WO2006099513A1 - Amine- and phthalocyanine dye-containing ink-jet inks with improved ozone fastness - Google Patents
Amine- and phthalocyanine dye-containing ink-jet inks with improved ozone fastness Download PDFInfo
- Publication number
- WO2006099513A1 WO2006099513A1 PCT/US2006/009423 US2006009423W WO2006099513A1 WO 2006099513 A1 WO2006099513 A1 WO 2006099513A1 US 2006009423 W US2006009423 W US 2006009423W WO 2006099513 A1 WO2006099513 A1 WO 2006099513A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- ink
- jet
- jet ink
- amine additive
- amine
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/30—Inkjet printing inks
- C09D11/32—Inkjet printing inks characterised by colouring agents
- C09D11/328—Inkjet printing inks characterised by colouring agents characterised by dyes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/0008—Organic ingredients according to more than one of the "one dot" groups of C08K5/01 - C08K5/59
- C08K5/0041—Optical brightening agents, organic pigments
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/17—Amines; Quaternary ammonium compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/30—Inkjet printing inks
- C09D11/38—Inkjet printing inks characterised by non-macromolecular additives other than solvents, pigments or dyes
Definitions
- the present invention relates generally to amine-containing ink-jet inks.
- the present invention relates to ink-jet inks having improved ozone fastness when printed on photo media, such as semi-metal oxide or metal oxide-containing porous media.
- image quality of high-resolution images can be a function of both the ink-jet ink used to produce an image, and the print medium upon which the image is printed. Desirable attributes of print quality include saturated colors, high gloss and gloss uniformity, and freedom of grain and coalescence, among other characteristics.
- an ink-jet ink that typically meets this criterion can comprise an ozone fastness enhancing amine.
- the present invention is drawn to an ink-jet ink comprising a metallized phthalocyanine dye and a liquid vehicle carrying the dye.
- the liquid vehicle can include an amine additive present in the ink-jet ink at an effective concentration to cause improvement of ozone fastness. The improvement can be determined by comparing the ozone fastness of the ink-jet ink with a control ink-jet ink that does not include the amine additive.
- a printing system for creating images with improved ozone fastness can comprise a printing medium including an ink- receiving layer coated thereon and an ink-jet ink.
- the ink-receiving layer of the printing medium can include semi-metal oxide or metal oxide particulates.
- the ink-jet ink can be configured for printing on printing medium, and can comprise a metallized phthalocyanine dye and a liquid vehicle carrying the dye.
- the liquid vehicle can include an amine additive that is present in the ink-jet ink at an effective concentration to cause improvement of ozone fastness performance, wherein the improvement is determined by comparing the ozone fastness of the ink-jet ink with a control ink-jet ink that does not include the amine additive.
- a method for printing images with improved ozone fastness can comprise jetting an ink-jet ink onto a printing medium.
- the printing medium can include an ink-receiving layer coated thereon comprising semi-metal oxide or metal oxide particulates.
- the ink-jet ink can comprise a metallized phthalocyanine dye and a liquid vehicle carrying the dye.
- the liquid vehicle can include an amine additive that is present in the ink-jet ink at an effective concentration to cause improvement of ozone fastness performance, wherein the improvement is determined by comparing the ozone fastness of the ink-jet ink with a control ink-jet ink that does not include the amine additive.
- liquid vehicle is defined to include liquid compositions that can be used to carry colorants to a substrate.
- Liquid vehicles are well known in the art, and a wide variety of ink vehicles may be used in accordance with embodiments of the present invention.
- ink vehicles may include a mixture of a variety of different agents, including without limitation, surfactants, solvents, co-solvents, buffers, biocides, viscosity modifiers, sequestering agents, stabilizing agents, and water.
- the liquid vehicle can also carry other additives such as polymers, UV curable materials, plasticizers, and/or co- solvents in certain embodiments.
- the term "metallized" when referring to the phthalocyanine dyes includes dyes that have a transition metal that is chelated, coordinated, or complexed to the dye molecule as an integral part of the dye structure. Metallized dyes do not include dyes that merely include a metal counter ion. For example, DB199Na is a copper phthalocyanine dye having a sodium counter ion. The copper component makes this particular dye a "metallized” dye, whereas the sodium counter ion does not.
- control ink-jet ink refers to ink-jet inks that are prepared in order to determine whether there is an improvement in ozone fastness for amine-containing inks prepared in accordance with embodiments of the present invention.
- a control ink-jet ink is prepared by duplicating an amine additive-containing ink, except for the replacement of the amine additive with an equivalent amount of water.
- the control ink-jet ink is relative to the specific ink-jet ink it is to be tested against.
