dismutation of superoxide.
2. The biomaterial of claim 1 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
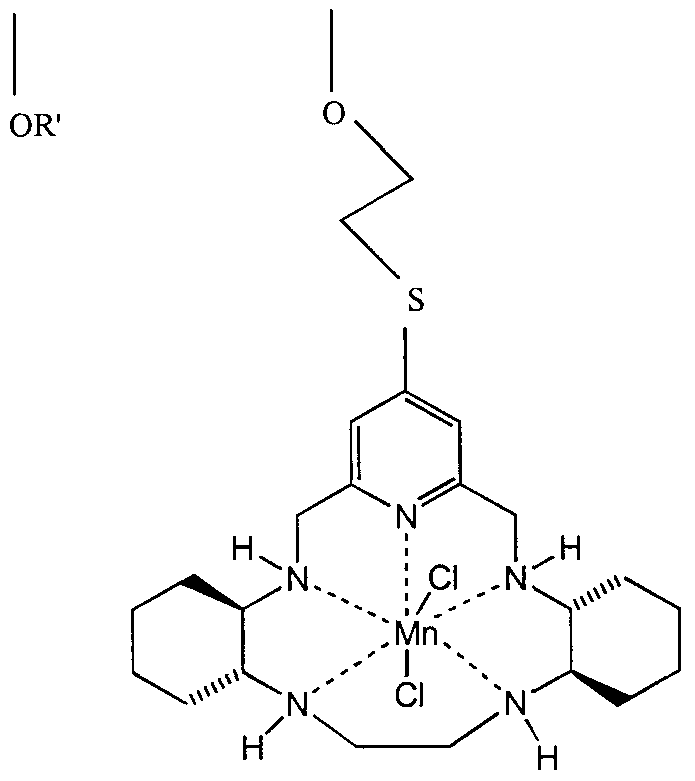
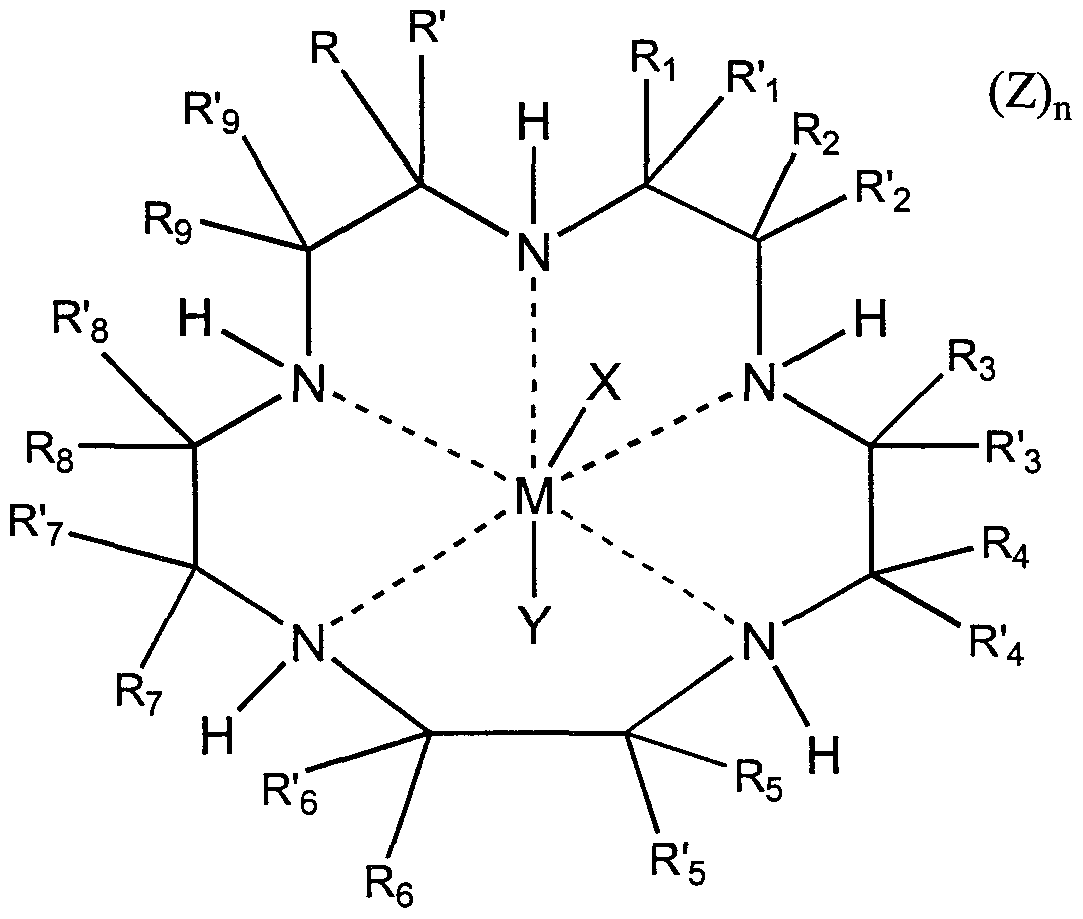
3. The biomaterial of claim 1 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds , which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R
lf R'
lf R
2, R'
2, R
3,
R'37 R4 / R'4/ R5 R'5 R-6 / R'β' R7 / R'7/ Re / R's/ R-9 / &nd R'9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; R1 or R\ and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R' and Rλ or R'x, R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rx and R'lf R2 and R' 2 R3 and R' 3 / R. and R'4/ R5 and R'5, R6 and R'6, R7 and R' 7 / R8 and R' 8 ' and R9 and R'9 , together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', Rl t R'1# R2, R'2, R3, R'3, R4, R'4, Rs, R's, R6, R'6, R7, R'7, R8, R'8, R9, and R'9 together with a different one of R, R', Rl r R'1# R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'g, R7, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate,
75 thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
4. The biomaterial of claim 1 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
5. The biomaterial of claim 1 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
6. The biomaterial of claim 2, 3, 4, or 5 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
7. The biomaterial of claim 2, 3, 4, or 5 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
8. The biomaterial of claim 2, 3, 4, or 5 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
9. The biomaterial of claim 1 wherein the unmodified biomaterial is selected from the group consisting of: metals, ceramics, polymers, biopolymers, and composites thereof.
