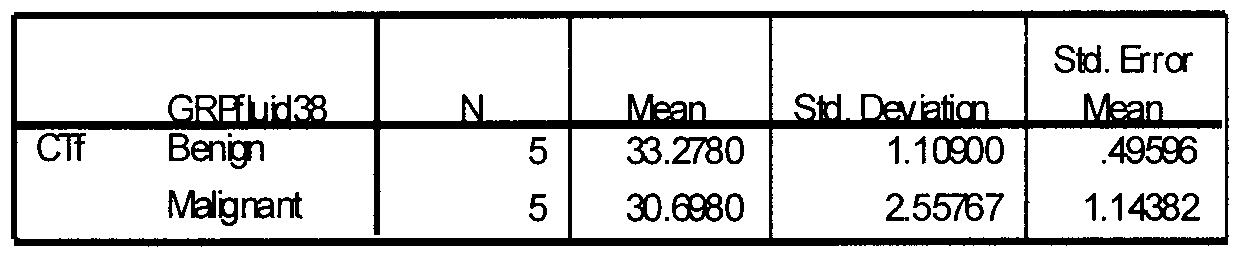WO2009039601A1 - 3.4 kb mitochondrial dna deletion for use in the detection of cancer - Google Patents
3.4 kb mitochondrial dna deletion for use in the detection of cancer Download PDFInfo
- Publication number
- WO2009039601A1 WO2009039601A1 PCT/CA2007/001711 CA2007001711W WO2009039601A1 WO 2009039601 A1 WO2009039601 A1 WO 2009039601A1 CA 2007001711 W CA2007001711 W CA 2007001711W WO 2009039601 A1 WO2009039601 A1 WO 2009039601A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- deletion
- mtdna
- sample
- cancer
- amount
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
- C12Q1/6886—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material for cancer
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6844—Nucleic acid amplification reactions
- C12Q1/6851—Quantitative amplification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6844—Nucleic acid amplification reactions
- C12Q1/686—Polymerase chain reaction [PCR]
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/574—Immunoassay; Biospecific binding assay; Materials therefor for cancer
- G01N33/57407—Specifically defined cancers
- G01N33/57415—Specifically defined cancers of breast
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/574—Immunoassay; Biospecific binding assay; Materials therefor for cancer
- G01N33/57407—Specifically defined cancers
- G01N33/57434—Specifically defined cancers of prostate
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/112—Disease subtyping, staging or classification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/118—Prognosis of disease development
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/156—Polymorphic or mutational markers
Definitions
- This invention is related to the field of mitochondrial genomics. In particular it is related to a 3.4 kb deletion in the mitochondrial genome and its utility as an indicator of cancer.
- DESCRIPTION OF THE PRIOR ART [0003] Mitochondrial DNA (MtDNA) as a Diagnostic Tool
- MtDNA sequence dynamics are important diagnostic tools. Mutations in mtDNA are often preliminary indicators of developing disease, often associated with nuclear mutations, and act as biomarkers specifically related to: disease, such as but not limited to, tissue damage and cancer from smoking and exposure to second hand tobacco smoke (Lee et al., 1998; Wei, 1998); longevity, based on accumulation of mitochondrial genome mutations beginning around 20 years of age and increasing thereafter (von Wurmb, 1998); metastatic disease caused by mutation or exposure to carcinogens, mutagens, ultraviolet radiation (Birch-Machin, 2000); osteoarthritis; cardiovascular, Alzheimer, Parkinson disease (Shoffher et al., 1993; Sherratt et al., 1997;Zhang et al, 1998); age associated hearing loss (Seidman et al., 1997); optic nerve degeneration and cardiac dysrhythmia (Brown et al., 1997; Wallace et al., 1988); chronic progressive external exophthalmoplegia (Taniike et
- Mutations at specific sites of the mitochondrial genome can be associated with certain diseases. For example, mutations at positions 4216, 4217 and 4917 are associated with Leber's Hereditary Optic Neuropathy (LHON) (Mitochondrial Research Society;
- these mutations or alterations include point mutations (transitions, transversions), deletions (one base to thousands of bases), inversions, duplications, (one base to thousands of bases), recombinations and insertions (one base to thousands of bases).
- specific base pair alterations, deletions, or combinations thereof have been found to be associated with early onset of prostate, skin, and lung cancer, as well as aging (e.g. Polyak et al., 1998), premature aging, exposure to carcinogens (Lee et al., 1998), etc.