- the control ink-jet ink is tested against the amine-containing ink-jet ink for ozone fastness by printing both inks identically on a media substrate (such as porous media or other coated media) and testing the printed images in an ozone chamber at a controlled ozone concentration, relative humidity, and temperature. Failure of each printed sample can be defined as a predetermined percentage OD loss.
- the amine additive is said to have improved ozone fastness.
- the comparing of ozone fastness of an amine-containing ink-jet ink with a control ink-jet ink can be carried out by several steps.
- steps include: a) preparing a first printed sample including the ink-jet ink printed on a media substrate (such as commercially available porous media, for example, Epson Premium Glossy Photo Paper); b) preparing a second printed sample including the control ink-jet ink printed on the media substrate so that the second printed sample is substantially identical to the first printed sample; c) placing the first printed sample and the second printed sample in a chamber having a 1 ppm ozone level by volume, a relative humidity of 50%, and a temperature of 30 0 C; and d) comparing time intervals required for each of the first printed sample and the second printed sample to reach failure in the chamber.
- failure can be determined by 30% optical density loss compared to initial optical density.
- a longer time interval to failure of the first printed sample relative to the time interval to failure of the second printed sample indicates improvement in ozone fastness.
- an effective concentration when referring to an amount of the amine additive that can be used to improve of ozone fastness, amounts that cause the amine additive-containing ink-jet ink to outperform a control ink-jet ink are included.
- certain amine additives will work to improve ozone fastness within a concentration range or at other specific levels, and will not improve ozone fastness at other levels.
- an effective concentration of an amine additive to improve ozone fastness includes any functional amount of that amine additive, or combination of amine additives, that may be present in an ink-jet ink which improves ozone fastness.
- association when referring to an interaction between the metallized phthalocyanine dye and the amine additive can include any number of molecular associations, but is associated to a greater degree than mere admixture or contact.
- an amine additive can be molecularly associated with a metallized phthalocyanine dye in the form of a van der Waal force or ionic attraction.
- the association can occur in the ink-jet ink and/or once the ink-jet ink is printed on a media substrate. However, preferably, the association will at least be present once the ink-jet ink has been printed or applied to the media substrate, as it is on the media substrate where ozone fastness becomes an issue.
- associations include covalent bonding, hydrogen bonding, or ionic attraction, such as by complexation, chelation, or salt formation.
- association can be characterized by intercalation or other general attraction.
- solubilizing moieties refers to groups, such as acidic groups, that can be attached to organic compositions in order to improve their solubility in the ink vehicle. Examples include sulfonic acid groups, carboxylic acid groups, quaternary amines, etc.
- a weight range of about 1 wt% to about 20 wt% should be interpreted to include not only the explicitly recited concentration limits of 1 wt% to about 20 wt%, but also to include individual concentrations such as 2 wt%, 3 wt%, 4 wt%, and sub-ranges such as 5 wt% to 15 wt% , 10 wt% to 20 wt% , etc.
- an ink-jet ink composition can comprise a metallized phthalocyanine dye and a liquid vehicle carrying the dye.
- the liquid vehicle can include an amine additive present in the ink-jet ink at an effective concentration to cause improvement of ozone fastness performance. Improvement can be determined by comparing the ozone fastness performance of the ink-jet ink which contains the amine additive to that of a control ink-jet ink that does not include the amine additive.
- a printing system for creating images with improved ozone fastness can comprise a printing medium including an ink- receiving layer coated thereon and an ink-jet ink.
- the ink-receiving layer of the printing medium can include semi-metal oxide or metal oxide particulates.
- the ink-jet ink can be configured for printing on printing medium, and can comprise a metallized phthalocyanine dye and a liquid vehicle carrying the dye.
- the liquid vehicle can include an amine additive present in the ink-jet ink at an effective concentration to cause improvement of ozone fastness performance, wherein the improvement is determined by comparing the ozone fastness of the ink-jet ink with a control ink-jet ink that does not include the amine additive.
- a method for printing images with improved ozone fastness can comprise jetting an ink-jet ink onto a printing medium.
- the printing medium can include an ink-receiving layer coated thereon comprising semi-metal oxide or metal oxide particulates.
- the ink-jet ink can comprise a metallized phthalocyanine dye and a liquid vehicle carrying the dye.
- the liquid vehicle can include an amine additive present in the ink-jet ink at an effective concentration to cause improvement of ozone fastness performance, wherein the improvement is determined by comparing the ozone fastness of the ink-jet ink with a control ink-jet ink that does not include the amine additive.
- the amine additive can be a polyamine additive or a heterocyclic amine additive.
- Polyamines include amines that have from two to about 20 amine groups in one embodiment, and from two to about 10 amines in an alternative embodiment. In still a further embodiment, the polyamines can have from two to about five amines.
- heterocyclic amine additives heterocyclic amines devoid of oxygen within its ring structure exemplify a particular group of materials that provide desirable results, though this observation is not intended to be limiting.