10. The biomaterial of claim 1 wherein the unmodified biomaterial is a metal selected from the group consisting of: stainless steel, tantalum, titanium, nitinol, gold, platinum, inconel, iridium, silver, tungsten, nickel, chromium, vanadium, and alloys comprising any of the foregoing metals and alloys.
11. The biomaterial of claim 10 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
12. The biomaterial of claim 10 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition meta , preferably manganese or iron; wherein R, R' , Rlr R'x, R2, R'2/ R3 '3 R4 R'4 R5 / '5/ Re ' R'β R7 ' R'7 Re 8/ 9 ' 3π R t independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rx or R'x and R2 or R'2, R3 or
R', and R, or R Rc or R' and Rfi or R' 6 / R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R ' and Rx or R'17 R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, Rs or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R1, R1 and R' __ , R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'6, R7 and R'7, R8 and R'8, and Rg and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', Rl; R'lf R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'6, R7, R'7, R8, R'8, R9, and R' 9 together with a different one of R, R', R1# R'1# R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'6, R7, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins .
13. The biomaterial of claim 10 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
14. The biomaterial of claim 10 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
15. The biomaterial of claim 11, 12, 13, 14 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
16. The biomaterial of claim 11, 12, 13, 14 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
17. The biomaterial of claim 11, 12, 13, 14 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
18. The biomaterial of claim 1 wherein the unmodified biomaterial is a ceramic selected from the group consisting of: hydroxyapatite, tricalcium phosphate, and aluminum-calcium-phosphorus oxide
19. The biomaterial of claim 18 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron(III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
20. The biomaterial of claim 18 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R
1# R'
17 R
2, R'
2, R
3,
R' 3 / R4 / R'4/ R5 ' R'5 Re R' 6/ R7 / R'7/ Re ' ' 8 ' R9/ and R' 9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rx or R' 1 and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R's, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R* and Rx or R'17 R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rτ and R'17 R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R's, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R1# R'x, R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R's, R7, R'7, R8, R'8/ Rg/ and R'9 together with a different one of R, R', Rl r R , R2 , R'2, R3, R'3, R4, R'4, R5, R'5, R6, R's, R7, R'7, R8 , R'8/ R 9/ and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate,
75 thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
21. The biomaterial of claim 18 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
22. The biomaterial of claim 18 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
23. The biomaterial of claim 19, 20, 21, or 22 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
24. The biomaterial of claim 19, 20, 21, or 22 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
25. The biomaterial of claim 19, 20, 21, or 22 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
26. The biomaterial of claim 1 wherein the unmodified biomaterial is a polymer selected from the group consisting of: polyurethane, polyureaurethane, polyalkylene glycols, polyethylene teraphthalate, ultra high molecular weight polyethylene, polypropylene, polyesters, polyamides, polycarbonates, polyorthoesters, polyesteramides, polysiloxane, polyolefins, polytetrafluoroethylene, polysulfones, polyanhydrides, polyalkylene oxides, polyvinyl halides, pol vinyledene halides, acrylic, methacrylic, polyacrylonitrile, polyvinyl, polyphosphazene, polyethylene-co-acrylic acid, silicone, block copolymer of any of the foregoing polymers, random copolymers of any of the foregoing polymers, graft copolymers of any of the foregoing polymers, crosslinked polymers of any of the foregoing polymers, hydrogels, and mixtures of any of the foregoing polymers .
27. The biomaterial of claim 26 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II] porphyrin complexes, and iron (III) porphyrin complexes.
28. The biomaterial of claim 26 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , Rlf R'χ, R2, R'2, R3,
R'3/ R4 / '4/ R5 / R'5/ Re / ' 6 / R7 / '7 Re R ' 8 R9 3-fl. ' 9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rt or RX and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R* and Rx or R'l; R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R's and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', R and R'17 R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R's, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R17 R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R's, R7, R'7, R8, R'8, R9, and R'9 together with a different one of R, R1, R1# R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R's, R7, R'7, R8/ R'8/ R9/ and R'g which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2) W-L- (CH2) Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl, phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, 95 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
100 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
105 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
29. The biomaterial of claim 26 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
30. The biomaterial of claim 26 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
31. The biomaterial of claim 27, 28, 29, or 30 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent
32. The biomaterial of claim 27, 28, 29, or 30 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
33. The biomaterial of claim 27, 28, 29, or 30 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
34. The biomaterial of claim 1 wherein the unmodified biomaterial is a polyethylene glycol .
35. The biomaterial of claim 34 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
36. The biomaterial of claim 34 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R
17 R'
17 R
2, R'
2, R
3,
R' 37 R4 / R 4 / R5 / '5/ Re R e/ R7 R'7 Re / R 8 / R9 / an R 9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rλ or R' 1 and R2 or R'2, R3 or R'3 and R4 or -R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and Ra or R'8, and R9 or R'9 and R or R ' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R1 and Rx or R'1# R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R's, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rx and R'17 R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R17 R'1# R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'g, R7/ R'7/ R8/ R'β/ R 9/ and R,9 together with a different one of R, R', R17 R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R's, R7, R'7, R8 , R'B/ 9 and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate,
75 thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl L irate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
37. The biomaterial of claim 34 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
38. The biomaterial of claim 34 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
39. The biomaterial of claim 35, 36, 37, or 38 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent,
40. The biomaterial of claim 35, 36, 37, or 38 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
41. The biomaterial of claim 35, 36, 37, or 38 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
42. The biomaterial of claim 1 wherein the unmodified biomaterial is a biopolymer selected from the group consisting of: chitin, chitosan, cellulose, methyl cellulose, hyaluronic acid, keratin, fibroin, collagen, elastin, and saccharide polymers.