- Prostate cancer is a frequently diagnosed solid tumour that most likely originates in the prostate epithelium (Huang et al. 1999). In 1997, nearly 10 million American men were screened for prostate specific antigen (PSA), the presence of which suggests prostate cancer (Woodwell, 1999). Indeed, this indicates an even higher number of men screened by an initial digital rectal exam (DRE). In the same year, 31 million men had a DRE (Woodwell, 1999). Moreover, the annual number of newly diagnosed cases of prostate cancer in the United States is estimated at 179,000 (Landis et al., 1999). It is the second most commonly diagnosed cancer and second leading cause of cancer mortality in Canadian men.
- PSA prostate specific antigen
- DRE digital rectal exam
- prostate cancer accounted for 19,800 of newly diagnosed cancers in Canadian men (28%) (National Cancer Institute of Canada). It is estimated that 30% to 40% of all men over the age of forty-nine (49) have some cancerous prostate cells, yet only 20% to 25% of these men have a clinically significant form of prostate cancer (SpringNet - CE Connection, internet, www.springnet.com/ce/j803a.htm). Prostate cancer exhibits a wide variety of histological behaviour involving both endogenous and exogenous factors, i.e. socio-economic situations, diet, geography, hormonal imbalance, family history and genetic constitution (Konishi et al. 1997; Hayward et al. 1998). Although certain mtDNA alterations have been previously associated with prostate cancer, the need exists for further markers for the detection of prostate cancer.
- the 3379 bp deletion (referred to as the 3.4 kb deletion) was determined to be located between nucleotides 10744-14124 of the mitochondrial genome. It was determined that the detection of this deletion could be used in the diagnosis of prostrate cancer when tissue samples are tested.
- the 3.4 kb deletion removes all or part of the following genes from the mtDNA genome: (i) NADH dehydrogenase subunit 4L, (ii) NADH dehydrogenase subunit 4, (iii) NADH dehydrogenase subunit 5, (iv) tRNA histidine, (v) tRNA serine2, and (vi) tRNA Ieucine2.
- Breast cancer is a cancer of the glandular breast tissue and is the fifth most common cause of cancer death. In 2005, breast cancer caused 502,000 deaths (7% of cancer deaths; almost 1% of all deaths) worldwide (World Health Organization Cancer Fact Sheet No. 297). Among women worldwide, breast cancer is the most common cancer and the most common cause of cancer death (World Health Organization Cancer Fact Sheet No. 297). Although certain mtDNA alterations have been previously associated with breast cancer, for example in Parrella et al. (Cancer Research: 61, 2001), the need exists for further markers for the detection of breast cancer.
- the present invention provides a method of detecting a cancer in an individual comprising;
- the present invention provides a method of detecting a cancer in an individual comprising; a) obtaining a biological sample from the individual;
- 21682168.2 b) extracting mitochondrial DNA, mtDNA, from the sample; c) quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence between residues 10743 and 14125 of the mtDNA genome; d) comparing the amount of mtDNA in the sample having the deletion to the amount of the deletion in a reference sample of mtDNA from known non-cancerous tissue or body fluid; wherein an elevated amount of the deletion in the biological sample compared to the reference sample is indicative of cancer.
- the present invention provides a method of detecting a cancer in an individual comprising; a) obtaining a biological sample from the individual; b) extracting mitochondrial DNA, mtDNA, from the sample; c) quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence between residues 10743 and 14125 of the mtDNA genome; d) comparing the amount of mtDNA in the sample having the deletion to the amount of the deletion in a reference sample of mtDNA from known cancerous tissue or body fluid; wherein a similar level of the deletion in the biological sample compared to the reference sample is indicative of cancer.
- the present invention provides a method of monitoring an individual for the development of a cancer comprising; a) obtaining a biological sample; b) extracting mtDNA from the sample; c) quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence between residues 10743 and 14125 of the mtDNA genome; d) repeating steps a) to c) over a duration of time; e) wherein an increasing level of the deletion over the duration of time is indicative of cancer.
- the present invention provides a method of detecting a cancer in an individual comprising; a) obtaining a biological sample from the individual; b) extracting mitochondrial DNA, mtDNA, from the sample;
- Figure 1 is a schematic diagram showing the design and sequence of a primer useful for the detection of the 3.4 kb deletion.
- Figure 2 is a graph showing a comparison of cycle threshold between malignant and symptomatic benign participants in the 3.4 kb study.
- Figure 3 is a graph showing cycle threshold as related to Example 1.
- Figure 4 shows a ROC curve illustrating the specificity and sensitivity of one embodiment of the present invention.