- amine compositions such as polyamines, heterocyclic amines, and other types of amines
- amines can improve ozone fastness of ink-jet inks, particularly when printed on porous media substrates such as semi-metal oxide or metal oxide coated media substrates.
- the amines used can come in a variety of forms, including in the form of a primary amines, secondary amines, tertiary amines, and heterocyclic aliphatic amines.
- the amines typically contain 20 carbons or less, and may contain oxygen, phosphorous, or sulfur.
- additives are particularly useful when added to metallized dye-containing ink-jet inks, such as copper- or nickel-containing dyes.
- metallized dye-containing ink-jet inks such as copper- or nickel-containing dyes.
- One class of dyes that can be used includes phthalocyanine dyes.
- Application of an ink-jet ink to a media substrate in accordance with embodiments of the present invention can be by thermal or piezo ink-jet application processes.
- the amine additive can be present in the ink-jet ink composition at an effective concentration to improve ozone fastness compared to a control ink-jet ink that does not include the amine additive.
- this concentration can be from 0.01 wt% to 10 wt%, though this range is not intended to be limiting. This being stated, ranges should be determined on a case by case basis, depending on vehicle components, specific metallized phthalocyanine dye, and/or specific amine additive selected for use, as would be ascertainable by one skilled in the art after considering the present disclosure. Some specific amine additives may actually have a more narrow functional range, and other may have a broader functional range.
- Exemplary amine compositions that can be used include N-methyl-1 ,3- propanediamine,1 ,10-phenanthroline, diethylenetriamine, triethyleneamine, N,N-diethylethylenediamine, homopiperazine, piperazine, piperidine, piperazine- 2-carboxylic acid dihydrochloride, 4-piperidineethanol, 1-(2-hydroxyethyl) piperidine, 1 ,8-bis-[dimethylamino] naphthalene (proton sponge), N-(2- hydroxyethyl) piperazine-N'-(2 ethane sulfonic acid) sodium salt (HEPES), 4-(2- hydroxyethyl) piperazine-1 -propane sulfonic acid (EPPS), and combinations thereof.
- N-methyl-1 ,3- propanediamine,1 ,10-phenanthroline diethylenetriamine, triethyleneamine, N,N-diethylethylenediamine, homopiperazine, pipe
- each of these amine compositions that are effective for use are polyamines, heterocyclic amines that do not include oxygen within the ring, or both.
- saturated ring structures often perform better than unsaturated ring structures. This being stated, provided the amine additive is effective at reducing ozone fade, such additives are included in accordance with embodiments of the present invention.
- amine(s) can also be used, provided the amine(s) are used in a concentration that improves ozone fastness compared to ink-jet inks prepared identically except for the removal of the amine additive and the replacement of the removed amine additive with water.
- glycine or other amino acids can also be included in certain ink-jet inks with improved ozone fastness.
- the ink-jet ink compositions of the present invention are typically prepared in an aqueous formulation or liquid vehicle which can include water, co-solvents, surfactants, buffering agents, biocides, sequestering agents, viscosity modifiers, humectants, and/or other known additives.
- the liquid vehicle can comprise from about 70 wt% to about 99.9 wt% by weight of the ink-jet ink composition.
- liquid vehicle can also carry polymeric binders, latex particulates, and/or other solids.
- co-solvents can be included in the ink-jet compositions of the present invention.
- Suitable co-solvents for use in the present invention include water soluble organic co-solvents, but are not limited to, aliphatic alcohols, aromatic alcohols, diols, glycol ethers, poly(glycol) ethers, lactams, formamides, acetamides, long chain alcohols, ethylene glycol, propylene glycol, diethylene glycols, triethylene glycols, glycerine, dipropylene glycols, glycol butyl ethers, polyethylene glycols, polypropylene glycols, amides, ethers, carboxylic acids, esters, organosulfides, organosulfoxides, sulfones, alcohol derivatives, carbitol, butyl carbitol, cellosolve, ether derivatives, amino alcohols, and ketones.
- co-solvents can include primary aliphatic alcohols of 30 carbons or less, primary aromatic alcohols of 30 carbons or less, secondary aliphatic alcohols of 30 carbons or less, secondary aromatic alcohols of 30 carbons or less, 1 ,2-diols of 30 carbons or less, 1,3-diols of 30 carbons or less, 1 ,5-diols of 30 carbons or less, ethylene glycol alkyl ethers, propylene glycol alkyl ethers, poly(ethylene glycol) alkyl ethers, higher homologs of poly(ethylene glycol) alkyl ethers, poly(propylene glycol) alkyl ethers, higher homologs of poly ⁇ ropylene glycol) alkyl ethers, lactams, substituted formamides, unsubstituted formamides, substituted acetamides, and unsubstituted acetamides.