43. The biomaterial of claim 42 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
44. The biomaterial of claim 42 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R17 R'17 R2, R'2, R3,
R'3 R4 / R'4/ R5/ R'5/ Re/ R'ε 7 / R'7/ Re / R'β/ R9 / nd R' 9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; R2 or R'x and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R' and Rx or R'1# R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R1, R and R' x , R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R1, R17 R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'g, R7/ R'7/ R8/ R's/ R9/ and R,9 together with a different one of R, R' , R17 R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'g, R7, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl , alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate, bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate, 95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
45. The biomaterial of claim 42 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
46. The biomaterial of claim 42 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
47. The biomaterial of claim 43, 44, 45, or 46 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
48. The biomaterial of claim 43, 44, 45, or 46 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
49. The biomaterial of claim 43, 44, 45, or 46 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
50. The biomaterial of claim 1 wherein the unmodified biomaterial is a composite material comprising a relatively inelastic phase selected from the group consisting of: carbon, hydroxy apatite, tricalcium
5 phosphate, silicates, ceramics, and metals, and a relatively elastic phase selected from the group consisting of: polymers and biopolymers.
51. The biomaterial of claim 50 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
52. The biomaterial of claim 50 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R17 R'17 R2, R'2, R3,
R'3/ R4/ '4/ R5/ '5/ Re/ R'ε 7 '7/ e / 'β/ R9 aπd R'9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rx or R and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'g, and R9 or R'9 and R or R* together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R ' and Rx or R'lf R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'g and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rx and R , R2 and R ' 2 , R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R ' 8 , and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R1 R'1# R2, R'2, R3, R'3, R4, R'4,
R5 / R'5/ Rζ, R'βi R / R'7/ Rs/ R's/ 9/ < and 9 together with a different one of R, R1, R17 R'17 R2, R' R3 / '3/ 4 / R R''44,/ RR55,/ RR''55,/ RR6e, RR''s6,/ RR77,/ RR''77,/ RR8s// RR''s8// R R99/, and R's which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
(CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2 2)' y wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins .
53. The biomaterial of claim 50 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
54. The biomaterial of claim 50 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
55. The biomaterial of claim 51, 52, 53, or 54 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent
56. The biomaterial of claim 51, 52, 53, or 54 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
57. The biomaterial of claim 51, 52, 53, or 54 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
58. The biomaterial of claim 1 comprising the non- proteinaceous catalyst for the dismutation of superoxide covalently bound to the surface of the biomaterial .
59. The biomaterial of claim 1 comprising a copolymer of the non-proteinaceous catalyst for the dismutation of superoxide and the biomaterial monomer.
60. The biomaterial of claim 1 comprising an admixture of the non-proteinaceous catalyst for the dismutation of superoxide and the biomaterial.
61. The biomaterial of claim 1 wherein, upon exposure to a biological fluid, dissociation of the non- proteinaceous catalyst for the dismutation of superoxide or the precursor ligand from the biomaterial is prevented by at least one covalent bond between the non- proteinaceous catalyst for the dismutation of superoxide or the precursor ligand and the biomaterial .
62. The biomaterial of claim 1 wherein, upon exposure to a biological fluid, dissociation of the non- proteinaceous catalyst for the dismutation of superoxide or the precursor ligand from the biomaterial is prevented by ionic interactions between the non-proteinaceous catalyst for the dismutation of superoxide or the precursor ligand and the biomaterial .
63. The biomaterial of claim 1 wherein, upon exposure to a biological fluid, dissociation of the non- proteinaceous catalyst for the dismutation of superoxide or the precursor ligand from the biomaterial is prevented by hydrophobic interactions between the non-proteinaceous catalyst for the dismutation of superoxide and the biomaterial .
64. A process for producing a biomaterial modified by surface covalent conjugation with at least one non- proteinaceous catalyst for the dismutation of superoxide or at least one precursor ligand of a non-proteinaceous catalyst for the dismutation of superoxide, the process comprising:
a. providing at least one reactive functional group on a surface of the biomaterial to be modified;
b. providing at least one complementary reactive functional group on the non-proteinaceous catalyst for the dismutation of superoxide or on the precursor ligand; and c. conjugating the non-proteinaceous catalyst for the dismutation of superoxide or the precursor ligand with the surface of the biomaterial through at least one covalent bond.
65. The process of claim 64 wherein the non- proteinaceous catalyst for the dismutation of superoxide is conjugated with the surface of the biomaterial by a photo-chemical reaction.
66. The process of claim 64 wherein the non- proteinaceous catalyst for the dismutation of superoxide or the precursor ligand is covalently bound directly to the surface of the biomaterial .
67. The process of claim 64 further comprising providing at least one linker capable of reacting with both the reactive functional group on a surface of the biomaterial to be modified and the complementary reactive functional group on the non-proteinaceous catalyst for the dismutation of superoxide or the precursor ligand, wherein during said conjugation at least one reactive functional group on the surface of the article and at least one complementary reactive functional group on the non-proteinaceous catalyst for the dismutation of superoxide or the precursor ligand form a covalent bond with the linker.
68. The process of claim 67 wherein the linker is selected from the group consisting of: polysaccharides, polyalkylene glycols, hexamethyl diimidi- isocyanate, silyl chloride, polypeptides, and polyaldehydes .
69. The process of claim 64 wherein the reactive functional group on the surface of the biomaterial is selected from the group consisting of: acid halide (XCO- wherein X= Cl , F, Br, I), amino (H2N-), isocyanate (OCN-), mercapto (HS-) , glycidyl (H2COCH-) , carboxyl (HOCO-) , hydroxy (HO-) , and chloromethyl (C1H2C-) .
70. The process of claim 64 wherein the complementary reactive functional group on the non- proteinaceous catalyst for the dismutation of superoxide or the precursor ligand is selected from the group consisting: of amino (-NH2) , carboxyl (-OCOH) , isocyanate (-NCO) , mercapto (-SH), hydroxy (-OH), silyl chloride (- SiCl2) , acid halide (-OCX wherein X= Cl , F, Br, I), halide (-X wherein X= Cl , F, Br, I), and glycidyl (-HCOCH2).
71. The process of claim 64 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of: manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
72. The process of claim 64 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R1# R'17 R2, R'2/ R3# R'3 R4 / 4 / R5 / R'5/ Rε R ' 6 R7 / R'7 e / R ' 8 R9 / aπd R ' 9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rx or R'x and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R ' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R ' and Rx or R'1# R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rx and R'17 R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R17 R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'g, R7, R'7, R8, R'8, R9, and R'9 together with a different one of R, R', R17 R'17 R2, R'2, R3, R'3, R4, R'4, R57 R'5, R6, R'g, R7, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate,
75 thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, asoorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
73. The process of claim 64 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
74. The process of claim 64 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
75. The process of claim 71, 72, 73, or 74 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
76. The process of claim 71, 72, 73, or 74 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
77. The process of claim 71, 72, 73, or 74 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
78. The process of claim 64 wherein the unmodified biomaterial is selected from the group consisting of: metals, ceramics, polymers, biopolymers, and composites thereof .