- Figure 5 shows a ROC curve illustrating the specificity and sensitivity of another embodiment of the present invention.
- Figure 6 shows real-time PCR data relating to 3.4kb mtDNA deletion levels associated with breast cancer.
- Figure 7 shows a ROC curve illustrating the specificity and sensitivity of another embodiment of the present invention.
- cycle threshold is the point at which target amplification using real-time PCR rises above background, as indicated by a signal such as a fluorescence signal.
- the C T is inversely related to the quantity of the sequence being investigated.
- sensitivity refers to the fraction of true positives (true positive rate) results obtained using the method of the present invention.
- the present invention may be used for detecting the presence of pre-neoplasia, neoplasia and progression towards potential malignancy of prostate cancer and breast cancer.
- the present invention involves the detection and quantification of the 3.4kb mtDNA deletion (SEQ ID NO: 1) for the detection, diagnosis, and/or monitoring of cancer.
- mtDNA is extracted from a biological sample (for example body tissue, or body fluids such as urine, prostate massage fluid). The extracted mtDNA is then tested in order to determine the levels (ie.
- the levels of the deletion were found to be elevated in samples obtained from subjects with cancer when compared to samples obtained from subjects without cancer. Based on the information and data supplied below, the inventors have concluded that elevated levels of the 3.4 kb deletion in the mtDNA is indicative of cancer.
- the 3.4kb deletion spans approximately nucleotides 10744 to 14124 of the mtDNA genome.
- the mtDNA genome is listed as SEQ ID NO:8 (Genbank accession no. AC 000021).
- the inventors have determined, as provided by example below, that this deletion is also associated with cancer and in particular prostate and breast cancer. Therefore, such deletion provides an accurate biomarker and, therefore, a valuable tool for the detection, diagnosis, or monitoring of cancer in at least these tissues.
- the deletion results in the creation of two deletion monomers, one of 3.4kb in size (small sublimon) and one of approximately 12.6kb in size (large sublimon).
- the occurrence of the deletion may be detected by either identifying the presence of the small sublimon, or by determining that the 3.4 kb sequence has been deleted from the large sublimon.
- the deletion is approximately 3379 bp, and comprises genes encoding NADH dehydrogenase subunit 4L, NADH dehydrogenase subunit 4, NADH dehydrogenase subunit 5, tRNA histidine, ⁇ RNAserine2, and tRNA Ieucine2.
- samples of, for example prostate tissue, prostate massage fluid, urine or breast tissue are obtained from an individual and tested over a period of time
- Age related accumulation of the 3.4 kb mtDNA deletion may predispose an individual to, for example, prostate cancer or breast cancer, which is prevalent in middle aged and older men, and middle aged and older women, respectively.
- a method is provided wherein regular cancer screening may take place by monitoring over time the amount of the 3.4 kb deletion in body tissues such as breast tissue or body fluids such as prostate massage fluid, or urine.
- the system and method of the present invention may be used to detect cancer at an early stage, and before any histological abnormalities.
- the system and method of the present invention may be used to detect pre-neoplasia in breast tissue.
- a pair of amplification primers are used to amplify a target region indicative of the presence of the 3.4 kb deletion.
- one of the pair of amplification primers overlaps a spliced region of mtDNA after deletion of the 3.4 kb sequence has occurred (ie. a splice at a position between 10743 and 14125 of the mtDNA genome). Therefore, extension of the overlapping primer can only occur if the 3.4 kb section is deleted.
- a pair of amplification primers are used to amplify a target region associated with the deleted 3.4 kb sequence.
- the deleted 3.4 kb sequence upon deletion, may reform as a circular mtDNA molecule.
- one of the pair of amplification primers overlaps the rejoining site of the ends of the 3.4 kb sequence.
- 21682168.2 sample is indicative of cancer.
- the below primer pair is preferred for the detection of the deleted 3.4 kb nucleic acid.
- kits for diagnosing cancer for example prostate or breast cancer, comprising means for extraction of mtDNA, primers having the nucleic acid sequences recited in SEQ ID NOS: 2 and 3, or SEQ ID NOS: 9 and 10, reagents and instructions, is provided.
- Another aspect of the invention provides methods for confirming or refuting the presence of a cancer biopsy test from a biopsy sample (eg. prostate or breast cancer), comprising: obtaining non-cancerous tissue from a biopsy sample; and detecting and quantifying the amount of the 3.4 kb mtDNA deletion in the non-diseased tissue.