- co-solvents that are preferably employed in the practice of this invention include, but are not limited to, 1 ,5-pentanediol, 2- pyrrolidone, 2-ethyl-2-hydroxymethyl-1 ,3-propanediol, diethylene glycol, 3- methoxybutanol, and 1 ,3-dimethyl-2-imidazolidinone.
- Co-solvents can be added to reduce the rate of evaporation of water in the ink-jet to minimize clogging or optimize other properties of the ink such as viscosity, pH, surface tension, optical density, and print quality.
- the co-solvent concentration can range from about 0.1 wt% to about 40 wt%, and in one embodiment is from about 5 wt% to about 15 wt%. Multiple co-solvents can also be used, as is known in the art.
- buffering agents can also be optionally used in the ink-jet ink compositions of the present invention.
- Typical buffering agents include such pH control solutions as hydroxides of alkali metals and amines, such as lithium hydroxide, sodium hydroxide, potassium hydroxide; citric acid; amines such as triethanolamine, diethanolamine, and dimethylethanolamine; hydrochloric acid; and other basic or acidic components which do not substantially interfere with the bleed control or optical density characteristics of the present invention. If used, buffering agents typically comprise less than about 10 wt% of the ink-jet ink composition.
- biocides can be used to inhibit growth of undesirable microorganisms.
- suitable biocides include benzoate salts, sorbate salts, commercial products such as NUOSEPT (Nudex, Inc., a division of HuIs America), UCARCIDE (Union Carbide), VANCIDE (RT Vanderbilt Co.), and PROXEL (ICI Americas) and other known biocides.
- NUOSEPT Nudex, Inc., a division of HuIs America
- UCARCIDE Union Carbide
- VANCIDE RT Vanderbilt Co.
- PROXEL ICI Americas
- surfactants can also be used as are known by those skilled in the art of ink formulation.
- suitable surfactants include alkyl polyethylene oxides, alkyl phenyl polyethylene oxides, polyethylene oxide block copolymers, acetylenic polyethylene oxides, polyethylene oxide (di)esters, polyethylene oxide amines, protonated polyethylene oxide amines, protonated polyethylene oxide amides, dimethicone copolyols, substituted amine oxides, commercial products such as TERGITOLS, SURFYNOLS, ZONYLS, TRITONS, MERPOLS, and combinations thereof.
- the amount of surfactant added to the ink-jet inks of this invention can range from 0 wt% to 10 wt%.
- the ink-jet ink can be configured for application from a thermal ink-jet pen.
- Thermal ink-jet systems are quite different in their jetting properties than piezo ink-jet systems.
- compositions that are effective for use in piezo ink-jet systems are not necessarily effective for use with thermal ink-jet ink systems.
- the converse is not necessarily true.
- polymers that work well with thermal ink-jet systems are more likely to work with piezo systems than wee versa. Therefore, the selection of liquid vehicle or other additives for use with thermal ink-jet systems often requires more care, as thermal ink-jet systems are less forgiving than piezo ink-jet systems.
- the coated print media typically includes a substrate and a porous ink- receiving layer deposited on the substrate.
- the substrate can be paper, plastic, coated paper, fabric, art paper, or other known substrate used in the ink-jet printing arts.
- photobase can be used as the substrate. Photobase is typically a three-layered system comprising a single layer of paper sandwiched by two polymeric layers, such as polyethylene layers.
- inorganic semi-metal or metal oxide particulates, a polymeric binder, and optionally, mordants and/or other porous coating composition agents can be present.
- the inorganic semi-metal or metal oxide particulates can be silica, alumina, boehmite, silicates (such as aluminum silicate, magnesium silicate, and the like), titania, zirconia, calcium carbonate, clays, and combinations thereof.
- the particulates can be alumina, silica, or aluminosilicate.
- Each of these inorganic particulates can be dispersed throughout a porous coating composition, which can be applied to a media substrate to form the porous ink-receiving layer.
- the inorganic particulates are present in the coating composition at from 60 wt% to 95 wt%.
- boehmite can be present in the coating composition at from 85 wt% to 95 wt%
- silica or silicates can be present in the coating composition at from 75 wt% to 85 wt%.
- a polymeric binder In order to bind the inorganic particulates together in the porous coating composition, a polymeric binder is typically included.
- Exemplary polymeric binders that can be used include polyvinyl alcohol including water-soluble copolymers thereof; polyvinyl acetate; polyvinyl pyrrolidone; modified starches including oxidized and etherified starches; water soluble cellulose derivatives including carboxymethyl cellulose, hydroxyethyl cellulose; polyacrylamide including its derivatives and copolymers; casein; gelatin; soybean protein; silyl- modified polyvinyl alcohol; conjugated diene copolymer latexes including maleic anhydride resin, styrene-butadiene copolymer, and the like; acrylic polymer latexes including polymers and copolymers of acrylic and methacrylic acids, and the like; vinyl polymer latexes including ethylene-vinyl acetate copolymers; functional group-modified latexes
- the polymeric binder can be present in the coating composition at from 5 wt% to 40 wt%.