79. The process of claim 64 wherein the unmodified biomaterial is a metal selected from the group consisting of: stainless steel, tantalum, titanium, nitinol, gold, platinum, inconel, iridium, silver, tungsten, nickel, chromium, vanadium, and alloys comprising any of the foregoing metals and alloys.
80. The process of claim 79 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of: manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
81. The process of claim 79 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R17 R'17 R2, R'2, R3,
R'37 R / R' / 5/ R'5/ Re/ R'ε/ R7 / R'7/ Rs/ R'β/ 9 and R'9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rλ or R' and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R ' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R ' and Rx or R'1# R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rλ and R'17 R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R1; R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'g, R7, R'7, R8, R'8, R9, and R'9 together with a different one of R, R', R17 R'17 R2, R'2, R3, R'3, R4, R'4, R5, R's, R6, R'6, R7, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl, phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof; and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate, 90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins .
82. The process of claim 79 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
83. The process of claim 79 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
84. The process of claim 80, 81, 82, or 83 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
85. The process of claim 80, 81, 82, or 83 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
86. The process of claim 80, 81, 82, or 83 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
87. The process of claim 64 wherein the unmodified biomaterial is a ceramic selected from the group consisting of: hydroxyapatite, tricalcium phosphate, and aluminum-calcium-phosphorus oxide .
88. The process of claim 87 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of: manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron(III) porphyrin complexes.
89. The process of claim 87 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , Rl7 R'x, R2, R'27 R3,
R'3/ R4 / R'4/ R5 / R'5/ e R'ε R7/ R'7/ s / R's R9/ and R'g independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rx or R'x and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R' and Rτ or R'17 R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R5 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rλ and R'17 R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R17 R'lf R2, R'2, R3, R'3, R4, R'4, R5, R'g, R6, R'g, R7, R'7, R8, R'8/ R9/ and R'g together with a different one of R, R', R17 R'17 R27 R'2, R3, R'3, R4, R'47 R5, R's, R6, R'g, R7, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y- wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins .
90. The process of claim 87 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
91. The process of claim 87 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
92. The process of claim 88, 89, 90, or 91 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
93. The process of claim 88, 89, 90, or 91 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
94. The process of claim 88, 89, 90, or 91 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
95. The process of claim 64 wherein the unmodified biomaterial is a polymer selected from the group consisting of: polyurethane, polyureaurethane, polyalkylene glycols, polyethylene teraphthalate, ultra high molecular weight polyethylene, polypropylene, polyesters, polyamides, polycarbonates, polyorthoesters, polyesteramides, polysiloxane, polyolefins, polytetrafluoroethylene, polysulfones, polyanhydrides, polyalkylene oxides, polyvinyl halides, polyvinyledene halides, acrylic, methacrylic, polyacrylonitrile, polyvinyl, polyphosphazene, polyethylene-co-acrylic acid, silicone, block copolymer of any of the foregoing polymers, random copolymers of any of the foregoing polymers, graft copolymers of any of the foregoing polymers, crosslinked polymers of any of the foregoing polymers, hydrogels, and mixtures of any of the foregoing polymers .
96. The process of claim 95 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of: manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
97. The process of claim 95 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R17 R'1# R2, R'27 R3,
R'3/ 4 / '4/ R5 / R'5/ ε / R'ε R7/ R'7/ e/ R'β/ 9 an- R'9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; R__ or R' __ and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R's, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R' and Rx or R'1# R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R1, Rx and R'17 R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g/ R7 nd R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R1, R17 R'17 R2, R'2, R3, R'3, R4, R'4, R5, R's, R6, R'g, R7, R'7, R8, R'8, R9, and R'9 together with a different one of R, R', R1# R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'g, R7, R'7, R8 , R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl, borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phtualic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
100 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
105 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins .
98. The process of claim 95 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
99. The process of claim 95 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
100. The process of claim 96, 97, 98, or 99 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
101. The process of claim 96, 97, 98, or 99 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
102. The process of claim 96, 97, 98, or 99 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
103. The process of claim 64 wherein the unmodified biomaterial is a biopolymer selected from the group consisting of: chitin, chitosan, cellulose, methyl cellulose, hyaluronic acid, keratin, fibroin, collagen, elastin, and saccharide polymers.
104. The process of claim 103 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of: manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
105. The process of claim 103 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R
17 R'
17 R
2, R'
2/ R
3# R'3/ 4 / '4 R5 / R'5/ Re/ 'ε/ R7 / R'7/ Rs R's/ R9
aπd R'
9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; R or R' ■__ and R
2 or R'
2, R
3 or R'
3 and R
4 or R'
4 R
5 or R'
5 and R
6 or R'
6, R
7 or R'
7 and R
8 or R'
8, and R
9 or R'
9 and R or R ' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R ' and R
λ or R'
lf R
2 or R'
2 and R
3 or R'
3, R
4 or R'
4 and R
5 or R'
5, R
6 or R'
6 and R
7 or R'
7, and R
8 or R'
8 and R
9 or R'
9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', R
x and R'
17 R
2 and R'
2, R
3 and R'
3, R
4 and R'
4, R
5 and R'
5, R
6 and R'g, R
7 and R'
7, R
8 and R'
8, and R
9 and R'
9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R
17 R'
17 R
2, R'
2, R
3, R'
3, R
4, R'
4, R
5, R'
5, R
6, R'g, R
7, R'
7, R
8 , R'
8, R
9, and R'
9 together with a different one of R, R
1, R
17 R'
17 R
2, R'
2, R
3, R'
3, R
4, R'
4, R
5, R'
57 R
6, R'
6, R
7, R'
7, R
8 , R'
8, R
9, and R'
9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamin , arylamino, heterocycloalkyl amino, heterocycloaryl ammo, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate,
75 thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
106. The process of claim 103 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
107. The process of claim 103 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
108. The process of claim 104, 105, 106, or 107 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
109. The process of claim 104, 105, 106, or 107 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
110. The process of claim 104, 105, 106, or 107 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
111. The process of claim 64 wherein the unmodified biomaterial is a composite material essentially consisting of a relatively inelastic phase selected from the group consisting of: carbon, hydroxy apatite, tricalcium phosphate, silicates, ceramics, and metals, and a relatively elastic phase selected from the group consisting of polymers and biopolymers.