- a cancer biopsy test from a biopsy sample (eg. prostate or breast cancer)
- obtaining non-cancerous tissue from a biopsy sample comprising: obtaining non-cancerous tissue from a biopsy sample; and detecting and quantifying the amount of the 3.4 kb mtDNA deletion in the non-diseased tissue.
- the present invention provides a method for screening individuals for prostate or breast cancer from a body fluid sample comprising; obtaining a body fluid sample, and detecting and quantifying the level of the 3.4 kb mtDNA deletion in the body fluid.
- the examples provided below illustrate that not only can this deletion be used for the detection of prostate cancer in prostate tissue, but can also be used to detect the presence of cancer in other biological samples, for example prostate massage fluid, urine, and breast tissue. Based on the findings in these examples, the 3.4 kb mtDNA deletion may be used as a biomarker for cancer.
- the amount of the 3.4 kb deletion was found to be higher in the samples obtained from subjects having cancer. This determination was made by comparing the amount of the 3.4 kb deletion in the test samples with amounts from known cancer cells and/or known non-cancer cells.
- Example 1 3.4 kb Deletion in the mtDNA of Prostate Tissue
- a deletion of approximately 3.4 kilobases (kb) was identified through full mitochondrial genome amplification of fresh frozen prostate tissue. Using linear regression, the size of the deletion was estimated to be between 3000 base pairs (bp) and 3500 bp.
- Two possible candidate deletions were identified using MitomapTM (Brandon, M. C, Lott, M. T., Nguyen, K. C, Spolim, S., Navathe, S. B., Baldi, P. & Wallace, D. C, MITOMAP: a human mitochondrial genome database— 2004 update.
- Figure 1 is a schematic diagram showing the design and sequence of the primer (ie. SEQ ID NO: 2). Positive amplification results for the amplicon corresponding to the 3379 bp deletion (referred to as the 3.4 kb deletion) at 10744- 14124 were obtained.
- the 3.4 kb deletion removes all or part of the following genes: (i) NADH dehydrogenase subunit 4L, (ii) NADH dehydrogenase subunit 4, (iii) NADH dehydrogenase subunit 5, (iv) tRNA histidine, (v) tRNA serine2, and (vi) tRNA Ieucine2.
- the 3.4kb deletion was determined to be present in 91% of 33 fresh frozen prostate samples. With the specific deletion primers, formalin fixed tissues were tested in order increase the n value.
- each sample consisted of a mixture of glandular prostate tissue as well as stromal prostate tissue.
- Example 2 3.4kb Deletion Blinded Study - Comparison of Cycle Threshold
- a tapelift was performed on each slide using Prep-Strips (Catalogue Number LCM0207) from Arcturus Bioscience Inc. This allowed the removal of any particulate matter or non-adhering tissue from the slide prior to DNA extraction. With the tissue still on the slides, the slides were rinsed with PBS (Phosphate Buffered Saline Solution) to remove as much fixative as possible. The 1-2 needle biopsy sections on the slides were scraped into sterile microcentrifuge tubes using individually wrapped, sterilized surgical razor blades. DNA was then isolated and purified using a QIAamp ® DNA Mini Kit (Qiagen, Cat. # 51304) according to manufacturer's specifications.
- PBS Phosphate Buffered Saline Solution
- a negative extract control was processed in parallel with the slide extractions as a quality control checkpoint.
- the total concentration of DNA and purity ratio for each sample was determined by spectrophotometry (Nano-DropTM ND- 1000) and dilutions of 2ng/ ⁇ l were prepared for the purpose of Quantitative Polymerase Chain Reaction (qPCR) .
- oligonucleotide primers were chemically synthesized by Invitrogen (California, USA). The sequences of the primers and the expected sizes of the PCR products amplified are listed in Table 1. In addition, PCR analysis for mtDNA deletions included positive controls (DNA from a source known to carry the mutant mtDNA). Each primer set with the exception of TNF (tumor necrosis factor) were checked against a mitochondria-free rho 0 cell line to confirm the absence of pseudogene coamplification.
- TNF tumor necrosis factor
- 5'TAGACTACGTACATACTAACCCTACTCCTA-S' SEQ ID NO: 2 3.4 reverse (14361-14379) 5'-GAGGTAGGATTGGTGCTGT-S' SEQ ID NO: 3 12s forward (708-728 ) 5'-CGTTCCAGTGAGTTCACCCTC-S" SEQ ID NO: 4 12s reverse (923-945) 5 '-CACTCTTTACGCCGGCTTCTATT-S ' SEQ ID NO: 5 TNF forward (3756-3775) 5' -CCTGCCCCAATCCCTTTATT-3' SEQ ID NO: 6 TNF reverse (3866-3886) 5 '-GGTTTCGAAGTGGTGGTCTTG-S 7 SEQ ID NO: 7
- PCRs Three separate PCRs were performed on each sample. Each reaction was 25 ⁇ l total volume and included template DNA, one pair of primers (12s or 3.4 Deletion or TNF ), an iQTM SYBR Green SupermixTM kit (Catalogue Number 170-8882, Bio-Rad Laboratories Inc.) and distilled deionized water (ddH 2 O).