- the polymeric binder can be present at from 3 wt% to 15 wt%.
- the polymeric binder can be present at from 10 wt% to 25 wt%.
- the binder can be polyvinyl alcohol or derivatives thereof.
- the porous ink-receiving layer can also be modified with an ionic binding species or mordant known to interact with a predetermined class of colorants, thereby increasing permanence.
- Typical mordants that can be included in the coating composition (and thus, included in the porous ink- receiving layer) include hydrophilic, water dispersible, or water soluble polymers having cationic groups (amino, tertiary amino, amidoamino, pyridine, imine, and the like). These cationically modified polymers can be compatible with water- soluble or water dispersible binders and have little or no adverse effect on image processing or colors present in the image.
- Suitable examples of such polymers include, but are not limited to, polyquaternary ammonium salts, cationic polyamines, polyamidins, cationic acrylic copolymers, guanidine- formaldehyde polymers, polydimethyl diallylammonium chloride, diacetone acrylamide-dimethyldiallyl ammonium chloride, polyethyleneimine, and a polyethyleneimine adduct with epichlorhydrin.
- porous ink-receiving layer can include anionic surfactants, cationic surfactants, biocides, plasticizers, optical brighteners, viscosity modifiers, leveling agents, UV absorbers, hindered amine stabilizers, anti-ozonants, silane coupling agents, and/or other known additives.
- the semi-metal oxide or metal oxide particulates can be chemically surface-modified using silane coupling agents having functional moieties attached thereto.
- the ink-receiving layer can be a single layer or multilayer coating designed to absorb sufficient quantities of ink to produce high quality printed images.
- the coating composition may be applied to the media substrate to form the ink-receiving layer by any means known to one skilled in the art, including blade coating, air knife coating, rod coating, wire rod coating, roll coating, slot coating, slide hopper coating, gravure, curtain, and cascade coating.
- the ink- receiving layer can be printed on one or both sides of the media substrate.
- the depth of the ink-receiving layer formed by the coating composition can be from about 20 ⁇ m to about 60 ⁇ m.
- the thickness for boehmite- containing coating compositions can be from 40 ⁇ m to 55 ⁇ m, the thickness for silica- or silicate-containing coating compositions can be from 25 ⁇ m to 35 ⁇ m. If applied as a media topcoat, the thickness can range from 0.1 ⁇ m to 10 ⁇ m, and in a more specific embodiment, from 1 ⁇ m to 5 ⁇ m.
- the metallized dyes included a copper phthalocyanine or a nickel phthalocyanine.
- Example 2 Evaluation of ozone fastness of ink-jet inks printed on porous media
- various experimental ink-jet inks which included various metallized dyes and amine additives were compared to corresponding control ink-jet inks which did not contain the amine additive to determine the effectiveness of the amine additive on ozone fastness.
- Epson Premium Glossy Photo Paper was chosen as the porous media for these experiments.
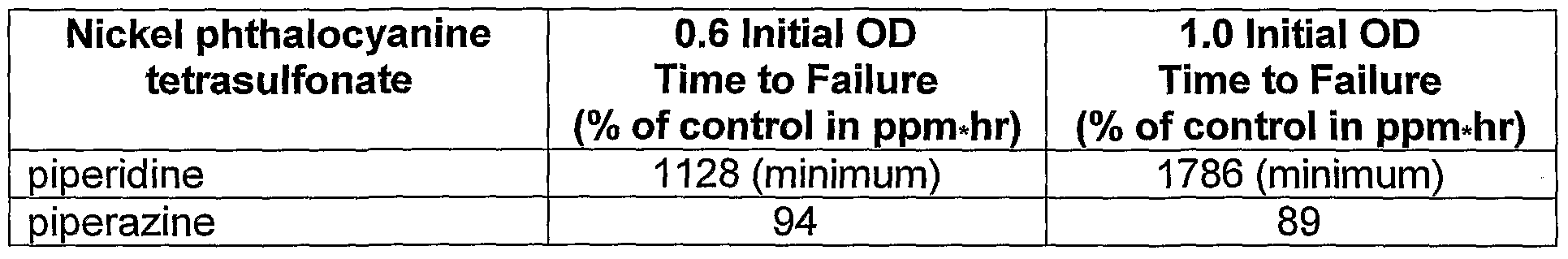
- the ozone fastness was evaluated using a copper j phthalocyanine dye, DB199 Na, with various amine additives.