112. The process of claim 111 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of: manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
113. The process of claim 111 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R1# R'17 R2, R'2, R3, R'3/ R4 / R'4/ R5 / R'5/ Re R'β/ R7 / R'7/ Rs / R'β and R'9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rx or R'x and R2 or R'2/ R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'g, and R9 or R'9 and R or R* together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R* and Rx or R'17 R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', R1 and R' 1 / R2 and R ' 2 / R3 and R ' 3 R4 and R ' 4 / R5 and R ' 5 7 Re and R' 6 / R7 and R' 7 / R8 and R ' 8 / and R9 and R ' g , together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R17 R'17 R2, R'2, R3, R'3, R4, R'4,
R5 / R ' 5 / Re / R ' e / R7 , 1 R ' 7 / R8, R'8, R9, and R'9 together with a different one of R , R 7 Ri / ' i / 2 / R ' 2 / R3 / R'3/ R4
RR'' 44 /, R55/, RR''55/, RR66z, RR'' 6g /, R77 /, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl, phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ; and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate, 90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
114. The process of claim 111 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
115. The process of claim 111 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
116. The process of claim 112, 113, 114, or 115 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent,
117. The process of claim 112, 113, 114, or 115 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
118. The process of claim 112, 113, 114, or 115 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
119. The process of claim 64 wherein the biomaterial is conjugated with a precursor ligand of a non-proteinaceous catalyst for the dismutation of superoxide, the process further comprising inserting a cation into the precursor ligand by reacting the biomaterial modified with the precursor ligand with a compound containing a transition metal selected from the group consisting of: manganese, and iron; said reaction yielding a biomaterial conjugated with an active non- proteinaceous catalyst for the dismutation of superoxide,
120. A process for producing a biomaterial modified by co-polymerization with at least one non-proteinaceous catalyst for the dismutation of superoxide or at least on ligand precursor of a non-proteinaceous catalyst for the dismutation of superoxide, the process comprising: a. providing at least one monomer; b. providing at least one least one non- proteinaceous catalyst for the dismutation of superoxide or at least one ligand precursor of a non-proteinaceous catalyst for the dismutation of superoxide containing at least one functional group capable of reaction with the monomer and also containing at least one functional group capable of propagation of the polymerization reaction; c. copolymerizing the monomers and the non- proteinaceous catalyst for the dismutation of superoxide or the ligand precursor in a polymerization reaction.
121. The process of claim 120 wherein the functional group capable of reaction with the monomer and the functional group capable of propagation of the polymerization reaction are the same functional group.
122. The process of claim 120 wherein the functional group capable of reaction with the monomer is selected from the group consisting of: amino (-NH2), carboxyl (-OCOH) , isocyanate (-NCO), mercapto (-SH), hydroxy (-OH) , silyl chloride (-SiCl2) , alkene (-C=CH2] and alkenyl halide (-C=CHX wherein X= Cl , F, Br, I) .
123. The process of claim 120 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of: manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
124. The process of claim 120 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R
17 R'
17 R
2, R'
2, R
3, '3/
4 R 4/ R5 / R 5/ Rε / 'e/ 7 / R'7/ Re/ R's/ R9 /
and R'
9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; R
x or R\ and R
2 or R'
2, R
3 or R'
3 and R
4 or R'
4, R
5 or R'
5 and R
6 or R'
6, R
7 or R'
7 and R
8 or R'g, and R
9 or R'
9 and R or R ' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R' and R
τ or R'
x, R
2 or R'
2 and R
3 or R'
3, R
4 or R'
4 and R
5 or R'
5, R
6 or R'
6 and R
7 or R'
7, and R
8 or R'g and R
9 or R'
9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', R
x and R' __ , R
2 and R'
2 , R
3 and R'
3, R
4 and R'
4, R
5 and R'
5, R
6 and R'g, R
7 and R'
7, R
8 and R'
8, and R
9 and R'
9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R
17 R'
x, R
2, R'
2, R
3, R'
3, R
4, R'
4, R
5, R'
5, R
6, R'g/ R
7/ R'
7/ R
8/ R's/ Rg
and RS together with a different one of R, R', R
17 R'
x, R
2, R'
2, R
3, R'
3, R
4, R'
4, R
5, R'
5, R
6, R'g, R
7, R'
7, R
8, R'
8, R
9, and R'
9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl, phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate,
75 thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
125. The process of claim 120 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
126. The process of claim 120 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
127. The process of claim 123, 124, 125, or 126 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
128. The process of claim 123, 124, 125, or 126 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
129. The process of claim 123, 124, 125, or 126 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
130. The process of claim 120 wherein the monomers are selected from the group consisting of alkylenes, vinyls, vinyl halides, vinyledenes, diacids, acid amines, diols, alcohol acids, alcohol amines, diamines, ureas, urethanes, phthalates, carbonic acids, orthoesters, esteramines, siloxanes, phosphazenes, olefins, alkylene halides, alkylene oxides, acrylic acids, sulfones, anhydrides, acrylonitriles, saccharides, and amino acids.
131. The process of claim 130 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of: manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron(III) porphyrin complexes.
132. The process of claim 131 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R17 R'1# R2, R'2 R3,
R'3/ R4 R'4 R5/ R's/ Rδ/ R' 6 / R7/ R'7/ Rs / R's/ Rg / and R' 9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rx or R'x and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R' 9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R* and Rτ or R'17 R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'g and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rλ and R' ! , R2 and R' 2 , R3 and R' 3 , R4 and R'4, R5 and R'5, R6 and R'g, R7 and R' 7, R8 and R' 8 , and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R1, R17 R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'g, R7, R'7, R8 , R'8, R9, and R'9 together with a different one of R, R', R17 R'17 R2, R'2, R3, R'3, R4,
R'4, R5, R'5, R6, R'g, R7, R'7, R8 , R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl, phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof; and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate, bromite, hypobromite, tetrahalomanganate, tetrafluoroborate, hexafluorophosphate, hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate, saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins .
133. The process of claim 130 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
134. The process of claim 130 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
135. The process of claim 131, 132, 133, or 134 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent,
136. The process of claim 131, 132, 133, or 134 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
137. The process of claim 131, 132, 133, or 134 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
138. The process of claim 120 wherein the biomaterial is co-polymerized with a precursor ligand of a non-proteinaceous catalyst for the dismutation of superoxide, the process further comprising inserting a cation into the precursor ligand by reacting the biomaterial modified with the precursor ligand with a compound containing a transition metal selected from the group consisting of: manganese and iron; said reaction yielding a biomaterial co-polymerized with an active non- proteinaceous catalyst for the dismutation of superoxide.
139. A process for producing a biomaterial modified by admixture with at least one non-proteinaceous catalyst for the dismutation of superoxide or a precursor ligand of a non-proteinaceous catalyst for the dismutation of superoxide, the process comprising: a. providing at least one unmodified biomaterial; b. providing at least one non-proteinaceous catalyst for the dismutation of superoxide or at least one ligand precursor of a non-proteinaceous catalyst for the dismutation of superoxide; and c. admixing the unmodified biomaterial and the non- proteinaceous catalyst for the dismutation of superoxide or the ligand precursor.
140. The process of claim 139 further comprising heating the constituents in order to melt at least one unmodified biomaterial constituent.
141. The process of claim 139 further comprising providing during admixture a solvent in which at least one the unmodified biomaterial and the non-proteinaceous catalyst for the dismutation of superoxide or the ligand precursor are soluble .
142. The process of claim 141 further comprising removing the solvent after admixing.
143. The process of claim 142 wherein said solvent removal is effected by a method selected from the group consisting of evaporation and membrane filtration.
144. The process of claim 139 wherein the biomaterial is admixed with a precursor ligand of a non- proteinaceous catalyst for the dismutation of superoxide, the process further comprising inserting a cation into the precursor ligand by reacting the biomaterial modified with the precursor ligand with a compound containing a transition metal selected from the group consisting of: manganese and iron; said reaction yielding a biomaterial admixed with an active non-proteinaceous catalyst for the dismutation of superoxide.
145. The process of claim 139 wherein the admixed constituents form a solution.
146. The process of claim 139 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
147. The process of claim 139 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R17 R'17 R2, R'2, R3,
R' R4 , R' R,, R' R« R' δ 7 R' RQ R' 8 / R--, and R', independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rλ or R\ and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R's and R6 or R's, R7 or R'7 and R8 or R'g, and R9 or R'9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R1 and Rλ or R'17 R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rλ and R'17 R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R1, R17 R'x, R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'g, R7, R'7/ R8/ R's/ Rg/ and R'9 together with a different one of R, R1, R17 R'17 R27 R'2, R3, R'3, R4,
R'4, R5, R'g, R6, R'g, R7, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate, bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate, saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins .
148. The process of claim 139 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
149. The process of claim 139 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
150. The process of claim 146, 147, 148, or 149 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent,
151. The process of claim 146, 147, 148, or 149 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
152. The process of claim 146, 147, 148, or 149 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
153. The process of claim 139 wherein the unmodified biomaterial is selected from the group consisting of: ceramics, polymers, biopolymers, and composites thereof.
154. The process of claim 139 wherein the unmodified biomaterial is a ceramic selected from the group consisting of: hydroxyapatite, tricalcium phosphate, and aluminum-calcium-phosphorus oxide.
155. The process of claim 154 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
156. The process of claim 154 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R17 R'17 R2, R'2, R3, R'3/ R4 / R'4/ R5/ R'5/ Rδ R'e R7 '7/ Re 8 Rg and R 9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; R or R'x and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R's and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R* and Rx or R'17 R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to tne included carbon atoms of the macrocycle are absent; R and R', Rx and R' x , R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R' 7, R8 and R' 8 , and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', Ri / ' 1 / 2 / '2/ R3 R 3 R4 R 4 / 5 / R'5/ Re/ R'ε/ R7 / R'7/ R8, R'8, R9, and R'9 together with a different one of R, R' , Rx , R ' 1 , R2 , R ' 2 1 R3 / R'3/ R4 /
R'4/ R5 / R's/ Re R'e R7 R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula - (CH2 ) X-M- ( CH2 ) W-L- ( CH2 ) Z - I - (CH2 ) y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl , alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins .
157. The process of claim 154 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
158. The process of claim 154 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
159. The process of claim 155, 156, 157, or 158 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
160. The process of claim 155, 156, 157, or 158 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
161. The process of claim 155, 156, 157, or 158 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
162. The process of claim 139 wherein the unmodified biomaterial is a polymer selected from the group consisting of: polyurethane, polyureaurethane, polyalkylene glycols, polyethylene teraphthalate, ultra high molecular weight polyethylene, polypropylene, polyesters, polyamides, polycarbonates, polyorthoesters, polyesteramides, polysiloxane, polyolefins, polytetrafluoroethylene, polysulfones, polyanhydrides, polyalkylene oxides, polyvinyl halides, polyvinyledene halides, acrylic, methacrylic, polyacrylonitrile, polyvinyl, polyphosphazene, polyethylene-co-acrylic acid, silicone, block copolymer of any of the foregoing polymers, random copolymers of any of the foregoing polymers, graft copolymers of any of the foregoing polymers, crosslinked polymers of any of the foregoing polymers, hydrogels, and mixtures of any of the foregoing polymers .