- TNF tumor necrosis factor
- the TNF comprised single copy nuclear gene primers
- 12s comprised total mitochondrial genome primers.
- the volume and concentrations for template DNA, primers, and reaction buffer are listed below.
- the determination of malignancy is based upon the quantity of the 3.4kb deletion present in the normalized sample as indicated by the location of the cycle threshold. This location may be either absolute, as in greater than 25 cycles but less than 35 cycles, or more likely a ratio between the total mitochondrial DNA present as indicated by the 12s amplicon, and the 3.4kb deletion. This may be expressed as a per cent of the total mitochondrial DNA.
- the number of cells, as represented by the TNF amplicon may be incorporated to refine the distinction between benign and malignant tissues.
- Figure 3 shows the actual movement and trends of the data.
- the x-axis is the patient number and the y-axis is the cycle threshold obtained from real time PCR.
- Supervised learning is based on the system trying to predict outcomes for known samples. Half of the data was used to train and the other half to test the algorithm. Supervised learning compares its predictions to the target answer and "learns" from its mistakes. But, if the predicted output is higher or lower than the actual outcome in the data, the error is propagated back through the system and the weights are adjusted accordingly.
- TNF Tumour Necrosis Factor
- Half of data set is used to train the ANN, and the remaining half is used to compare the accuracy.
- DNA was isolated and purified from the samples using a QIAamp ® DNA Mini Kit (Qiagen, Cat. # 51304) according to manufacturer's specifications.
- oligonucleotide primers were chemically synthesized by Invitrogen (California, USA). The sequences of the primers and the expected sizes of the PCR products amplified are listed in Table 1 above.
- the determination of malignancy was based upon the quantity of the 3.4kb deletion present in the normalized sample as indicated by the location of the cycle threshold. This location may be either absolute, as in greater than 25 cycles but less than 30 cycles, or more likely a ratio between the total mitochondrial DNA present as indicated by the 12s amplicon, and the 3.4kb deletion. This may be expressed as a percent of the total mitochondrial DNA.
- Table 6 and figure 7 show the difference in the mean C T scores for samples from malignant tissue and benign tissue.
- the mean C T value for normal tissue was 30.5889, while the mean C T for malignant tissue was 27.8533 thereby illustrating a difference in the quantity of mtDNA having the 3.4 kb deletion in malignant breast tissue compared to normal breast tissue.
- Figure 8 is an ROC curve illustrating the specificity and sensitivity of the 3.4 kb mtDNA deletion as a marker for breast cancer when testing breast tissue. These results were obtained using a cutoff C T of 29.1900. The sensitivity of the marker at this Cj was 77.8%, while the specificity was 77.8%.
- Table 7 shows the calculation of the area under the curve for the present example. As a measure of the accuracy of the test.
- Example 5 The 3.4kb Deletion in the Prostate Massage Fluid of Individuals with Prostate Cancer as Compared to the Fluid from those without Histological Evidence of Prostate Cancer
- Figure 5 is a Receiver Operating Characteristic (ROC) curve illustrating the 6 specificity and sensitivity of the 3.4 kb mtDNA deletion as a marker for prostate cancer when
- the smallest cutoff value is the minimum observed test value -1, and the largest cutoff value is the maximum observed test value plus 1 All the other cutoff values are the average of two consecutive ordered, observed test values
- Example 6 The 3.4kb Deletion in the Urine of Individuals with Prostate Cancer as Compared to the fluid from those without Histological Evidence of Prostate Cancer
- Urine samples were collected from 5 patients who were diagnosed with prostate cancer and 5 who have had a needle biopsy procedure which was unable to detect prostate malignancy. These samples were collected following a digital rectal exam (DRE) to facilitate the collection of prostate cells.
- DRE digital rectal exam
- Figure 6 is a Receiver Operating Characteristic (ROC) curve illustrating the specificity and sensitivity of the 3.4 kb mtDNA deletion as a marker for prostate cancer when testing urine. These results were obtained using a cutoff C T of 31.575. The sensitivity of the marker at this C 7 is 80%, while the specificity is 100%.