- the amine compounds which showed effectiveness in improving the ozone fastness were triethylenediamine, N 1 N- diethylethylenediamine, homopiperazine, piperazine, piperidine, piperazine-2- carboxylic acid dihydrochloride, 4-piperidineethanol, 1-(2-hydroxyethyl) piperidine, and proton sponge (1 ,8-bis-[dimethylamino] naphthalene).
- the level of ozone fastness improvement increased at higher amine concentration for certain additives, as illustrated in the examples of piperazine.
- the ink-jet ink demonstrated improved ozone fastness comparing to that of the control ink.
- An even higher level of improvement was attained at the concentration of 1.0 wt %.
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI0608017A BRPI0608017B1 (en) | 2005-03-14 | 2006-03-14 | inkjet ink composition and method for printing ozone-enhanced images |
| DE602006015940T DE602006015940D1 (en) | 2005-03-14 | 2006-03-14 | AMIN AND PHTHALOCYANINE DYEIN INK IRON INK WITH IMPROVED OZONE THICKNESS |
| AT06738483T ATE476481T1 (en) | 2005-03-14 | 2006-03-14 | INKJET INKS CONTAINING AMINE AND PHTHALOCYANINE DYES WITH IMPROVED OZONE FASTNESS |
| JP2008502027A JP5199065B2 (en) | 2005-03-14 | 2006-03-14 | Ink jet ink containing amine and phthalocyanine dyes with improved ozone resistance |
| CA2600803A CA2600803C (en) | 2005-03-14 | 2006-03-14 | Amine- and phthalocyanine dye-containing ink-jet inks with improved ozone fastness |
| CN200680008307XA CN101155885B (en) | 2005-03-14 | 2006-03-14 | Amine- and phthalocyanine dye-containing ink-jet inks with improved ozone fastness |
| EP06738483A EP1877505B1 (en) | 2005-03-14 | 2006-03-14 | Amine- and phthalocyanine dye-containing ink-jet inks with improved ozone fastness |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/080,209 | 2005-03-14 | ||
| US11/080,209 US7435291B2 (en) | 2005-03-14 | 2005-03-14 | Amine- and phthalocyanine dye-containing ink-jet inks with improved ozone fastness |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2006099513A1 true WO2006099513A1 (en) | 2006-09-21 |
Family
ID=36579086
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2006/009423 WO2006099513A1 (en) | 2005-03-14 | 2006-03-14 | Amine- and phthalocyanine dye-containing ink-jet inks with improved ozone fastness |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US7435291B2 (en) |
| EP (1) | EP1877505B1 (en) |
| JP (1) | JP5199065B2 (en) |
| CN (1) | CN101155885B (en) |
| AT (1) | ATE476481T1 (en) |
| BR (1) | BRPI0608017B1 (en) |
| CA (1) | CA2600803C (en) |
| DE (1) | DE602006015940D1 (en) |
| RU (1) | RU2371462C2 (en) |
| TW (1) | TWI397562B (en) |
| WO (1) | WO2006099513A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018057032A1 (en) * | 2016-09-26 | 2018-03-29 | Hewlett-Packard Development Company, L.P. | Aqueous ink compositions |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006063330A (en) * | 2004-07-29 | 2006-03-09 | Canon Inc | Inkjet ink, inkjet recording method, ink cartridge, recording unit and inkjet recorder |
| US20070095250A1 (en) * | 2005-10-28 | 2007-05-03 | Uhlir-Tsang Linda C | Ink Compositions and Methods for Improving Neutrality, Hue Angle and/or Media Independence |
| US7811369B2 (en) * | 2007-01-31 | 2010-10-12 | Hewlett-Packard Development Company, L.P. | Phthalocyanine dyes formulated with salts and anti-flocculent aromatic polyamines for ink-jet imaging |
| US7897203B2 (en) * | 2007-04-23 | 2011-03-01 | Hewlett-Packard Development Company, L.P. | Intercalated compound |
| US7641728B2 (en) * | 2007-04-23 | 2010-01-05 | Hewlett-Packard Development Company, L.P. | Ink composition and method for forming the same |
| US20080265211A1 (en) * | 2007-04-30 | 2008-10-30 | Rehman Zia Ur | Shipping, handling, and testing fluids for ink dispensing systems |
| JP5125701B2 (en) * | 2007-05-07 | 2013-01-23 | コニカミノルタホールディングス株式会社 | Inkjet ink |
| WO2009114684A2 (en) * | 2008-03-12 | 2009-09-17 | Sun Chemical Corporation | Latex paints with improved fade resistance |
| US8673272B2 (en) * | 2009-07-27 | 2014-03-18 | Isp Investments Inc. | Ultraviolet-absorbing compounds |