163. The process of claim 162 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
164. The process of claim 162 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R1# R'1# R2, R'2, R3, '3/ R4 / R'4/ R5/ R'5/ Rε R'ε/ 7 R'7/ Rs / R'β' g and R'9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rx or R' λ and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R's and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R ' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R* and R or R'17 R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, Rs or R'6 and R7 or R'7, and R8 or R'8 and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R', Rx and R' l r R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R17 R'17 R2, R'2, R3, R'3, R4, R'4, R5, R's, R6, R'g, R7, R'7, R8 , R'8, R9, and R'9 together with a different one of R, R', R17 R'lf R2, R'2, R3, R'3, R4, R'4, R5, R'g, R6, R's, R7, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, 85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
165. The process of claim 162 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
166. The process of claim 162 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
167. The process of claim 163, 164, 165, or 166 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent
168. The process of claim 163, 164, 165, or 166 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
169. The process of claim 163, 164, 165, or 166 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
170. The process of claim 139 wherein the unmodified biomaterial is a biopolymer selected from the group consisting of: chitin, chitosan, cellulose, methyl cellulose, hyaluronic acid, keratin, fibroin, collagen, elastin, and saccharide polymers.
171. The process of claim 170 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
172. The process of claim 170 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R
1# R'
x, R
2, R'
2, R
3,
R'3/ R4 R'4' R5/ R's/ Re R' e/ R7/ R'7/ e / R' δ / R9 and R' 9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rλ or R'x and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R'6, R7 or R'7 and R8 or R'8, and R9 or R'9 and R or R ' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R' and Rλ or R'lf R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'g and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R1, Rλ and R'17 R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', R1# R'17 R2, R'2, R3, R'3, R4, R'4, R5, R'5, R6, R'6, R7, R'7, R8, R'8/ R9/ and R'9 together with a different one of R, R', R1# R'17 R2, R'2, R3, R'3, R4, R'4, R5, R's, R6, R'g, R7, R'7, R8, R'8, R9, and R'9 which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl, phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof;
and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate,
75 thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic
80 acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate,
85 alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate,
90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
173. The process of claim 170 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
174. The process of claim 170 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
175. The process of claim 171, 172, 173, or 174 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
176. The process of claim 171, 172, 173, or 174 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
177. The process of claim 171, 172, 173, or 174 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
178. The process of claim 139 wherein the unmodified biomaterial is a composite material comprising a relatively inelastic phase selected from the group consisting of: carbon, hydroxy apatite, tricalcium phosphate, silicates, ceramics, and metals, and a relatively elastic phase selected from the group consisting of: polymers and biopolymers.
179. The process of claim 178 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese (II) pentaaza complexes, manganese (III) pentaaza complexes, iron (II) pentaaza complexes, iron (III) pentaaza complexes, manganese (II) salen complexes, manganese (III) salen complexes, iron (II) salen complexes, iron (III) salen complexes, manganese (II) porphyrin complexes, manganese (III) porphyrin complexes, iron (II) porphyrin complexes, and iron (III) porphyrin complexes.
180. The process of claim 178 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of manganese and iron chelates of pentaazacyclopentadecane compounds, which are represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R, R' , R17 R'x, R2, R'2, R3,
R'3/ R4 / R'4/ R5/ R'5/ Re / R'δ/ 7 / R'7/ e R's/ R9 and R'9 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; Rx or R' 1 and R2 or R'2, R3 or R'3 and R4 or R'4, R5 or R'5 and R6 or R's, R7 or R'7 and R8 or R'g, and R9 or R'9 and R or R ' together with the carbon atoms to which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R or R' and Rx or R'17 R2 or R'2 and R3 or R'3, R4 or R'4 and R5 or R'5, R6 or R'6 and R7 or R'7, and R8 or R'g and R9 or R'9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R and R1, Rx and R' l t R2 and R'2, R3 and R'3, R4 and R'4, R5 and R'5, R6 and R'g, R7 and R'7, R8 and R'8, and R9 and R'9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R, R', Rlf R'1# R2, R'2, R3, R'3, R4, R'4, R5, R'g, R6, R'g, R7, R'7/ R8, R'8, R9, and R'9 together with a different one of R, R', R1# R'17 R2, R'2, R3, R'3, R4, R'4, R5, R's, R6, R'g/ R7, R'7/ R8 / R's/ Rg/ and R' g which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof ; and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate, 90 bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate, tartrate, salicylate, succinate, citrate, ascorbate,
95 saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins.
181. The process of claim 178 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 1-54 of Table 1.
182. The process of claim 178 wherein the non- proteinaceous catalyst for the dismutation of superoxide is selected from the group consisting of Compounds 16, 27, 38, 40, 42, 43, 51, 52, 53, and 54 of Table 1.
183. The process of claim 179, 180, 181, or 182 wherein the non-proteinaceous catalyst is present at a concentration of about 0.001 to about 25 weight percent.
184. The process of claim 179, 180, 181, or 182 wherein the non-proteinaceous catalyst is present at a concentration of about 0.01 to about 10 weight percent.
185. The process of claim 179, 180, 181, or 182 wherein the non-proteinaceous catalyst is present at a concentration of about 0.05 to about 5 weight percent.
186. A biomaterial modified by a combination of methods selected from the group consisting of the method of claim 64, the method of claim 120, and the method of claim 139.
187. A biocompatible article comprising a biomaterial modified with at least one non-proteinaceous catalyst for the dismutation of superoxide or a ligand precursor of a non-proteinaceous catalyst for the dismutation of superoxide, wherein said catalyst or ligand precursor is presented on a surface of said article .
188. The biocompatible article of claim 187 wherein at least a portion of the article comprising the modified biomaterial is implanted within a mammal .
189. The biocompatible article of claim 187 wherein said surface is exposed to biological fluids.
190. The biocompatible article of claim 187 further comprising at least one other biomaterial modified with at least one non-proteinaceous catalyst for the dismutation of superoxide or a ligand precursor of a non- proteinaceous catalyst for the dismutation of superoxide.
191. The biocompatible article of claim 187, wherein the article is a stent, and the modified biomaterial is a metal .
192. The biocompatible article of claim 187, wherein the article is a nerve growth channel, and the modified biomaterial is a hyaluronic acid ester.
193. The biocompatible article of claim 187, wherein the article is a woven vascular graft, and the modified biomaterial is a polymer.
194. The biocompatible article of claim 190, wherein the article is a cardiac stimulator lead wire, and wherein one modified biomaterial is a metal and one other modified biomaterial is a polymer.