- ROC Receiver Operating Characteristic
- T he smallest cutoff value isthe m in im um observed test val ue mi nus 1
- the largest cutoff va lue isthe maximum observed test value pl us 1. All the other cutoff val ues a re the averages of two consecutive ordered observed test val ues.
- Example 7 Detection of Re-circularized 3.4kb Deleted Sequence in Prostate Malignant and Benign Tissue
- the amount of re-circularized 3.4 kb deleted mtDNA molecules in samples was tested as an indicator for prostate cancer.
- the 3.4 kb sequence upon deletion, may reform as a circular mtDNA molecule.
- Amplification of a target region from the deleted 3.4 kb mtDNA sublimon was conducted using a primer pair (SEQ ID NOS: 9 and 10).
- the forward primer (SEQ ID NO: 9) overlaps the rejoining site of the ends of the 3.4 kb sequence.
- Prostate tissue was formalin- fixed paraffin embedded prostate tissue needle biopsies.
- Table 16 below provides a summary of testing conducted for the detection of the actual 3.4 kb deleted in mtDNA obtained from malignant and benign prostate tissue. Using a C T score of 30.0, a clear identification of malignant and benign tissue was possible. As such, an increase in the amount of the 3.4 kb molecule present in a sample was indicative of cancer.
- Birch-Machin MA Online Conference Report (Sunburnt DNA), International Congress of Biochemistry and Molecular Biology, New Scientist, 2000(a)
- MITOMAP A human mt genome database (www.gen.emorv.edu/mitomap.html)
- Woodwell DA National Ambulatory Medical Care Survey: 1997 Summary. Advance data from vital and health statistics; no. 305. Hyattsville, Maryland: National Center for Health Statistics. 1999
Abstract
Description
Claims
Priority Applications (18)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ES07815900.1T ES2484044T3 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb deletion in mitochondrial DNA for use in cancer detection |
| NZ584815A NZ584815A (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
| CA2700941A CA2700941A1 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
| ES13169334.3T ES2539001T3 (en) | 2007-09-26 | 2007-09-26 | Deletion of 3.4 kb mitochondrial DNA for use in cancer detection |
| EP13169334.3A EP2634267B1 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial DNA deletion for use in the detection of cancer |
| AU2007359381A AU2007359381B2 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial DNA deletion for use in the detection of cancer |
| SG2012071478A SG185254A1 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
| EP07815900.1A EP2203570B1 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
| CN200780101671.5A CN101874119B (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial DNA deletion for use in the detection of cancer |
| PCT/CA2007/001711 WO2009039601A1 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
| JP2010526114A JP5518715B2 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial DNA deletion for use in cancer detection |
| KR1020107007603A KR101449562B1 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
| US12/748,120 US20130022979A1 (en) | 2005-04-18 | 2010-03-26 | 3.4kb MITOCHONDRIAL DNA DELETION FOR USE IN THE DETECTION OF CANCER |
| HK10111822.9A HK1145343A1 (en) | 2007-09-26 | 2010-12-17 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer 34kb dna |
| US14/507,027 US20150037804A1 (en) | 2005-04-18 | 2014-10-06 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
| US14/874,155 US20160376660A1 (en) | 2005-04-18 | 2015-10-02 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
| US15/470,175 US10308987B2 (en) | 2005-04-18 | 2017-03-27 | 3.4 kb mitochondrial DNA deletion for use in the detection of cancer |
| US16/429,739 US11111546B2 (en) | 2005-04-18 | 2019-06-03 | 3.4 KB mitochondrial DNA deletion for use in the detection of cancer |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CA2007/001711 WO2009039601A1 (en) | 2007-09-26 | 2007-09-26 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CA2006/000652 Continuation WO2006111029A1 (en) | 2005-04-18 | 2006-04-18 | Mitochondrial mutations and rearrangements as a diagnostic tool for the detection of sun exposure, prostate cancer and other cancers |