| CN104419259A (en) * | 2013-09-06 | 2015-03-18 | 北京联创佳艺影像新材料技术有限公司 | Method for processing dye high in fastness to ozone and application of dye in digital ink jet industry |
| AR100280A1 (en) * | 2014-05-22 | 2016-09-21 | Rohm & Haas | POLYMER BINDERS FOR INK JET PRINTERS |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20030079650A1 (en) * | 2001-10-29 | 2003-05-01 | Prasad Keshava A. | Ink-jet inks for improved image quality and reliability |
| EP1308489A1 (en) * | 2001-10-30 | 2003-05-07 | Hewlett-Packard Company | Enhancement of waterfastness using a polyamine/anionic dye mixture with an acidic fixer |
| EP1405883A1 (en) * | 2002-10-02 | 2004-04-07 | Canon Kabushiki Kaisha | Aqueous ink, ink jet recording method, ink tank, recording unit and ink jet recording apparatus |
| EP1616918A1 (en) * | 2004-07-16 | 2006-01-18 | Hewlett-Packard Development Company, L.P. | Metallized dye-based ink-jet inks with improved ozone fastness |
Family Cites Families (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4155768A (en) * | 1977-12-05 | 1979-05-22 | International Business Machines Corporation | Waterfast ink for use in ink jet printing |
| US5026426A (en) * | 1989-09-15 | 1991-06-25 | Hewlett-Packard Company | Ink compositions having improved waterfastness |
| US5100470A (en) * | 1990-10-25 | 1992-03-31 | Hewlett-Packard Company | Waterfast ink formulations for thermal ink-jet using organic amines |
| US5254159A (en) * | 1992-12-17 | 1993-10-19 | Xerox Corporation | Ink compositions |
| GB9322257D0 (en) * | 1993-10-28 | 1993-12-15 | Ciba Geigy Ag | Pigment composition |
| JPH11228883A (en) * | 1998-02-13 | 1999-08-24 | Seiko Epson Corp | Ink and ink jet recording using the same |
| JPH11323226A (en) * | 1998-05-13 | 1999-11-26 | Kao Corp | Aqueous ink |
| JP4009198B2 (en) * | 2001-03-08 | 2007-11-14 | 松下電器産業株式会社 | Ink for ink jet recording, method for producing the ink, and ink cartridge and recording apparatus provided with the ink |
| EP1308488B1 (en) * | 2001-10-30 | 2007-04-11 | Hewlett-Packard Company | Ink-jet inks for improved image quality and reliability |
| US7293867B2 (en) * | 2002-01-24 | 2007-11-13 | Fujifilm Corporation | Ink for ink jet recording, process for the production of ink and ink jet recording process |
| JP3844336B2 (en) * | 2002-01-25 | 2006-11-08 | 富士写真フイルム株式会社 | Ink jet recording ink and ink jet recording method |
| DE60316408T2 (en) * | 2002-02-08 | 2008-06-19 | Fujifilm Corp. | INK AND INK INSERTS FOR INK JET PRINTING AND INK JET PRINTING METHOD |
| JP3849626B2 (en) * | 2002-03-13 | 2006-11-22 | ブラザー工業株式会社 | Water-based ink for inkjet recording |
| JP4072487B2 (en) * | 2002-10-02 | 2008-04-09 | キヤノン株式会社 | Aqueous ink, ink jet recording method, ink tank, recording unit, and ink jet recording apparatus |
| JP4414783B2 (en) * | 2003-03-24 | 2010-02-10 | 富士フイルム株式会社 | Inkjet ink |
| US7704575B2 (en) * | 2003-07-28 | 2010-04-27 | Hewlett-Packard Development Company, L.P. | Additives to eliminate bronzing of ink-jet inks |
| US7052535B2 (en) * | 2003-07-28 | 2006-05-30 | Hewlett-Packard Development Company, L.P. | Additives to eliminate bronzing of inkjet ink formulations on specialty quick-dry inkjet photographic media |
| JP4508706B2 (en) * | 2004-04-09 | 2010-07-21 | キヤノン株式会社 | Inkjet recording method |
| JP2006063330A (en) * | 2004-07-29 | 2006-03-09 | Canon Inc | Inkjet ink, inkjet recording method, ink cartridge, recording unit and inkjet recorder |
| US20070098927A1 (en) * | 2005-10-28 | 2007-05-03 | Uhlir-Tsang Linda C | Ink compositions and methods for controlling color on a print medium |
| US20070095250A1 (en) * | 2005-10-28 | 2007-05-03 | Uhlir-Tsang Linda C | Ink Compositions and Methods for Improving Neutrality, Hue Angle and/or Media Independence |
-
2005
- 2005-03-14 US US11/080,209 patent/US7435291B2/en not_active Expired - Fee Related
-
2006
- 2006-02-14 TW TW095104905A patent/TWI397562B/en not_active IP Right Cessation
- 2006-03-14 AT AT06738483T patent/ATE476481T1/en not_active IP Right Cessation
- 2006-03-14 DE DE602006015940T patent/DE602006015940D1/en active Active
- 2006-03-14 RU RU2007137856/02A patent/RU2371462C2/en not_active IP Right Cessation