195. A process for making a bisimine intermediate in the synthesis of a transition metal chelated pentaazacyclopentadecane complex having superoxide dismutating activity, said intermediate being represented by the following formula:
wherein M is a cation of a transition metal, preferably manganese or iron; wherein R
2, R
3, R'
3, R
4, R'
4, R
5, R'
5, R
6, R's/ R
7/ R'
7/ Rs/ R's/
R9/
Rιo,
Rιι,
and Ri2 independently represent hydrogen, or substituted or unsubstituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylcycloalkyl, cycloalkenylalkyl, alkylcycloalkyl, alkylcycloalkenyl, alkenylcycloalkyl, alkenylcycloalkenyl, heterocyclic, aryl and aralkyl radicals; R
3 or R'
3 and R
4 or R'
4, R
5 or R'
5 and R
6 or R'
6, R
7 or R'
7 and R
8 or R'
8, together with the carbon atoms to .which they are attached independently form a substituted or unsubstituted, saturated, partially saturated or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; R
2 or and R
3 or R'
3, R
4 or R'
4 and R
5 or R'
s, R
6 or R'g and R
7 or R'
7, and R
8 or R'
B and R
9 together with the carbon atoms to which they are attached independently form a substituted or unsubstituted nitrogen containing heterocycle having 2 to 20 carbon atoms, provided that when the nitrogen containing heterocycle is an aromatic heterocycle which does not contain a hydrogen attached to the nitrogen, the hydrogen attached to the nitrogen as shown in the above formula, which nitrogen is also in the macrocyclic ligand or complex, and the R groups attached to the included carbon atoms of the macrocycle are absent; R
2 , R
3 and R'
3, R
4 and R'
4, R
5 and R'
5, R
6 and R'
6, R
7 and R'
7, R
8 and R'
8, and R
9, together with the carbon atom to which they are attached independently form a saturated, partially saturated, or unsaturated cyclic or heterocyclic having 3 to 20 carbon atoms; and one of R
2,
3 / R '
3 / R
4 , R' 4 / R
5 / R ' s / R-6 / R ' 6 / R
7 , R ' 7 / R
8 , R /
together : wi th a dif ferent one of R
2 / R '
2 ! /
R3 / RS . R
4 , R '
4 /
Rs / R' s / Re / R ' e / R7 , R ' 7 / Rs R ' 8 / Re, / and which is attached to a different carbon atom in the macrocyclic ligand may be bound to form a strap represented by the formula
- (CH2)X-M- (CH2)W-L- (CH2)Z-I- (CH2)y-
wherein w, x, y and z independently are integers from 0 to 10 and M, L and J are independently selected from the group consisting of alkyl, alkenyl, alkynyl, aryl, cycloalkyl, heteroaryl, alkaryl, alkheteroaryl , aza, amide, ammonium, oxa, thia, sulfonyl, sulfinyl, sulfonamide, phosphoryl , phosphinyl , phosphino, phosphonium, keto, ester, alcohol, carbamate, urea, thiocarbonyl , borates, boranes, boraza, silyl, siloxy, silaza and combinations thereof; and combinations thereof; and wherein X, Y and Z are independently selected from the group consisting of halide, oxo, aquo, hydroxo, alcohol, phenol, dioxygen, peroxo, hydroperoxo, alkylperoxo, arylperoxo, ammonia, alkylamino, arylamino, heterocycloalkyl amino, heterocycloaryl amino, amine oxides, hydrazine, alkyl hydrazine, aryl hydrazine, nitric oxide, cyanide, cyanate, thiocyanate, isocyanate, isothiocyanate, alkyl nitrile, aryl nitrile, alkyl isonitrile, aryl isonitrile, nitrate, nitrite, azido, alkyl sulfonic acid, aryl sulfonic acid, alkyl sulfoxide, aryl sulfoxide, alkyl aryl sulfoxide, alkyl sulfenic acid, aryl sulfenic acid, alkyl sulfinic acid, aryl sulfinic acid, alkyl thiol carboxylic acid, aryl thiol carboxylic acid, alkyl thiol thiocarboxylic acid, aryl thiol thiocarboxylic acid, alkyl carboxylic acid (such as acetic acid, trifluoroacetic acid, oxalic acid) , aryl carboxylic acid (such as benzoic acid, phthalic acid) , urea, alkyl urea, aryl urea, alkyl aryl urea, thiourea, alkyl thiourea, aryl thiourea, alkyl aryl thiourea, sulfate, sulfite, bisulfate, bisulfite, thiosulfate, thiosulfite, hydrosulfite, alkyl phosphine, aryl phosphine, alkyl phosphine oxide, aryl phosphine oxide, alkyl aryl phosphine oxide, alkyl phosphine sulfide, aryl phosphine sulfide, alkyl aryl phosphine sulfide, alkyl phosphonic acid, aryl phosphonic acid, alkyl phosphinic acid, aryl phosphinic acid, alkyl phosphinous acid, aryl phosphinous acid, phosphate, thiophosphate, phosphite, pyrophosphite, triphosphate, hydrogen phosphate, dihydrogen phosphate, alkyl guanidino, aryl guanidino, alkyl aryl guanidino, alkyl carbamate, aryl carbamate, alkyl aryl carbamate, alkyl thiocarbamate aryl thiocarbamate, alkyl aryl thiocarbamate, alkyl dithiocarbamate, aryl dithiocarbamate, alkyl aryl dithiocarbamate, bicarbonate, carbonate, perchlorate, chlorate, chlorite, hypochlorite, perbromate, bromate, bromite, hypobromite, tetrahalomanganate, tetrafluoroborate , hexafluorophosphate , hexafluoroantimonate, hypophosphite, iodate, periodate, metaborate, tetraaryl borate, tetra alkyl borate,
90 tartrate, salicylate, succinate, citrate, ascorbate, saccharinate, amino acid, hydroxamic acid, thiotosylate, and anions of ion exchange resins;
said process comprising combining a 2,6 dicarbonyl substituted pyridine, which is represented by the 95 following formula:
and a tetraamine, which is represented by the following formula:
and a transition metal ion, under basic conditions, whereby the tetraamine and the 2,6 dicarbonyl substituted pyridine are cyclized around the transition metal ion to form a bisimine chelated with the transition metal ion.
196. A process for making a transition metal chelated pentaazacyclodecane complex catalyst for the dismutation of superoxide comprising reducing a bisimine produced by the process of claim 195 with ammonium formate in the presence of a palladium catalyst.