| US12/748,120 Continuation US20130022979A1 (en) | 2005-04-18 | 2010-03-26 | 3.4kb MITOCHONDRIAL DNA DELETION FOR USE IN THE DETECTION OF CANCER |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009039601A1 true WO2009039601A1 (en) | 2009-04-02 |
Family
ID=40510689
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CA2007/001711 WO2009039601A1 (en) | 2005-04-18 | 2007-09-26 | 3.4 kb mitochondrial dna deletion for use in the detection of cancer |
Country Status (11)
| Country | Link |
|---|---|
| EP (2) | EP2634267B1 (en) |
| JP (1) | JP5518715B2 (en) |
| KR (1) | KR101449562B1 (en) |
| CN (1) | CN101874119B (en) |
| AU (1) | AU2007359381B2 (en) |
| CA (1) | CA2700941A1 (en) |
| ES (2) | ES2539001T3 (en) |
| HK (1) | HK1145343A1 (en) |
| NZ (1) | NZ584815A (en) |
| SG (1) | SG185254A1 (en) |
| WO (1) | WO2009039601A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2220252A1 (en) * | 2007-11-09 | 2010-08-25 | Genesis Genomics Inc. | Mitochondrial dna deletion between about residues 12317-16254 for use in the detection of cancer |
| WO2010115261A1 (en) | 2009-03-27 | 2010-10-14 | Genesis Genomics Inc. | Aberrant mitochondrial dna, associated fusion transcripts and translation products and hybridization probes therefor |
| US8715960B2 (en) | 2008-03-28 | 2014-05-06 | Mitomics Inc. | Aberrant mitochondrial DNA, associated fusion transcripts and translation products and hybridization probes therefor |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050026167A1 (en) * | 2001-06-11 | 2005-02-03 | Mark Birch-Machin | Complete mitochondrial genome sequences as a diagnostic tool for the health sciences |
| WO2006111029A1 (en) * | 2005-04-18 | 2006-10-26 | Genesis Genomics Inc. | Mitochondrial mutations and rearrangements as a diagnostic tool for the detection of sun exposure, prostate cancer and other cancers |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2450403A1 (en) * | 2001-06-11 | 2002-12-19 | 1304854 Ontario Ltd. | Complete mitochondrial genome sequences as a diagnostic tool for the health sciences |
-
2007
- 2007-09-26 CA CA2700941A patent/CA2700941A1/en not_active Abandoned
- 2007-09-26 CN CN200780101671.5A patent/CN101874119B/en active Active
- 2007-09-26 JP JP2010526114A patent/JP5518715B2/en active Active
- 2007-09-26 EP EP13169334.3A patent/EP2634267B1/en not_active Not-in-force
- 2007-09-26 NZ NZ584815A patent/NZ584815A/en unknown
- 2007-09-26 EP EP07815900.1A patent/EP2203570B1/en active Active
- 2007-09-26 KR KR1020107007603A patent/KR101449562B1/en active IP Right Grant
- 2007-09-26 ES ES13169334.3T patent/ES2539001T3/en active Active
- 2007-09-26 WO PCT/CA2007/001711 patent/WO2009039601A1/en active Application Filing
- 2007-09-26 SG SG2012071478A patent/SG185254A1/en unknown
- 2007-09-26 AU AU2007359381A patent/AU2007359381B2/en not_active Expired - Fee Related
- 2007-09-26 ES ES07815900.1T patent/ES2484044T3/en active Active
-
2010
- 2010-12-17 HK HK10111822.9A patent/HK1145343A1/en unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050026167A1 (en) * | 2001-06-11 | 2005-02-03 | Mark Birch-Machin | Complete mitochondrial genome sequences as a diagnostic tool for the health sciences |
| WO2006111029A1 (en) * | 2005-04-18 | 2006-10-26 | Genesis Genomics Inc. | Mitochondrial mutations and rearrangements as a diagnostic tool for the detection of sun exposure, prostate cancer and other cancers |
Non-Patent Citations (5)
| Title |
|---|
| CANCER RESEARCH, vol. 61, 2001 |
| HE, L. ET AL.: "Detection and quantification of mitochondrial DNA deletions in individual cells by real-time PCR", NUCLEIC ACIDS RESEARCH, vol. 30, no. 14, 15 July 2002 (2002-07-15), pages E68, XP002372964, ISSN: 1362-4962, Retrieved from the Internet <URL:http://nar.oxfordjournals.org/cgi/reprint/30/14/e68> [retrieved on 20080304] * |
| MAKI, J. ET AL.: "Mitochondrial genome deletion aids in the identification of false- and true-negative prostate needle core biopsy specimens", AMERICAN JOURNAL OF CLINICAL PATHOLOGY, vol. 129, no. 1, January 2008 (2008-01-01), pages 57 - 66, XP008132881 * |
| See also references of EP2203570A4 * |
| ZHU, W. ET AL.: "Large-scale mitochondrial DNA deletion mutations and nuclear genome instability in human breast cancer", CANCER DETECTION AND PREVENTION, vol. 28, no. 2, 2004, pages 119 - 126, XP002524596 * |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2220252A1 (en) * | 2007-11-09 | 2010-08-25 | Genesis Genomics Inc. | Mitochondrial dna deletion between about residues 12317-16254 for use in the detection of cancer |