- 2006-03-14 BR BRPI0608017A patent/BRPI0608017B1/en not_active IP Right Cessation
- 2006-03-14 JP JP2008502027A patent/JP5199065B2/en not_active Expired - Fee Related
- 2006-03-14 EP EP06738483A patent/EP1877505B1/en not_active Not-in-force
- 2006-03-14 WO PCT/US2006/009423 patent/WO2006099513A1/en active Application Filing
- 2006-03-14 CA CA2600803A patent/CA2600803C/en not_active Expired - Fee Related
- 2006-03-14 CN CN200680008307XA patent/CN101155885B/en not_active Expired - Fee Related
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20030079650A1 (en) * | 2001-10-29 | 2003-05-01 | Prasad Keshava A. | Ink-jet inks for improved image quality and reliability |
| EP1308489A1 (en) * | 2001-10-30 | 2003-05-07 | Hewlett-Packard Company | Enhancement of waterfastness using a polyamine/anionic dye mixture with an acidic fixer |
| EP1405883A1 (en) * | 2002-10-02 | 2004-04-07 | Canon Kabushiki Kaisha | Aqueous ink, ink jet recording method, ink tank, recording unit and ink jet recording apparatus |
| EP1616918A1 (en) * | 2004-07-16 | 2006-01-18 | Hewlett-Packard Development Company, L.P. | Metallized dye-based ink-jet inks with improved ozone fastness |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018057032A1 (en) * | 2016-09-26 | 2018-03-29 | Hewlett-Packard Development Company, L.P. | Aqueous ink compositions |
| US11401429B2 (en) | 2016-09-26 | 2022-08-02 | Hewlett-Packard Development Company, L.P. | Aqueous ink compositions |
Also Published As
| Publication number | Publication date |
|---|---|
| BRPI0608017A2 (en) | 2009-11-03 |
| RU2371462C2 (en) | 2009-10-27 |
| US20060201383A1 (en) | 2006-09-14 |
| CA2600803C (en) | 2011-02-15 |
| TW200641062A (en) | 2006-12-01 |
| JP2008536962A (en) | 2008-09-11 |
| RU2007137856A (en) | 2009-04-20 |
| EP1877505A1 (en) | 2008-01-16 |
| TWI397562B (en) | 2013-06-01 |
| CA2600803A1 (en) | 2006-09-21 |
| DE602006015940D1 (en) | 2010-09-16 |
| CN101155885A (en) | 2008-04-02 |
| CN101155885B (en) | 2012-05-09 |
| US7435291B2 (en) | 2008-10-14 |
| JP5199065B2 (en) | 2013-05-15 |
| EP1877505B1 (en) | 2010-08-04 |
| ATE476481T1 (en) | 2010-08-15 |
| BRPI0608017B1 (en) | 2017-04-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2600803C (en) | Amine- and phthalocyanine dye-containing ink-jet inks with improved ozone fastness | |
| EP1715011B1 (en) | Ink-jet inks containing sulfonated aromatic compounds for reducing ozone fade | |
| EP2762533B1 (en) | Pre-Treatment Coating | |
| EP2762534A1 (en) | Pre-Treatment Coating | |
| TW200902643A (en) | Ink-jet ink formulations containing imidazole | |
| JP2006083387A (en) | Additives to eliminate bronzing of ink-jet inks printed on photo media | |
| JP2006009005A (en) | Additive eliminating bronzing of inkjet ink | |
| EP2951253B1 (en) | Swellable pre-treatment coating | |
| MXPA05006548A (en) | Ink-jet ink and recording material. | |
| US6719833B2 (en) | Recording medium, and image forming method and recorded matter using same | |
| EP1616918B1 (en) | Metallized dye-based ink-jet inks with improved ozone fastness | |
| WO2006012075A1 (en) | Ink jet printing media | |
| JP2005219498A (en) | Modification of weak base of porous inkjet medium coating for upgrading image quality | |
| JP2003128964A (en) | Ink jet recording liquid, method for forming image with the recording liquid and recorded product and ink jet recording liquid/ink jet recording medium set | |
| US8158221B2 (en) | Reactive ink and ink-media system for reduced air fade on porous media | |
| JP2006502885A (en) | How to improve ozone fastness |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200680008307.X Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 3688/CHENP/2007 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2600803 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2008502027 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006738483 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007137856 Country of ref document: RU |
|
| ENP | Entry into the national phase |
Ref document number: PI0608017 Country of ref document: BR Kind code of ref document: A2 |