| CN101883864A (en) * | 2007-11-09 | 2010-11-10 | 起源基因组学公司 | Mitochondrial DNA deletion between about residues 12317-16254 for use in the detection of cancer |
| EP2220252A4 (en) * | 2007-11-09 | 2012-08-22 | Mitomics Inc | Mitochondrial dna deletion between about residues 12317-16254 for use in the detection of cancer |
| EP3409793A1 (en) * | 2007-11-09 | 2018-12-05 | MDNA Life Sciences Inc. | Mitochondrial dna deletion between about residues 12317-16254 for use in the detection of cancer |
| US10400290B2 (en) | 2007-11-09 | 2019-09-03 | Mdna Life Sciences, Inc. | Mitochondrial DNA deletion between about residues 12317-16254 for use in the detection of cancer |
| US8715960B2 (en) | 2008-03-28 | 2014-05-06 | Mitomics Inc. | Aberrant mitochondrial DNA, associated fusion transcripts and translation products and hybridization probes therefor |
| US10266899B2 (en) | 2008-03-28 | 2019-04-23 | Mdna Life Sciences Inc. | Aberrant mitochondrial DNA, associated fusion transcripts and hybridization probes therefor |
| WO2010115261A1 (en) | 2009-03-27 | 2010-10-14 | Genesis Genomics Inc. | Aberrant mitochondrial dna, associated fusion transcripts and translation products and hybridization probes therefor |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101874119B (en) | 2015-01-28 |
| HK1145343A1 (en) | 2011-04-15 |
| EP2634267B1 (en) | 2015-02-18 |
| EP2203570B1 (en) | 2014-04-23 |
| EP2203570A4 (en) | 2010-09-01 |
| ES2484044T3 (en) | 2014-08-08 |
| NZ584815A (en) | 2012-07-27 |
| JP2010539908A (en) | 2010-12-24 |
| CA2700941A1 (en) | 2009-04-02 |
| CN101874119A (en) | 2010-10-27 |
| KR101449562B1 (en) | 2014-10-13 |
| SG185254A1 (en) | 2012-11-29 |
| EP2634267A1 (en) | 2013-09-04 |
| ES2539001T3 (en) | 2015-06-25 |
| JP5518715B2 (en) | 2014-06-11 |
| EP2203570A1 (en) | 2010-07-07 |
| AU2007359381B2 (en) | 2015-06-18 |
| KR20100069670A (en) | 2010-06-24 |
| AU2007359381A1 (en) | 2009-04-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7443436B2 (en) | Detection of advanced pancreatic dysplasia | |
| JP6871926B2 (en) | Detecting gastric neoplasms | |
| US11111546B2 (en) | 3.4 KB mitochondrial DNA deletion for use in the detection of cancer | |
| CN107630093B (en) | Reagent, kit, detection method and application for diagnosing liver cancer | |
| US10400290B2 (en) | Mitochondrial DNA deletion between about residues 12317-16254 for use in the detection of cancer | |
| EP2634267B1 (en) | 3.4 kb mitochondrial DNA deletion for use in the detection of cancer | |
| CN116144782A (en) | Combined marker for lung cancer detection and application thereof | |
| WO2014160829A2 (en) | Unbiased dna methylation markers define an extensive field defect in histologically normal porstate tissues associated with prostate cancer: new biomarkers for men with prostate cancer | |
| CN111088358B (en) | Colorectal cancer molecular marker combination, application thereof, primer group and detection kit | |
| US20190382852A1 (en) | Mitochondrial DNA deletion between about residues 12317-16254 for use in the detection of cancer | |
| CN111363817B (en) | Lung cancer diagnostic agent and kit based on HOXD12 gene | |
| CN117106918A (en) | Method for differential diagnosis of benign lung nodules and malignant tumors by gene methylation and kit thereof | |
| CN113249484A (en) | Detection application of mutation number of group of genes as prostate cancer biomarker |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780101671.5 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07815900 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010526114 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2700941 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20107007603 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2680/DELNP/2010 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007815900 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007359381 Country of ref document: AU Ref document number: 584815 Country of ref document: NZ |
|
| ENP | Entry into the national phase |
Ref document number: 2007359381 Country of ref document: AU Date of ref document: 20070926 Kind code of ref document: A |